Neurogenic locus Notch homolog 1 (Notch1), also known as TAN1, acts as a receptor for the membrane-bound ligands Jagged-1 (JAG1), Jagged-2 (JAG2), and Delta-1 (DLL1), regulating cell fate decisions. Upon ligand activation via the released Notch intracellular domain (NICD), Notch forms a transcriptional activation complex with RBPJ/RBPSUH, activating genes at enhancer cleavage sites. This complex influences the execution of differentiation, proliferation, and apoptosis programs. It participates in angiogenesis, negatively regulating endothelial cell proliferation and migration, as well as the formation of angiogenic sprouts.

(Data source: Shi Q, et al. Signal Transduct Target Ther. 2024)
Notch1 expression distribution
Notch1 is widely expressed in cells, mainly in astrocytes, endothelial cells, adipocytes, and can be seen in glandular epithelial cells, squamous epithelial cells, specialized epithelial cells, undifferentiated cells, and immune cells (T cells, B cells, etc.).

(Data source: Uniprot)
Structure and signaling pathway of Notch1
The Notch1 gene is located on chromosome 9q34.3 and encodes the Notch1 protein, which is a type I transmembrane protein. The Notch1 receptor consists of a single transmembrane heterodimer and is divided into multiple functional domains, mainly divided into three regions: the extracellular domain (NECD), the negative regulatory region (NRR); and the intracellular domain (NICD).
NECD is mainly composed of 36 consecutive epidermal growth factor (EGF)-like domains, which are responsible for ligand binding .
The NRR is a simple yet complex system of folded domains that regulate receptor activation and consists of three Lin12/Notch repeats ( LNRs ) spatially protected by a heterodimerization domain (HD).
NICD consists of a protein-binding RBP-Jκ-associated molecule (RAM), seven ankyrin ( ANK) repeats sandwiched between two nuclear localization signals, a transcription activation domain ( TAD ) , and a glutamate, serine, and threonine ( PEST ) domain.
The PEST domain regulates NICD stability and degradation through multiple phospho-signals, and the WSSSSP pattern is recognized by E3 ubiquitin ligases.
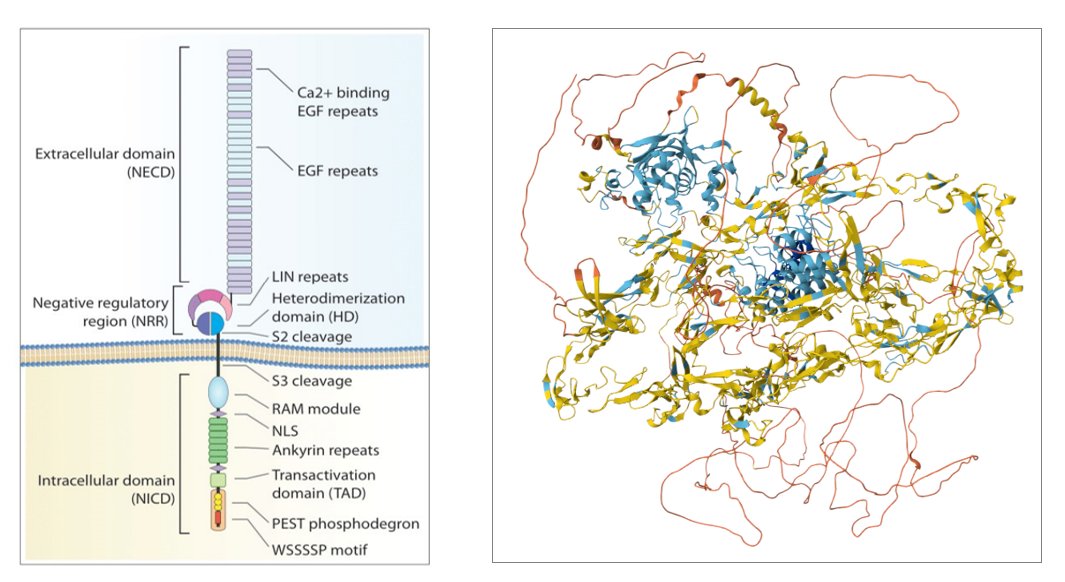
( Data source: Pozzo F, et al. Cancers. 2022 )
Ligand-induced signaling : Ligand-bound NECD is " pulled " toward the signaling cell (the cell expressing the ligand), causing the NRR's three-dimensional protein structure to unfold, exposing the HD. Nearby ADAM metalloproteinases cleave NOTCH1 at the S2 site. The residual TMIC fragment is then readily cleaved at the S3 site by γ-secretase . The released NICD is now free to translocate to the nucleus.
After translocation, NICD interacts with the DNA-binding protein RBP-Jκ/CSL via its RAM domain. Further recruitment of other adaptors, such as MAML ( Mastermind-like ), induces conformational changes, releasing RBP-Jκ-bound corepressors ( SMRT , SKIP , SPEN , HDACs ) , which are then replaced by coactivators ( p300 , PCAF ) to initiate target gene transcription .
Notch1 binds to its ligands in an antiparallel manner via the EGF repeats of the Notch intracellular domain (NECD) . Upon activation by Notch1 ligands JAG1, JAG2, and Delta-1, Notch1 forms a transcriptional activation complex with RBPJ/RBPSUH, activating genes at enhancer cleavage sites. This influences the execution of differentiation, proliferation, and apoptosis programs.
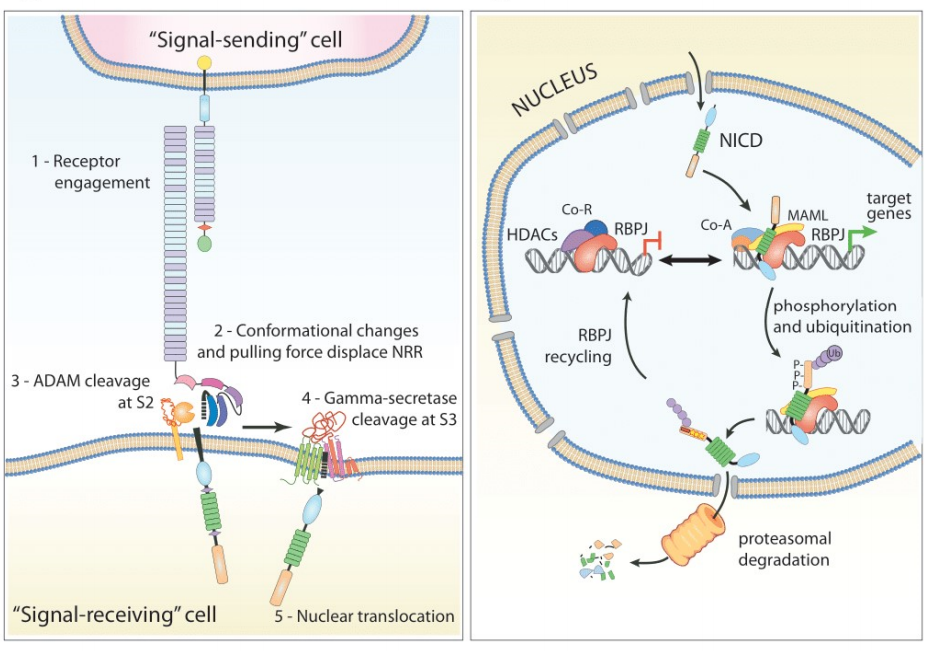
( Data source: Pozzo F, et al. Cancers. 2022 )
Notch1 signaling pathway in cancer
EMT is a complex process in which epithelial cells undergo a transformation, losing their intrinsic properties and adopting a mesenchymal phenotype. The Notch signaling pathway plays a crucial role in regulating EMT and is a key mechanism by which tumor cells acquire malignant properties. Activation of Notch1 not only promotes the EMT process in hepatocellular carcinoma (HCC), enabling it to acquire stem cell-like properties, but also significantly enhances cell migration, invasion, and chemotherapy resistance. Notch1 induces EMT and chemotherapy resistance in TNBC cells by directly activating the MCAM promoter. Downregulation of Notch1 significantly inhibits MCAM expression, thereby reversing EMT and cisplatin chemotherapy resistance in TNBC cells.

(Data source: Shi Q, et al. Signal Transduct Target Ther. 2024)
The role of the Notch signaling pathway in tumor angiogenesis: The Notch signaling cascade has been shown to play a key role in regulating tumor angiogenesis. In drug-resistant non-small cell lung cancer (NSCLC), Notch1 upregulates the hypoxia-inducible factor HIF1α, further promoting vascular endothelial growth factor (VEGF) expression, thereby stimulating angiogenesis. Differentiated glioma cells secrete fibromodulin ( FMOD ), which activates Notch1 signaling through an integrin-dependent pathway, promoting angiogenesis .
DLL4 (Notch ligand) induces Notch1 activation in the tumor microenvironment, triggering the expression of downstream target genes (such as Notch-regulated ankyrin repeat protein NRARP), and synergizing with VEGF to enhance angiogenesis.
Targeting Notch1 or its downstream pathways (such as HIF1α-VEGF) may provide a new strategy for inhibiting tumor angiogenesis.

Notch1 targeted therapy
Targeted therapies for Notch1 mainly target the Notch1 signaling pathway, such as γ-secretase inhibitors ( GSIs ) , ADAM inhibitors , monoclonal antibodies against NOTCH1, direct inhibition of the NOTCH1 transcription factor complex, SERCA inhibitors, etc. Currently, there are few monoclonal antibodies developed for Notch1.

(Data source: Zheng R, et al. Exp Hematol Oncol. 2020)
Notch1 , developed by OncoMed Pharmaceuticals , for the treatment of colorectal cancer, lymphoma , and solid tumors . Brontictuzumab inhibits pathway activation. OMP -52M51 reduced levels of the NOTCH1 intracellular domain in T-ALL cell lines in vitro and significantly inhibited cell growth in T-ALL xenograft models. In a Phase I clinical trial of brontictuzumab in patients with various solid tumor types, patients with ACC showed the best efficacy, with a partial response (PR) rate of 17% and a stable disease (SD) rate of 25% (3/12), lasting for more than six months. NCT03031691 is a study of brontictuzumab combined with chemotherapy in previously treated patients with metastatic colorectal cancer. In 2016, GSK terminated its collaboration with OncoMed for brontictuzumab, and the rights returned to OncoMed and were subsequently acquired by Mereo BioPharma. No updates on this drug are available at this time.

(Data source: clinicaltrials)
