Killer cell lectin-like receptor subfamily G member 1 (KLRG1), also known as CLEC15A, MAFA, and MAFAL, is an immune checkpoint receptor primarily expressed on natural killer (NK) and T cell subsets. It downregulates immune cell activation and proliferation and participates in cell-mediated immune responses. KLRG1's best-characterized inhibitory functions include inducing immune cell death or exhaustion through autophagy, inhibiting cytotoxicity, and suppressing cytokine production. KLRG1 plays a key role in the pathogenesis and progression of autoimmune diseases, infectious diseases, and malignancies, highlighting its potential utility as a promising immune cell marker for disease prediction, diagnosis, and prognosis.
Expression distribution of KLRG1
KLRG1 is mainly expressed in NK cells, granulocytes, and T cells, and is particularly prominent in CD8+ T cells. It is a key marker for cells that have undergone multiple antigen stimulations, are moving towards terminal differentiation and functional exhaustion.

(Data source: uniprot)
Structure of KLRG1 and its receptor
KLRG1 is a type II transmembrane glycoprotein receptor composed of 195 amino acids characterized by an extracellular C-type lectin domain, a transmembrane domain, and an inhibitory motif for a cytoplasmic immunoreceptor tyrosine-based inhibitory domain (ITIM).
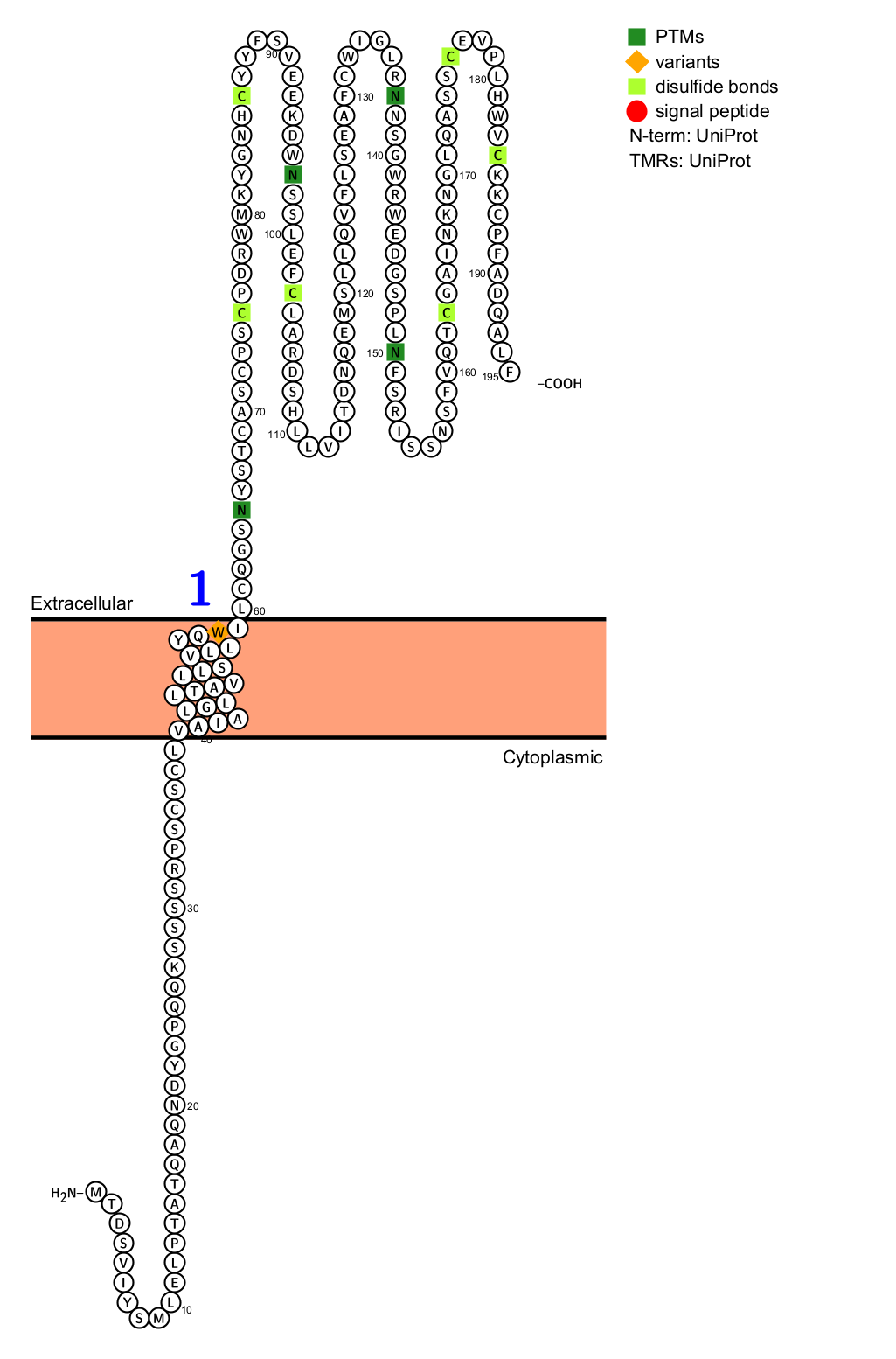
(Data source: uniprot)
Signaling pathway and regulation of KLRG1
T cells, interacts with cadherin ligands expressed on cancer cells or APCs, promoting intracellular phosphorylation of its ITIM domain and subsequently recruiting the tyrosine protein phosphatases SHIP-1 and SHP-2. Unlike PI3K, SHIP-1 and SHP-2 inhibit AKT phosphorylation by degrading PIP3 to PIP2, thereby attenuating activation of the mTOR pathway and leading to decreased T cell effector function and proliferation dysfunction. In HCV- infected CD4+ T cells, increased KLRG1 expression inhibits AKT phosphorylation, thereby activating the transcription factor FOXO and increasing expression of the cell cycle inhibitor p27kip1 or directly activating p16ink4a to inhibit T cell cycle progression. In NK cells, in addition to influencing the AKT pathway, KLRG1 can also be internalized after binding to cadherin ligands. It then directly binds to AMPK, preventing AMPK dephosphorylation through the protein phosphatase PP2C, thereby amplifying AMPK activity and causing loss of NK cell function.
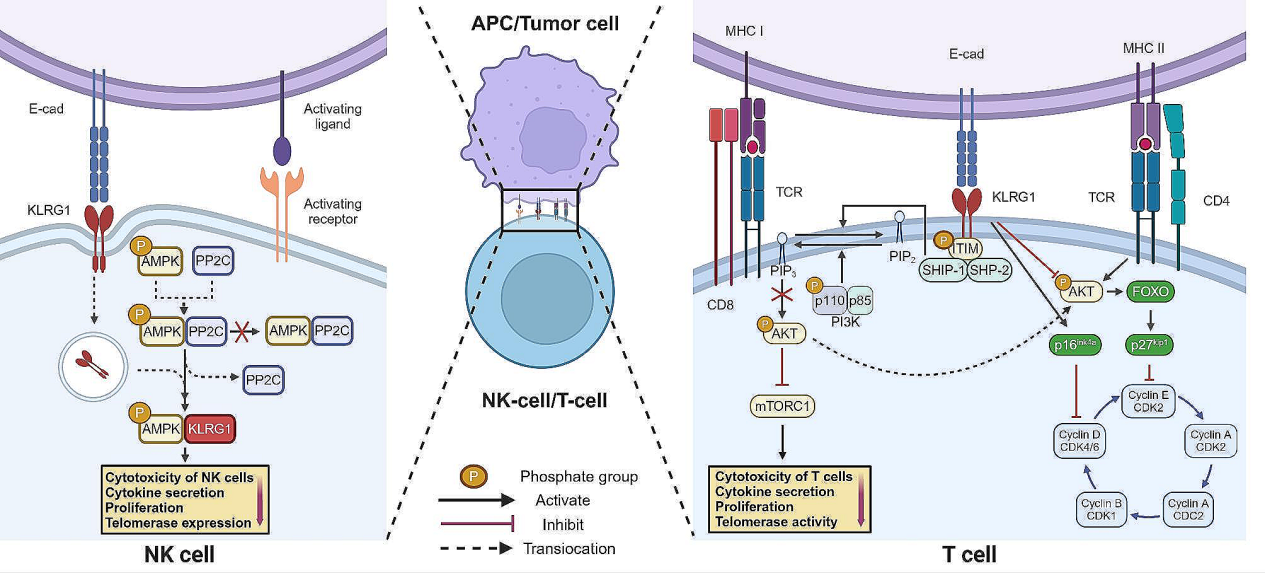
(Data source: Zhang Y, et al. Cell Commun Signal. 2024)
Targeted therapy of KLRG1
Ulviprubart(ABC008 ) is a first-in-class anti-KLRG1 antibody being developed by Abcuro that selectively depletes highly cytotoxic T cells while sparing naïve, regulatory, and central memory T cells. Ulviprubart is designed to treat diseases mediated by highly cytotoxic T cells, including the autoimmune muscle disease inclusion body myositis (IBM), T-cell large granular lymphocytic leukemia (T-LGLL), and mature T-cell malignancies. NCT06450886 is a Phase 3 long-term extension (LTE) study to determine the long-term safety and efficacy of Ulviprubart (ABC008) in subjects with inclusion body myositis.


(Data source: Abcuro official website)
