The neurogenic locus Notch homolog 3 (Notch3) regulates cell fate as a receptor for the membrane-bound ligands Jagged1, Jagged2, and Delta1. Upon ligand activation via the released Notch intracellular domain (NICD), Notch3 forms a transcriptional activator complex with RBPJ/RBPSUH and activates genes within the cleavage site enhancer, influencing differentiation, proliferation, and apoptosis programs. Mutations in Notch3 are the direct cause of cerebral autosomal dominant arteriopathy with subcortical infarcts and leukoencephalopathy (CADASIL).
Notch3 expression distribution
Under normal physiological conditions, Notch3 is primarily expressed in smooth muscle cells, suprabasal keratinocytes, and squamous epithelial cells. Under pathological conditions, abnormally high Notch3 expression is commonly seen in a variety of malignant tumors (such as glioma, breast cancer, renal cancer, and pancreatic cancer), and is often associated with tumor malignant progression (proliferation, invasion, and metastasis) and poor prognosis.
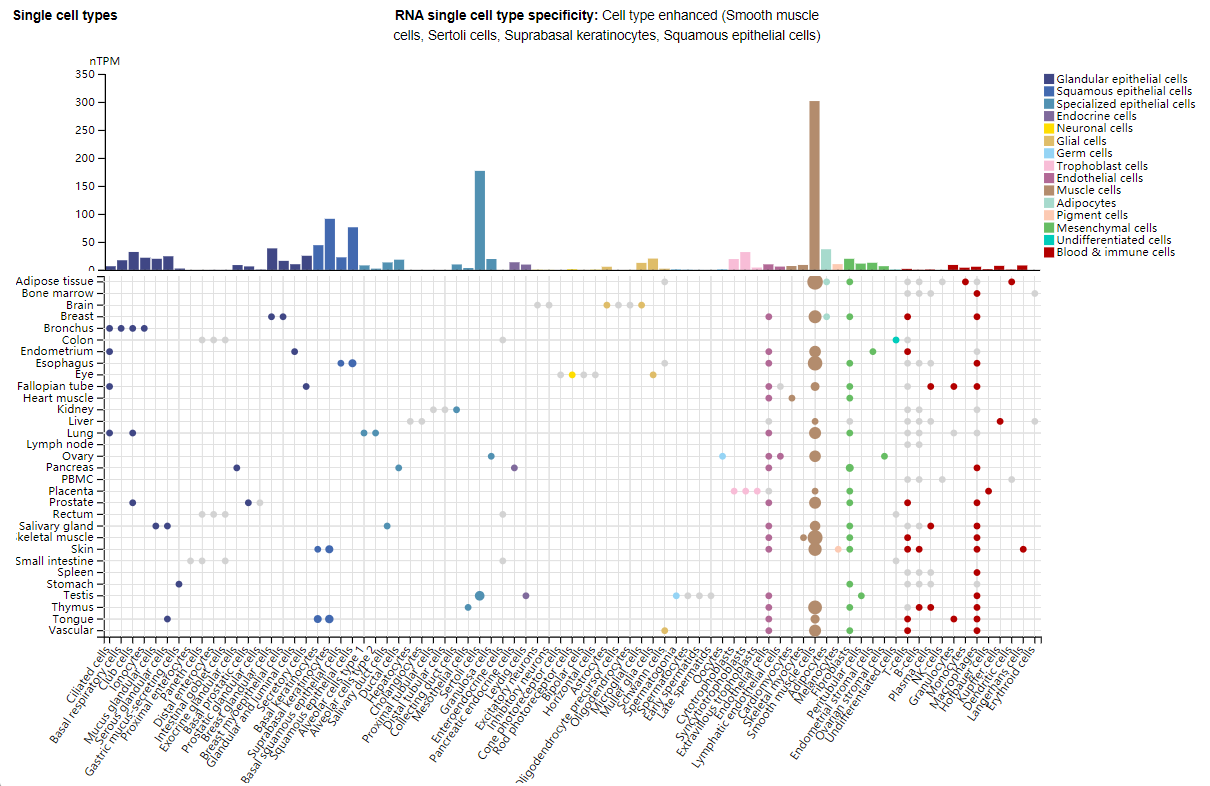
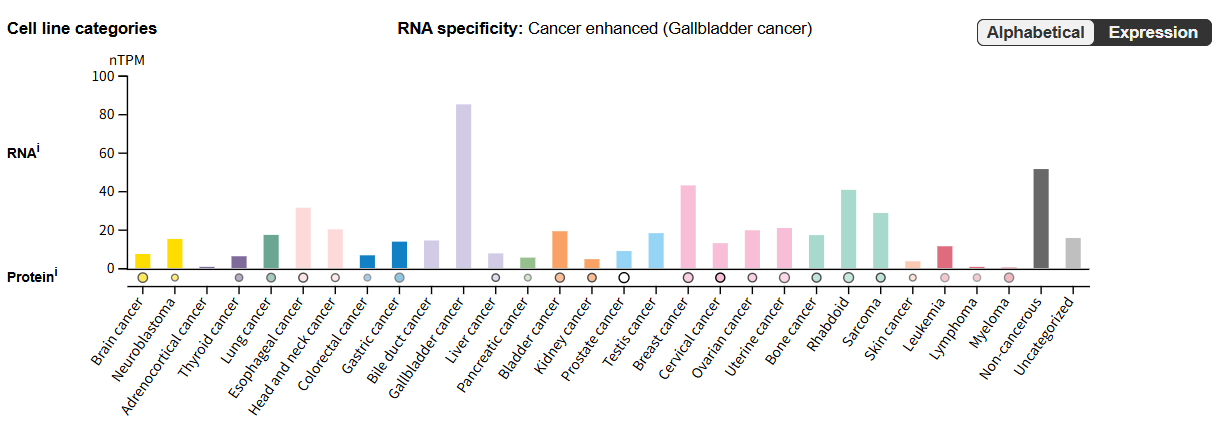
(Data source: Uniprot)
Structure and mutations of Notch3
Notch3 contains 33 exons and encodes a transmembrane protein called Notch3. The Notch3 protein consists of an extracellular domain (ECD) and an intracellular domain (ICD).
EGF repeat domain: The ECD contains 34 EGFr repeats, with the ligand binding site located at EGFr 10-11. Each EGFr domain contains six cysteines with conserved numbers and positions, which form three disulfide bridges that stabilize the EGFr domain and contribute to the tertiary structure of the protein.
LNR domain: Located between the EGF-like repeats and the transmembrane domain, it consists of three cysteine-rich LNR modules. When the receptor is not activated, the LNR region, together with another part of the extracellular domain, forms a " negative regulatory region " (NRR).
Transmembrane region: The S2 site is located at the extracellular end. Upon ligand binding, it undergoes a conformational change and is subsequently cleaved by the ADAM10/17 metalloproteinase. The S3 site, located intracellularly, is cleaved by the γ-secretase complex. This cleavage is crucial for releasing the transcriptionally active intracellular domain (NICD).
Intracellular domain: The cell contains ANK repeat sequences, RAM domain, transcription activation domain (TAD), and PEST sequence. After S2 and S3 cleavage, the intracellular domain (NICD) is released and enters the cell nucleus to perform transcriptional regulatory functions.
Notch3 mutations are concentrated in the exon region encoding the EGF-like repeat sequence. Missense mutations that change cysteine (such as the p.G111C mutation found in EGFr domain 2) result in an unpaired cysteine residue. ScanProsite predicts that this mutation will disrupt disulfide bond formation within the EGFr domain.

(Data source Yuan L, et al. J Adv Res. 2024)
Notch3 signal transduction in diseases
Notch3 gene are the direct cause of CADASIL, an inherited cerebral small vessel disease that primarily affects the small arteries in the brain.
Notch3 receptor undergoes a complex process involving cleavage and activation. In the absence of Notch3 mutations, the Notch3 receptor binds to its ligand. Ligand binding leads to engulfment of the N3 TMIC via clathrin-dependent endocytosis. The N3ICD is then cleaved by γ-secretase and translocated to the nucleus, triggering subsequent activation of Notch3 target genes. In the absence of Notch3, RhoA expression is downregulated, leading to inhibition of Rho kinase and, in turn, reduced myosin phosphorylation. Conversely, mutations in the receptor, particularly in its extracellular domain (p.Cys428Ser and p.Cys455Arg), disrupt this process, impairing ligand binding and, consequently, signal transduction. GOM deposition at the basement membrane involves the accumulation of Notch3 ECD (N3ECD) multimers. Mutated Notch3 triggers the generation of these multimers, promoting their intracellular aggregation (N3ICD). When clearance of these aggregates is impaired, endoplasmic reticulum stress, cell death, and abnormal growth of vascular smooth muscle cells (VSMCs) occur. GOMs may also disrupt VSMC function, potentially affecting periarterial drainage within the intramural arteries and leading to white matter degeneration. The Notch3 signaling pathway regulates vascular tone through interactions with LTBP-1 and TGF-β in GOMs and RhoA, driving CADASIL pathology.

(Data source: Heidari P, et al. J Headache Pain. 2025)
Notch3 targeted therapy
Tarextumab is a fully humanized monoclonal antibody that targets both Notch2 and Notch3 receptors. Early clinical studies in pancreatic cancer have shown promising results, with exploratory analyses suggesting that patients with high Notch3 expression may benefit more. NCT01647828 is a Phase 1b/2 study (ALPINE) of OMP-59R5 (Tarextumab) in combination with nab-paclitaxel and gemcitabine in previously untreated patients with Stage IV pancreatic cancer.
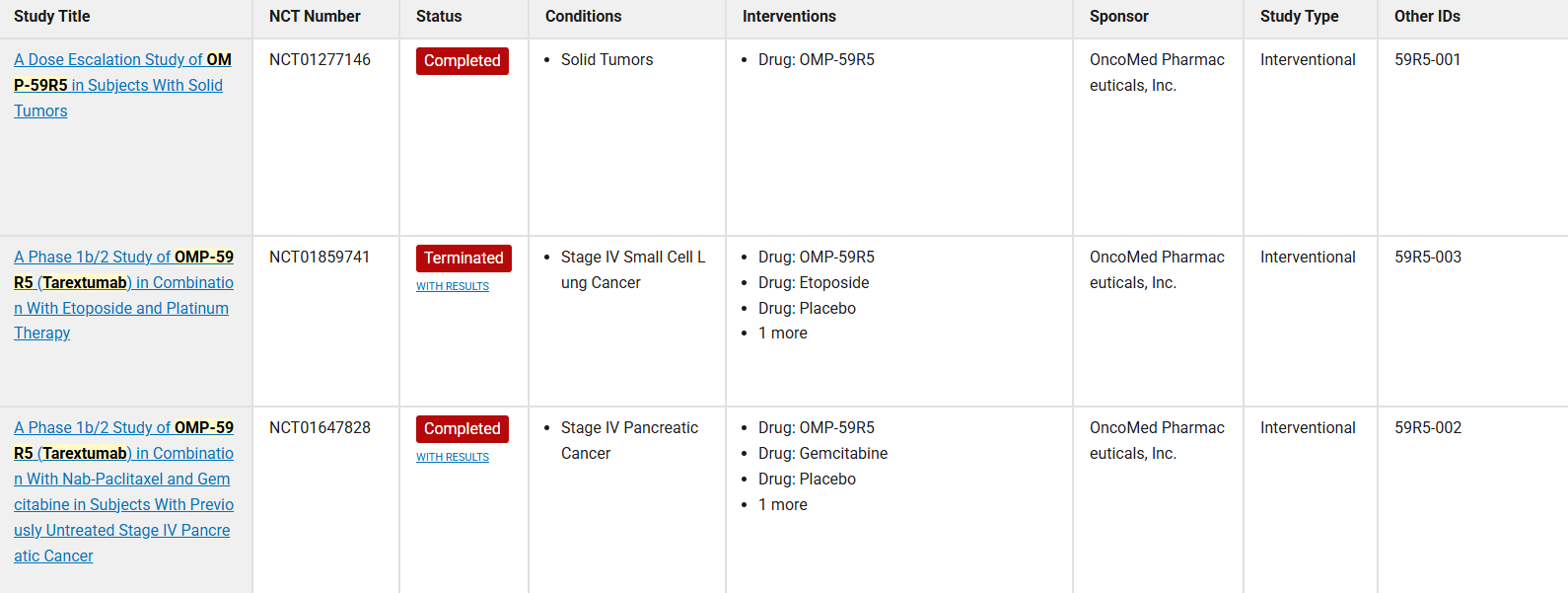
(Data source: clinicaltrials)
AV-353 is a monoclonal antibody targeting Notch3, developed by AVEO Pharmaceuticals for the treatment of multiple myeloma and currently in the preclinical research stage.
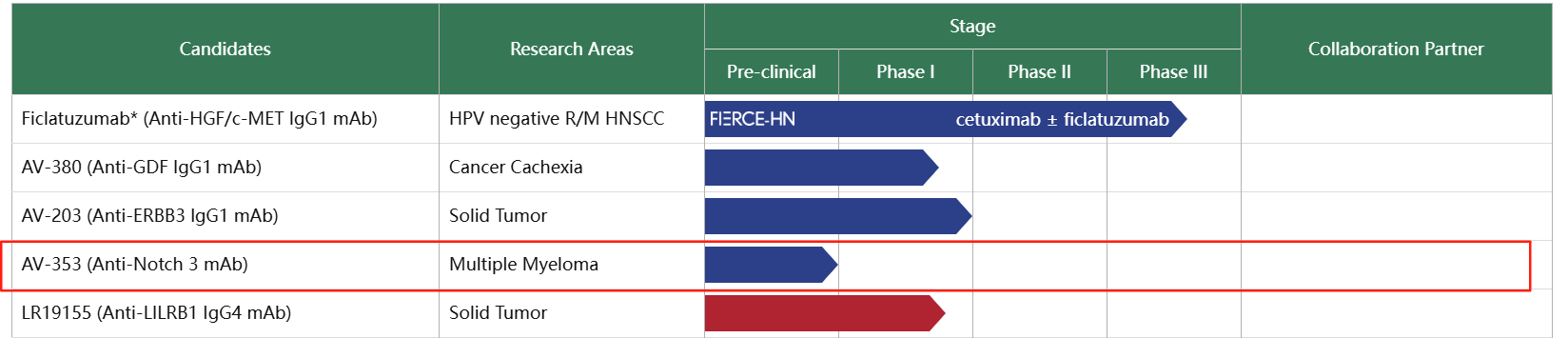
(Data source: AVEO Pharmaceuticals official website)
