Integrins play a crucial role in regulating the adhesion and migration of phagocytes to inflamed organs and other immune processes. ITGB2 is a component of the β2 integrin subunit, which is crucial for immune cell function, particularly white blood cells. Integrins help them adhere to blood vessel walls and migrate to sites of infection or injury to initiate immune responses. Mutations in the ITGB2 gene lead to leukocyte adhesion defects, preventing them from effectively exiting the bloodstream and resulting in recurrent bacterial infections.

(Data source: Wen L, et al. Blood. 2022)
Integrin family
Integrins are heterodimeric αβ transmembrane receptors, comprising 17 α subunits and 8 β subunits, making up 24 types of integrins. The β2 integrin family includes four members: αMβ2 , αXβ2 , αLβ2(CD11a&CD18/ITGB2), and αDβ2(CD11d&CD18/ITGB2).

(Data source: Xiaocong P , et al. Signal Transduct Target Ther. 2023)
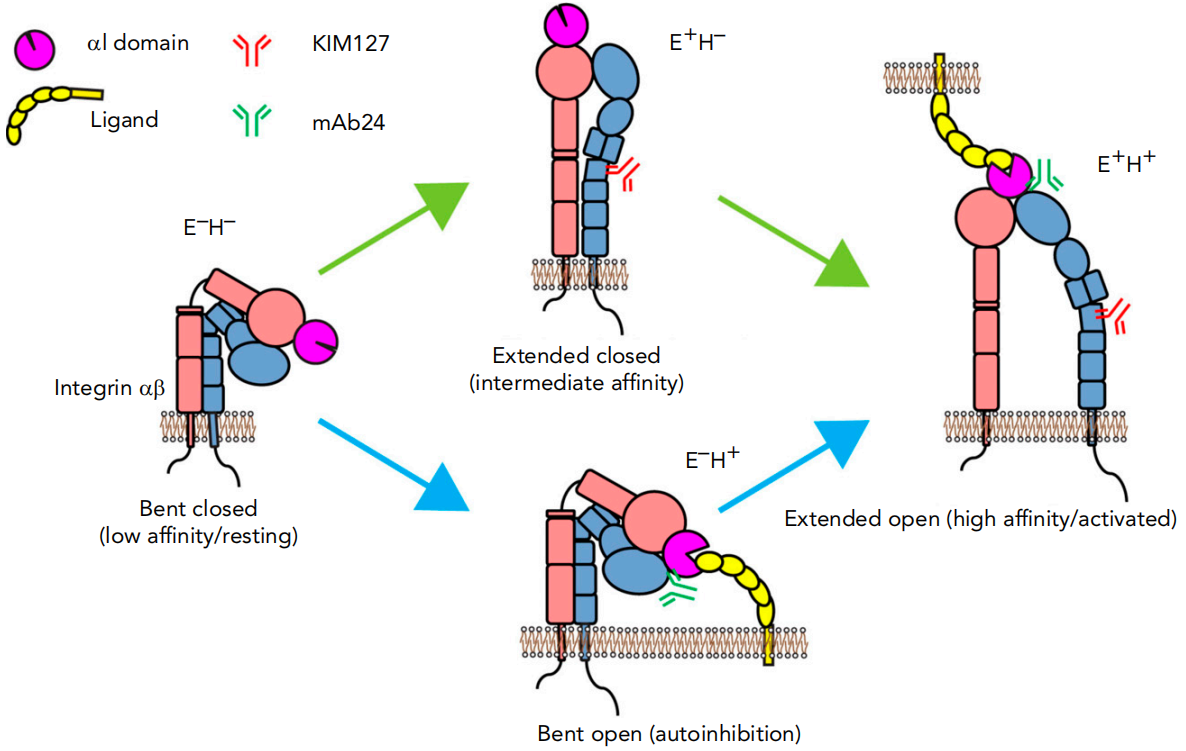
Both the α and β subunits of β2 integrins have long extracellular domains, transmembrane regions (TMDs), and flexible cytoplasmic tails. Integrins adopt at least three conformational states: inactive (curved epitope domain, closed head, curved-closed), intermediate (extended epitope domain, closed head, extended-closed), and activated (extended epitope domain, open head, extended-closed, extended-open). These conformational changes in the extracellular domain rapidly regulate cell adhesion and migration.

(Data source: Hao S , et al. Front Immunol. 2021)
Integrin signaling regulation
Integrin activation is regulated by a variety of external signals, which can be divided into two main types:
"Outside-in" mechanisms: Such signals can be mediated by the ECM, mechanotransduction, or non-ECM ligands (including growth factor receptors, hormones, and small molecules). Binding of ECM or non-ECM ligands and the application of force lead to integrin aggregation, which, through the recruitment of talin and vinculin, initiates downstream signaling to the actin cytoskeleton. Actin can simultaneously pull on integrins and further promote force generation. This "outside-in" mechanism then triggers various signaling cascades, ultimately leading to cell survival, proliferation, cell expansion, and even tumorigenesis and metastasis.
"Inside-out" mechanisms: This regulatory mechanism controls the displacement of integrin inhibitors within the cell and allows talin or kindlin to bind to the β-tail of the integrin, thereby controlling the affinity of the integrin for extracellular matrix components. For example, in neutrophils, talin-1 and kindlin-3 are rapidly recruited to activate β2 integrins induced by extracellular chemokines binding via GPCRs (G protein-coupled receptors).

(Data source: Xiaocong P , et al. Signal Transduct Target Ther. 2023)
Integrin- targeted therapy of ITGB2
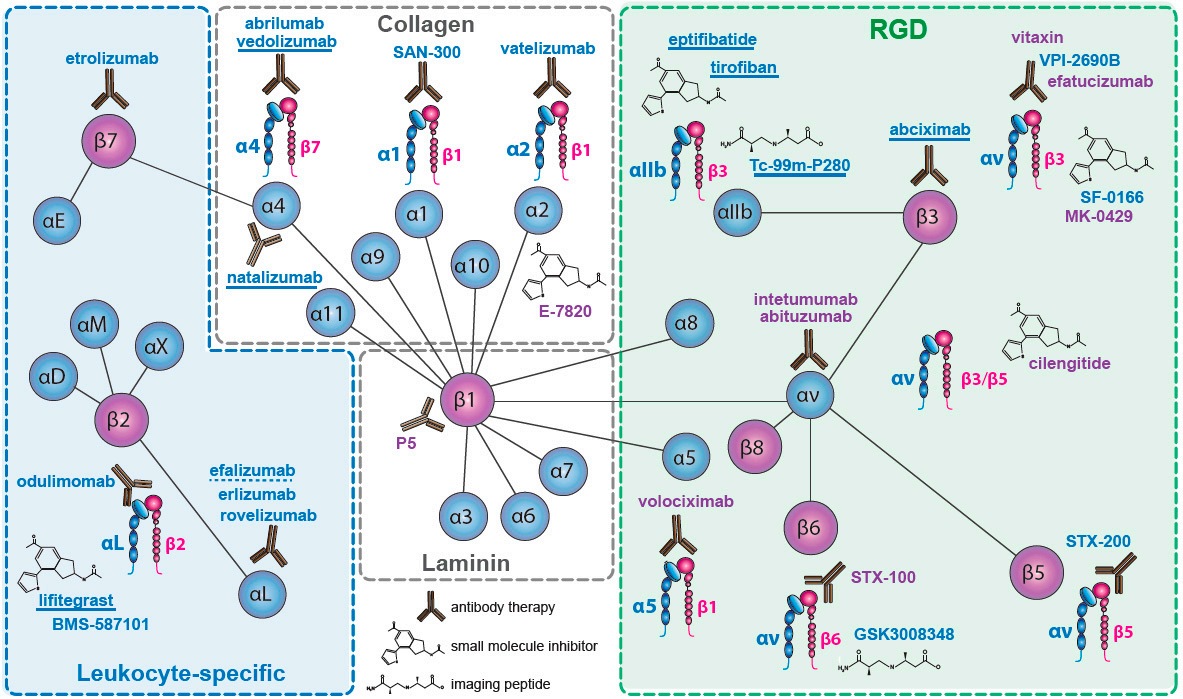
There are two approaches to developing integrin drugs: one is to block the integrin activation state, and the other is to block the integrin activation pathway.
(Data source: Elisabetta B , et al. Biomedicines. 2023)
Early drugs targeting ITGB2 mainly include Rovelizumab (heterodimer co-recognition, Phase 3 trial for cerebral ischemia, did not show clinical benefit on neurological function recovery, and development has been stopped) and Erlizumab (ITGB2 single chain, as a therapy for hemorrhagic shock, failed to reach the endpoint in the Phase 2 trial, and development has been stopped.).
(Data source: Sabine Raab-W , et al. Cancers. 2017)
Although there is currently no clinically advanced ITGB2-targeted antibody therapy, research on the ITGB2 target has not stopped in recent years, including the identification of ITGB2 as a key regulatory factor related to immunity and matrix in glial cell types, which is positively correlated with tumor infiltration of immune and stromal cells and can serve as a potential target for primary glioblastoma multiforme.

(Data source: AVS Sai K , et al. Comput Biol Med. 2023)
Inhibition or knockout of the integrin β2 subunit (ITGB2/CD18) in malignant B cells significantly reduced the cytotoxicity of CD19 and CD20 CAR-T cells against B-lineage tumor cells. ITGB2-mediated cytotoxicity alters the formation of immune synapses within the tumor microenvironment, thereby affecting CAR-T cell-mediated cytotoxicity against B-cell malignancies.

(Data source: Yang S , et al. Cancer Lett. 2025)
