Background
The discovery and development of multispecific antibodies presents unique challenges in optimizing their physicochemical properties for developability and manufacturability. Common developability challenges include increased risk of aggregation, high viscosity, poor solubility, low expression yields, complex purification requirements, increased fragmentation propensity, immunogenicity, or pharmacokinetics.

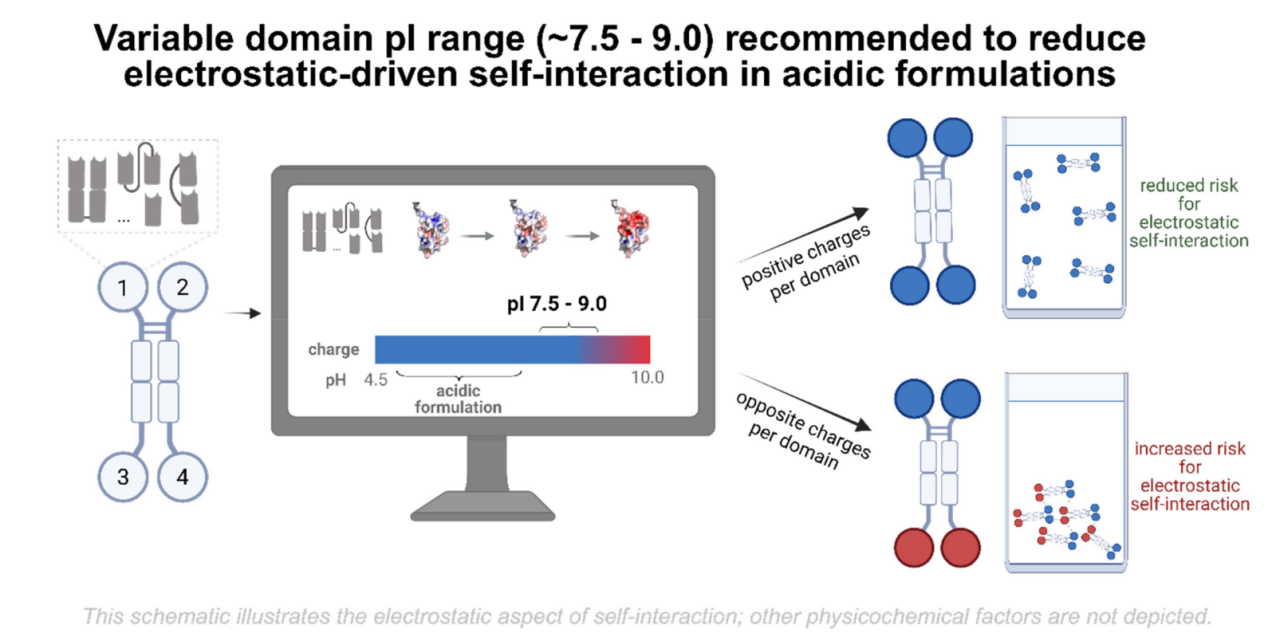
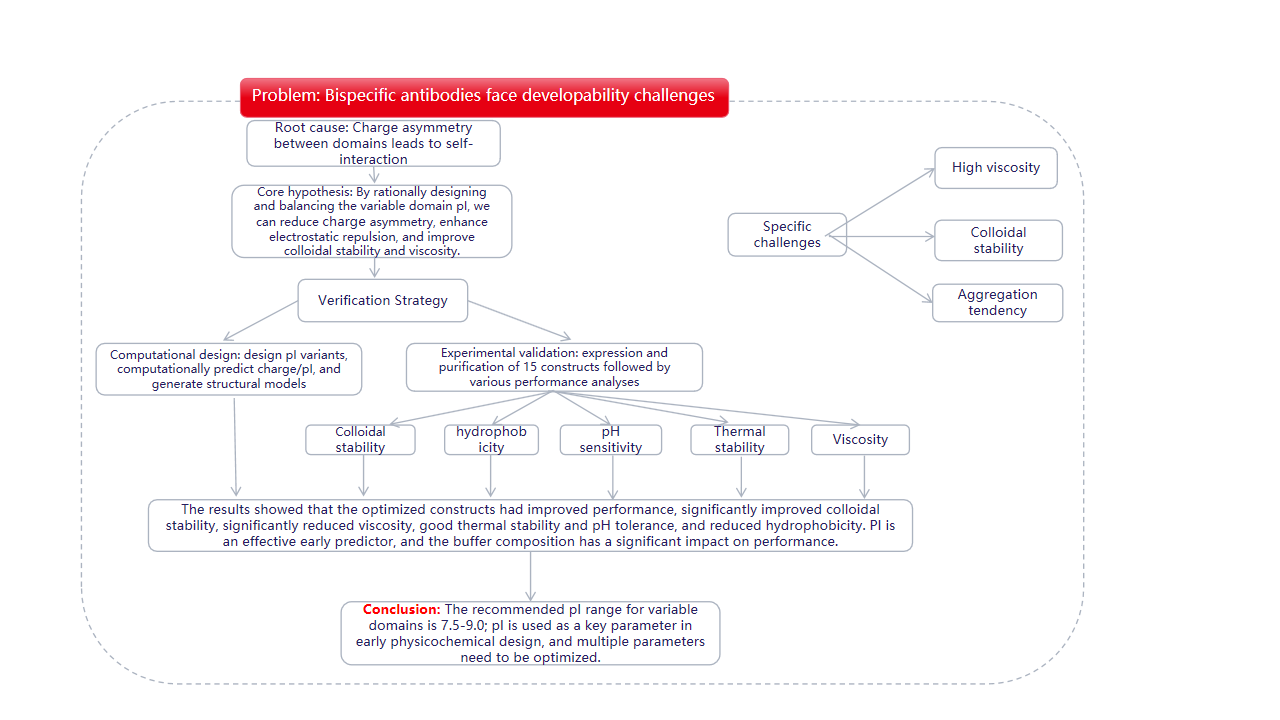
On September 1, 2025, Vanessa Siegmund's team published a study in MAbs titled "Optimizing colloidal stability and viscosity of multispecific antibodies at the drug discovery-development interface: a systematic predictive case study." The study investigated the solution behavior, colloidal stability, hydrophobicity, thermal stability, pH sensitivity, and viscosity of engineered bispecific IgG1-VHH constructs derived from the parental NKp30 × EGFR natural killer cell engager (NKCE) molecule. By combining computer simulation predictions and experimental evaluations, variants with altered antigen-binding fragments (Fab) and variable domains (VHH) were engineered and characterized over a wide formulation-relevant pH range (pH 4.5–8.0). The results showed that making the pI value slightly alkaline (approximately 7.5–9.0) in the variable domain of the bispecific antibody can effectively alleviate the charge asymmetry that may be induced in standard acidic formulations, leading to unfavorable solution behavior. Rational design and early experimental validation yielded optimized variants with significantly improved colloidal stability and viscosity compared to the starting molecule. This systematic study, the first of its kind for a bispecific antibody, highlights the value of integrating domain-level computational simulations. Early assessments in antibody design facilitate efficient optimization of multispecific biotherapeutics for improved solution performance.
Design of Fab and VHH variants and in silico evaluation of domain and complete constructs
Preliminary in silico evaluation of the bispecific NKp30 × EGFR NKCE, Fab M-Fc-VHHL, revealed a calculated pI of 8.4 for the Fab domain and 4.5 for the VHH. This results in opposite charges between these domains in the pH range of 5.5 to 8.0, creating a charge asymmetry that triggers electrostatically induced self-interactions and may lead to aggregation and poor solution behavior.
Design variants covering a wide range of pI and charge profiles. Specific mutations were introduced into the VHH and Fab regions to generate variants with low, intermediate, and high pI values (designated VHHL, FabL, VHHM, FabM, VHHH, and FabH ).
SEC experiments revealed that the Fab H-Fc-VHHH and Fab M-Fc-VHH H constructs had the lowest titers (36.9 and 34.5 mg/L, respectively).


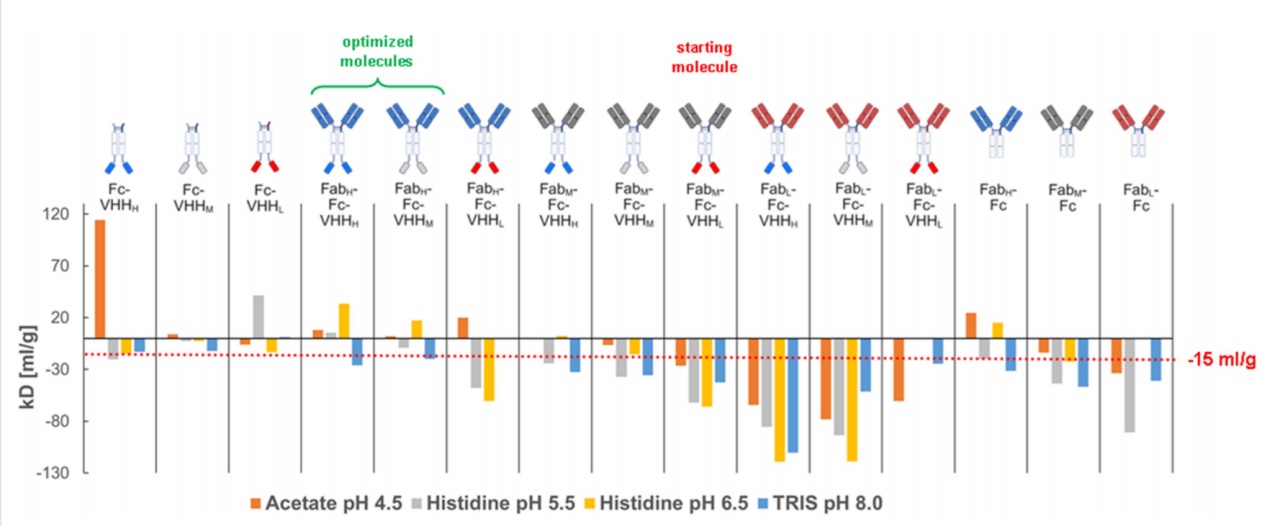
Protein engineering significantly improves colloidal stability in relevant formulation buffers
FabH-Fc-VHHM and FabH-Fc-VHHH, which combine Fab and VHH variants designed for higher pI, exhibited good colloidal stability in formulation buffers at pH 4.5, 5.5, and 6.5, but unfavorable stability only at pH 8.0. These findings are in good agreement with the calculated charge profiles of the FabH, VHHH, VHHM, and Fc domains at these pH values, where all domains exhibited positive charge at pH 6.5 and charge asymmetry only at pH 8.0.
Fab-Fc variants: Colloidal stability improved with increasing Fab domain pI. The high-pI variant, FabH-Fc, performed best across the entire pH range.
Fc-VHH variants: In most cases, a higher VHH pI also resulted in better colloidal stability, but there were exceptions (e.g., Fc-VHHL showed unexpectedly better stability at pH 5.5), suggesting that factors other than net charge (e.g., hinge region flexibility) are at play.
Bispecific constructs: Combining high-pI Fab and VHH domains is an effective strategy for achieving optimal colloidal stability. Results show that electrostatic repulsion is maximized and stability is best when all domains (Fab, Fc, VHH) carry sufficiently high positive charges at the formulation pH.
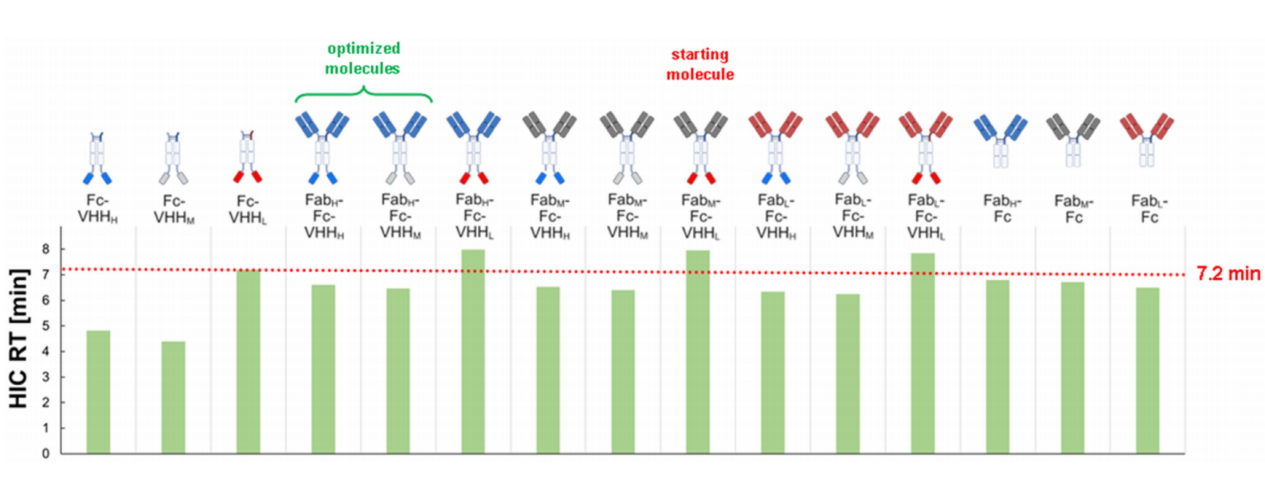
The optimized molecule has lower hydrophobicity
HIC analysis revealed that constructs containing low-pI VHH domains (VHHLs) exhibited longer retention times, suggesting either increased hydrophobicity or less stable conformation. This difference in hydrophobicity was not caused by the introduction of hydrophobic mutations but rather indirectly by altered charge distribution, highlighting the complexity of protein properties. Optimized constructs (using either VHHH or VHHM) exhibited shorter HIC retention times, indicating more favorable hydrophobic properties, which contributes to a reduced propensity to aggregate.
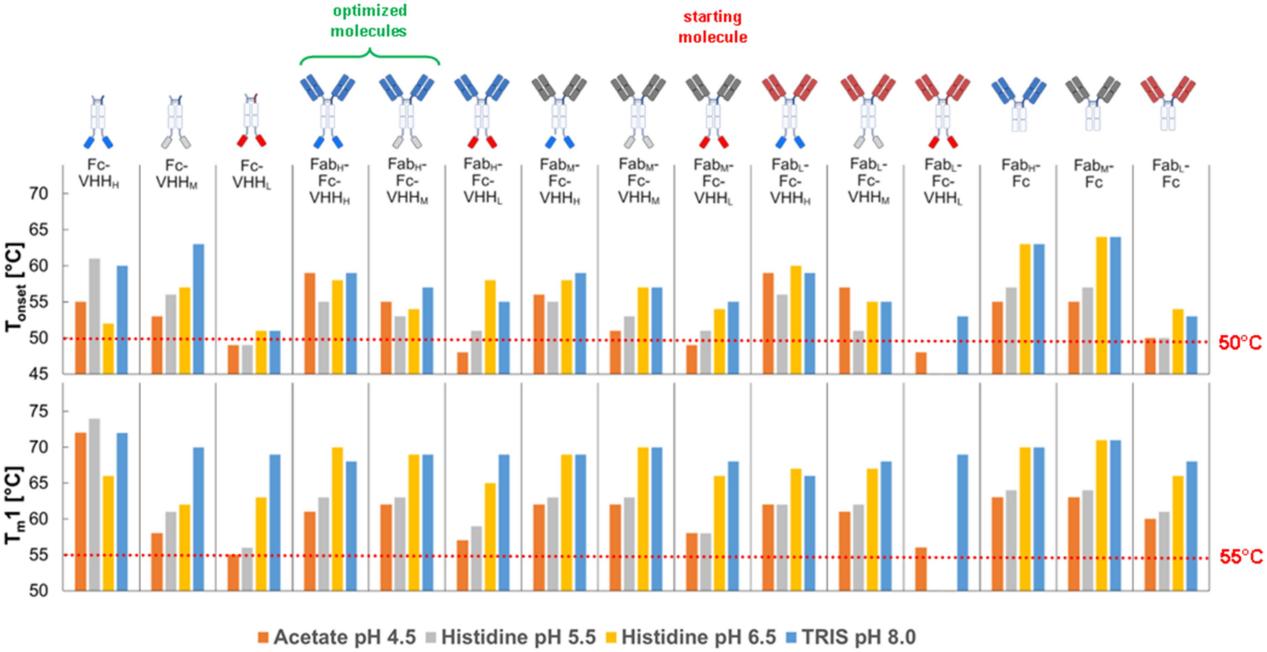
Protein engineering improved the thermal stability of the optimized construct in all formulation buffers
All constructs exhibited acceptable thermal stability (Tm1 > 55°C). Several constructs displayed Tonset values below 50°C at pH 4.5 and 5.5, compared to pH 6.5 and 8.0. The optimized constructs exhibited superior thermal stability compared to the starting molecule across the entire pH range. Thermal stability is primarily determined by the least stable domain. In constructs containing VHHL, the VHHL is the first domain to unfold; in other constructs, the CH2 domain of the Fc domain generally exhibits low conformational stability, especially at acidic pH .
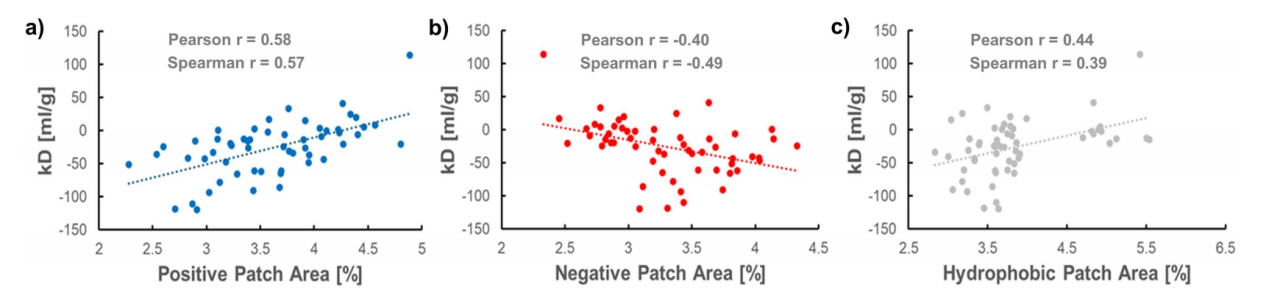
Confirmation of the correlation between computer and experimental data
Correlation analysis revealed a moderate but clear correlation between experimentally measured colloidal stability (kD values) and in silico structural descriptors: correlations were identified with the proportion of positively charged surface patches (Spearman r = 0.57) , negatively charged surface patches, and the proportion of hydrophobic patches . This validates surface charge distribution as a key physical factor driving colloidal stability and supports the use of in silico tools for early predictions.
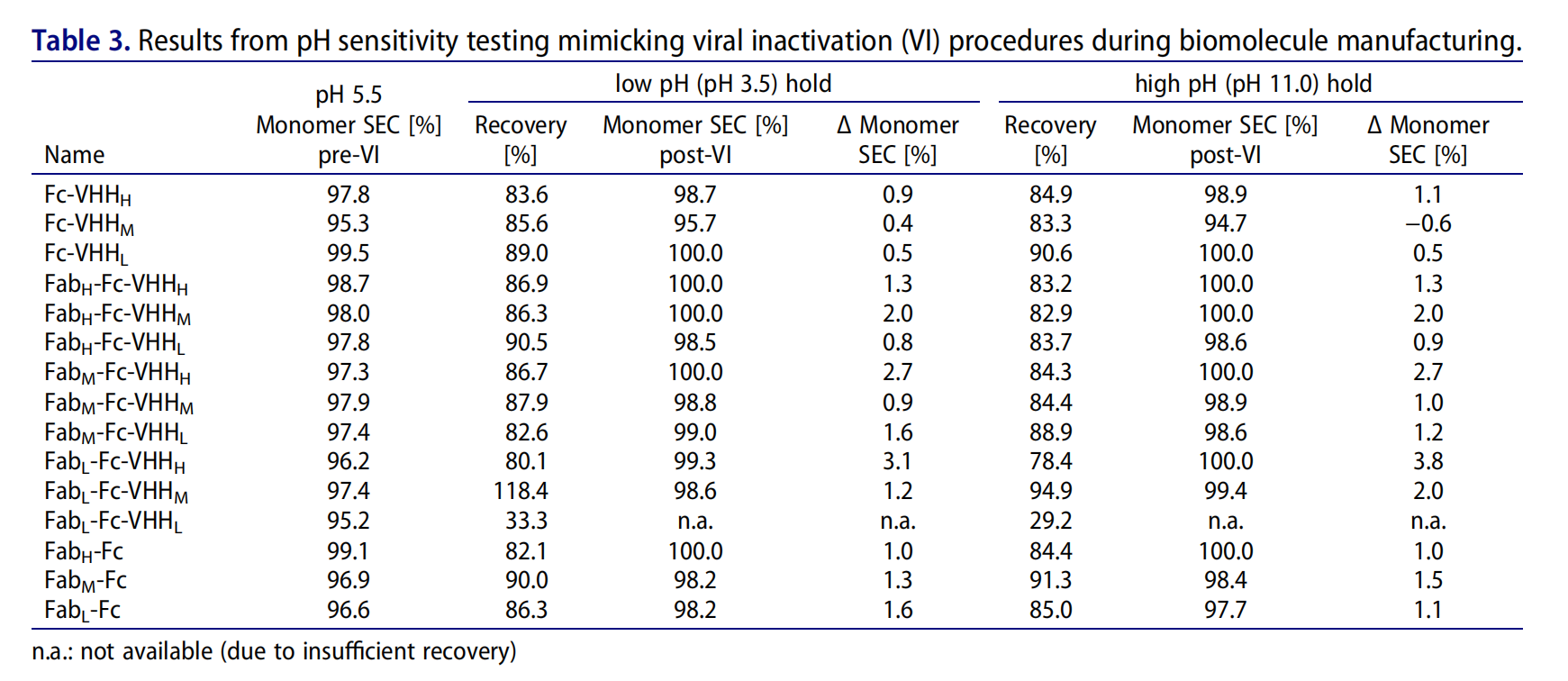
Engineered variants show good pH sensitivity (viral inactivation stability)
pH sensitivity testing is crucial for evaluating the stability and safety of therapeutic antibodies, particularly with regard to their susceptibility to viral inactivation (VI). With the exception of Fab L-Fc-VHHL, all constructs demonstrated robust tolerance to extreme pH conditions and rebuffering at 2 mg/ml . Most engineered variants, including the parental and optimized constructs (Fab H-Fc-VHH H and Fab H-Fc-VHH M), maintained robust stability under extreme pH conditions at the given concentration (2 mg/ml), highlighting their suitability for manufacturing processes involving viral inactivation steps.
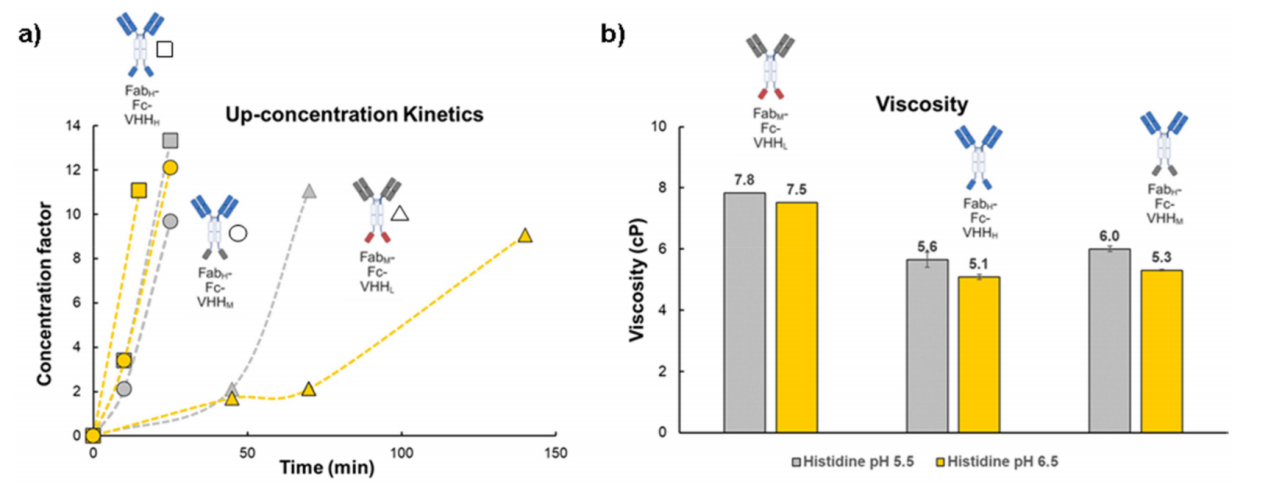
Optimized bispecific constructs display improved viscosity properties
Compared to the starting molecule, the optimized constructs exhibited significantly faster concentration kinetics, highlighting their enhanced suitability for high-concentration formulations. The optimized constructs exhibited significantly lower viscosity than the starting molecule. This viscosity behavior is a result of the strong electrostatic repulsion provided by the highly charged Fab H domain and the absence of charge asymmetry or attractive interactions between the VHH M or Fc domains at this pH. By mitigating charge asymmetry, high viscosity caused by electrostatic attraction can be effectively reduced, which is crucial for developing high-concentration subcutaneous formulations.
In conclusion
Our results demonstrate that electrostatically driven self-interactions can be effectively reduced through rational charge and pI engineering, thereby improving the colloidal stability and viscosity of multispecific antibodies in acidic formulation buffers. The variable region pI range was optimized to approximately 7.5–9.0 to specifically reduce electrostatic self-interactions in acidic formulations. While pI and charge descriptors provide a practical early-stage strategy to reduce the risks associated with electrostatic self-association, antibody solution behavior is inherently multifactorial. Other physicochemical properties, such as conformational stability, hydrophobicity, linker design, and formulation architecture, are governed by other, partially independent determinants and may require case-by-case screening and optimization. Future studies will extend these insights to different multispecific antibody formats and systematically explore other molecular factors, including linker design, as well as formulation parameters, which will further refine predictive models and support the rational design of next-generation antibody therapeutics.