Neurotrophin receptor 1 (NRP1), also known as CD304, is a cell surface receptor involved in cardiovascular development, angiogenesis, the formation of certain neuronal circuits, and the development of organs outside the nervous system. NRP1 is expressed on regulatory T cells (Tregs), promoting their survival and function, while Tregs suppress the activity of immune cells. It plays a crucial role in immune regulation and the tumor microenvironment (TEM).
Expression distribution of NRP1
NRP1 was initially discovered to be expressed in brain neurons and was later found to be widely expressed in various cell types, such as osteoblasts, neurons, immune cells, adipocytes, glomerular interstitial cells, endothelial cells, and hepatic stellate cells. It is expressed in tumors such as leukemia, malignant melanoma, malignant glioma, osteosarcoma (OS), lung cancer, and gastric cancer.
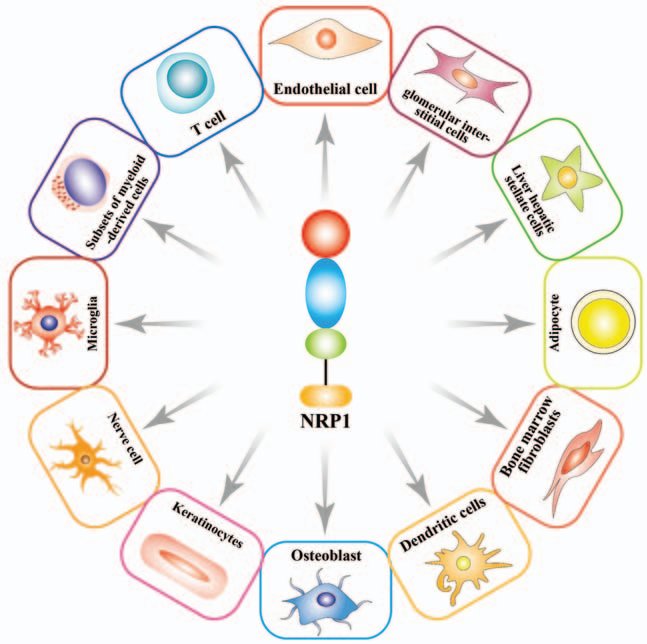
(Data source: Liu SD, et al. Chin Med J (Engl). 2020)
The structure of NRP1 and its receptor
NRP1 is a transmembrane protein located on human chromosome 10q12. It contains 17 exons and 16 introns. It has intracellular, transmembrane, and extracellular domains. The intracellular domain is relatively small, lacks an intrinsic kinase domain, and is not involved in signal transduction. The extracellular domain is composed of five subdomains: a1, a2, b1, b2, and c, each associated with different molecular and/or cellular interactions. The a1a2b1 domains bind to SEMA3, the b1b2 domains bind to vascular endothelial growth factor (VEGF), and the c domain is thought to play a role in NRP1 oligomerization. The C-terminus of NRP contains a three-amino acid sequence (serine-glutamic acid-alanine) called SEA, which binds to kinases through the PDZ domain.
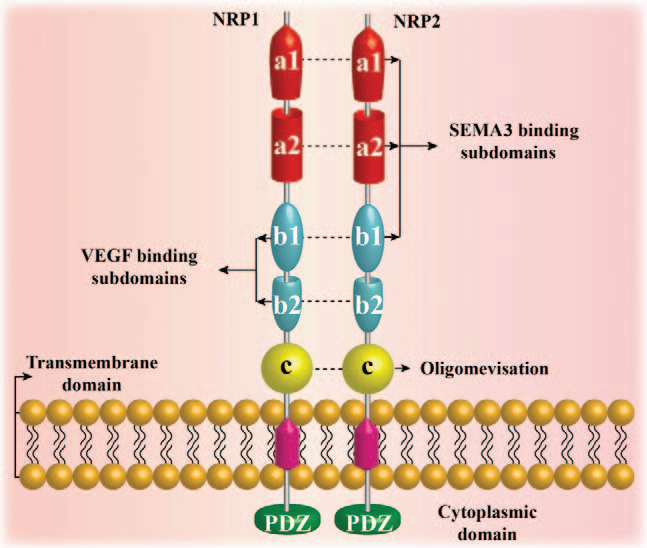
(Data source: Liu SD, et al. Chin Med J (Engl). 2020)
The role of NRP1 in immunity and tumor microenvironment
At the tumor-immune interface, NRP1 promotes immune evasion by orchestrating multiple inhibitory processes within the immune compartment of the TME. On the one hand, the physiological effects of NRP1 in DCs and macrophages, which contribute to the anti-inflammatory phenotype of these cells, are " hijacked " by tumors to promote tumor angiogenesis and tumor-associated immunosuppression. On the other hand, NRP1 has a direct impact on adaptive antitumor immunity by influencing both cell-specific and cell-specific inhibitory pathways that regulate intratumoral Treg cells and CD8+ T cells.
The role of NRP1 in regulatory T cells (Tregs)
Enhance Treg cell stability: NRP1 is highly expressed in Treg cells and maintains the phenotypic and functional stability of Treg cells by interacting with ligands such as Sema4A, enabling them to maintain their immunosuppressive function in the tumor microenvironment.
Promote the recruitment of Treg cells to tumor sites: NRP1 acts as a co-receptor for VEGFR2, helping Treg cells migrate to the tumor area along the VEGF gradient, thereby enhancing immunosuppression at the tumor site.
Maintaining the suppressive function of Treg cells: NRP1 inhibits Akt activity in Treg cells through PTEN, thereby stabilizing the suppressive function of Treg cells. Treg cells lacking NRP1 exhibit functional impairments, such as increased production of IFNγ, which weakens their suppressive function.
Plasticity of Treg cells: NRP1-deficient Treg cells switch from a suppressive phenotype to a pro-inflammatory phenotype, which is of great significance in tumor immunity.
The role of NRP1 in CD8+ T cells
Marking Exhausted CD8+ T Cells: NRP1 is highly expressed in exhausted CD8+ T cells and, together with inhibitory receptors such as PD1, marks these functionally impaired T cells. These cells exhibit decreased migration and cytotoxicity.
Inhibit the cytotoxicity of CD8+ T cells: NRP1 inhibits the migration and cytotoxic function of CD8+ T cells by binding to Sema3A, thereby limiting their ability to kill tumor cells.
Impact on memory formation of CD8+ T cells: NRP1 has a unique effect on memory formation in CD8+ T cells. NRP1-deficient CD8+ T cells are more able to maintain a memory/exhausted precursor cell phenotype, thereby forming a larger pool of tumor-specific memory CD8+ T cells.
These effects indicate that NRP1 plays an important role in tumor immunity by regulating the functions of Treg cells and CD8+ T cells, thereby influencing the immunosuppression and anti-tumor immune response of the tumor microenvironment.
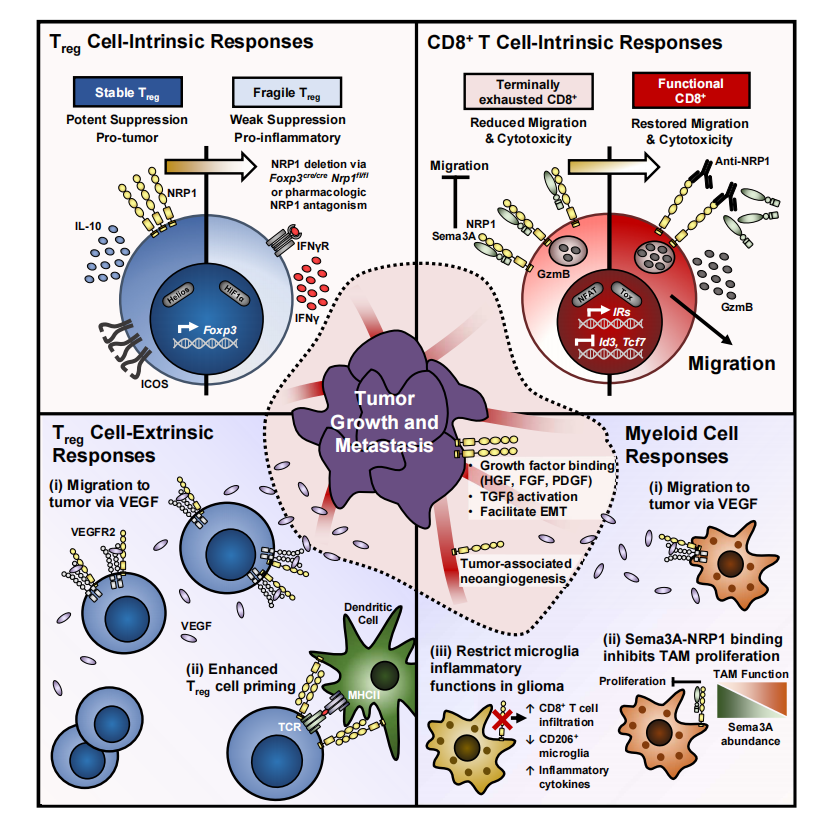
(Data source: Chuckran CA, et al. J Immunother Cancer. 2020)
Targeted therapy for NRP1
Vesencumab is an IgG1 monoclonal antibody targeting NRP1, developed and produced by Genentech, primarily for the treatment of locally advanced malignancies. Vesencumab binds to NRP-1, preventing its interaction with vascular endothelial growth factor receptor 2 (VEGFR-2), thereby interfering with the VEGF-mediated signaling pathway, inhibiting endothelial cell proliferation and angiogenesis, and thereby preventing tumor-associated angiogenesis. Because it interferes with the primary pathway of tumor angiogenesis, vesencumab has potential for treating various solid tumor types, particularly those that are often highly metastatic. However, safety and efficacy issues remain, and no new developments have been reported on this antibody.
Developed by Astellas Pharma Inc., ASP-1948 is a humanized IgG4 monoclonal antibody targeting the immunomodulatory receptor Neuropilin-1 (NRP1). ASP-1948 blocks the interaction between NRP1 and its ligand, thereby reversing the suppressive activity of Tregs, releasing the inhibitory effect on cytotoxic T cells and exerting anti-tumor effects. In 2023, Astellas announced the termination of further development of ASP-1948. In a Phase Ib study, the drug failed to demonstrate clinical benefit in combination with nivolumab across multiple tumor types. A PoC evaluation determined that the drug did not achieve the expected efficacy, leading to the project's termination.
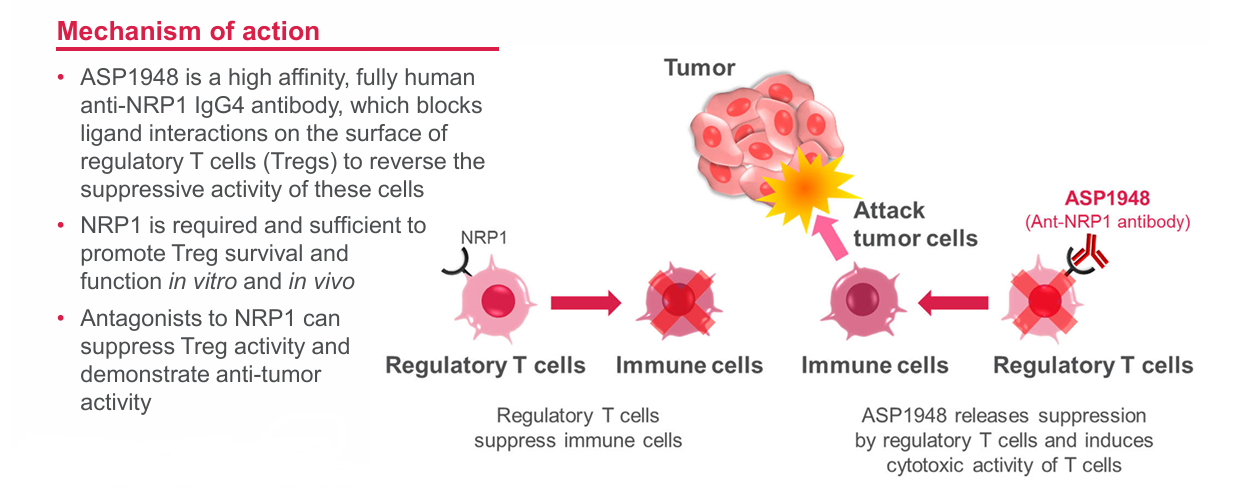
(Data source: Astellas official website)
