β-nerve growth factor (NGF), also known as NGFB, is an extracellular ligand for NTRK1 and NGFR receptors. During growth and development, NGF promotes neuronal survival, proliferation, and differentiation, and repairs and regenerates damaged nerve cells. In adulthood, NGF's main function is to regulate the activity of nociceptive neurons and pain responses. NGF levels are elevated in trauma, inflammation, and chronic pain, and it acts on TrkA receptors on the surface of nociceptors, activating intracellular signaling and triggering pain mechanisms. Therefore, selectively inhibiting NGF can help prevent or alleviate pain signals from tissues or organs from being transmitted to the brain.
NGF expression distribution
NGF is mainly expressed in female tissues, muscle tissues, connective and soft tissues, and is expressed in supporting cells, interstitial cells, smooth muscle cells, inhibitory neurons, ovarian stromal cells, and oligodendrocyte precursor cells.

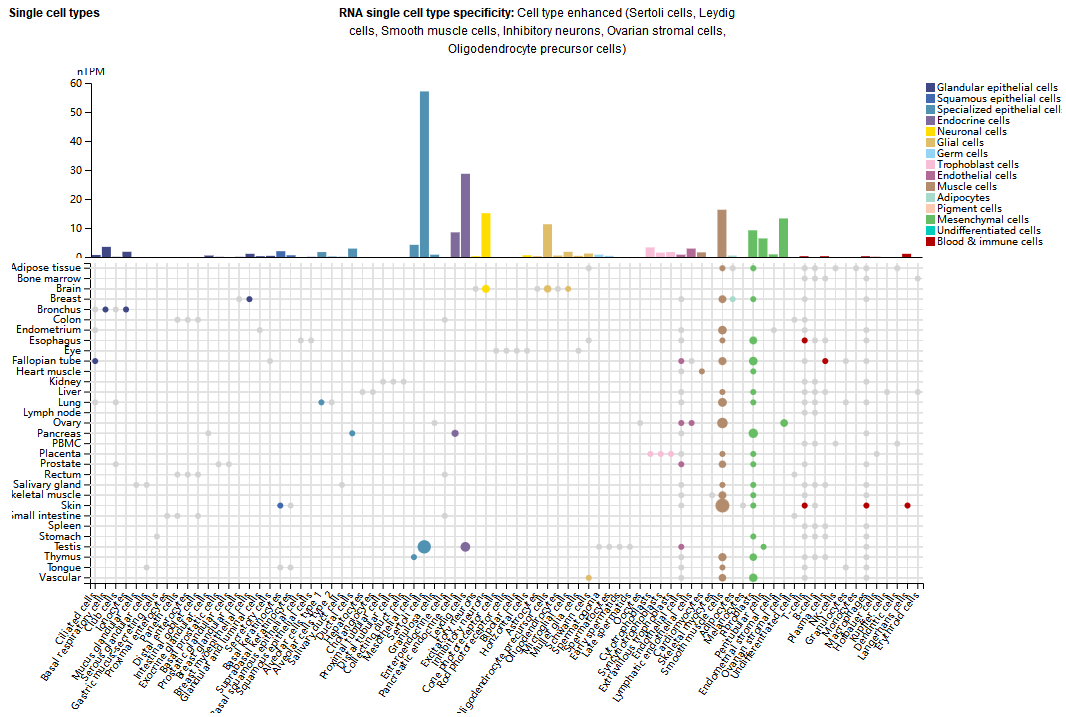
(Data source: uniprot)
Structure, signaling pathways, and regulation of NGF
NGF is a secretory protein composed of 241 amino acids. The precursor of NGF (proNGF, a 32 kDa molecule) is synthesized in the form of a homodimer, which is then cleaved by extracellular proteases (such as plasmin and neuroserine protease inhibitors) or proteases in the Golgi apparatus and secretory vesicles (such as furin and proconvertase) to eventually form mature NGF.
Immature NGF precursor (proNGF) acts as a ligand for the heterodimer receptor formed by SORCS2 and NGFR, activating the cell signaling cascade, leading to RAC1 and/or RAC2 inactivation, actin cytoskeleton remodeling, and collapse of the neuronal growth cone.
ProNGF and NGF bind to receptor TrkA (a receptor with tyrosine kinase activity) and p75 neurotrophic factor receptor (p75NTR, a member of the tumor necrosis factor receptor (TNFR) superfamily), respectively.
ProNGF binds to the p75NTR receptor with high affinity, while NGF preferentially binds to the TrkA receptor and has a lower affinity for p75NTR.
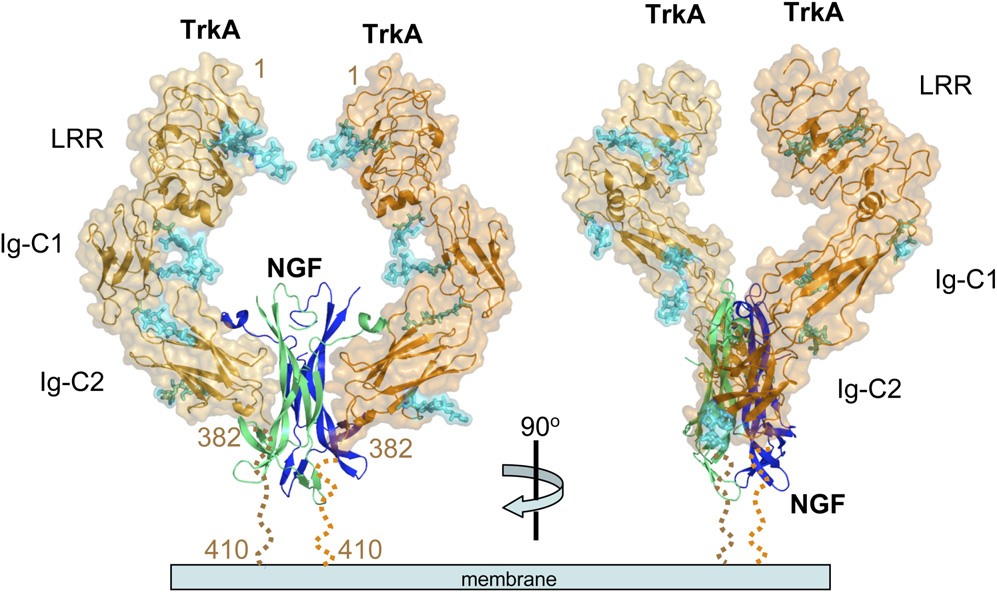

(Data source: Wehrman T, et al. Neuron. 2007)
The binding of NGF to TrkA induces dimerization and transphosphorylation of tyrosine residues located in the cytoplasmic domain of TrkA. For example, phosphorylation at Y785 induces the activity of its effector protein phospholipase c-γ (PLC-γ), promoting cell differentiation and survival. On the other hand, phosphorylation at Y490 activates the MAPK and phosphatidylinositol 3-kinase ( PI3K ) /AKT pathways, inducing cell proliferation, survival, and growth.
The binding of NGF to p75NTR induces conformational changes in the receptor's intracellular domain, thereby recruiting effector proteins, including TNF-receptor-associated factor 6 (TRAF6) and receptor-interacting serine/threonine protein kinase 2 ( RIPK2 or RIP2 ) . Similar to TrkA activity, TRAF6 and RIPK2 promote cell survival by activating the NF-κB and PI3K/Akt pathways. p75NTR may promote TrkA activity because the binding of these two receptors allows NGF to form a high-affinity binding site.
p75NTR forms a heterodimer with sortilin, inducing a pro-apoptotic pathway via c-Jun N-terminal kinase (JNK) and activated caspases 3, 6, and 9. Furthermore, the interaction between p75NTR and sortilin increases the affinity of p75NTR for proNGF, suggesting that exposure to high levels of proNGF may lead to a pro-apoptotic pathway. In addition to these classical pathways associated with proliferation, survival, and apoptosis, NGF receptors are also involved in metabolic homeostasis. Therefore, alterations in the proNGF/NGF ratio and the abundance of TrkA and p75NTR in peripheral tissues may lead to metabolic abnormalities.
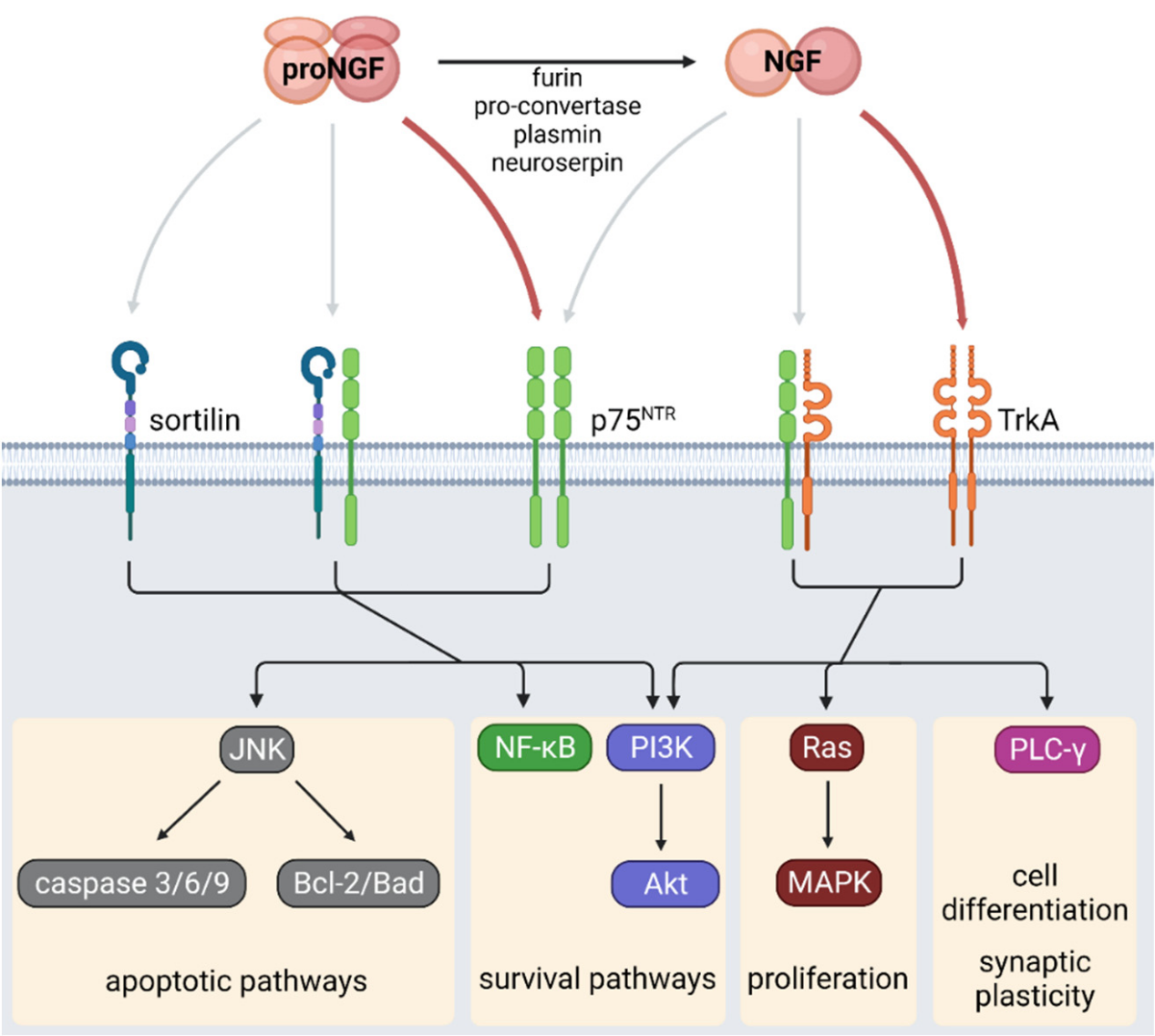
(Data source: Samario-Román J, et al.) Int J Mol Sci . 2023)
NGF -targeted therapy
Fasinumab is a monoclonal antibody targeting NGF, developed by Regeneron Pharmaceutica. Its highest development stage was Phase 3 clinical trials. Currently, research on this antibody has been terminated.
Tanezumab, developed by Pfizer, is a monoclonal antibody targeting NGF. It was later developed in collaboration with Eli Lilly for the treatment of neuropathic pain and schwannoma. It was the first NGF inhibitor to receive FDA Fast Track designation, possessing the ability to selectively bind to and inhibit NGF. NGF levels rise in the body when experiencing pain caused by injury, inflammation, or chronic pain. Therefore, by selectively inhibiting NGF, tanezumab may help block pain signals generated in muscles, skin, and organs from reaching the spinal cord and brain.
AK115 is a monoclonal antibody targeting NGF developed by Akeso, Inc for the treatment of pain, and is currently in Phase 1b/II clinical trials.

(Data source: Akeso Biopharma official website)
DS-002, developed by Dartsbio, is currently the only known monoclonal antibody drug in China targeting NGF, and ranks third globally in terms of development speed among drugs under development. Its clinical trial application was accepted by the NMPA in October 2020, and clinical trials will commence. The overall market potential for this drug is estimated at 30 billion RMB. It can bind to NGF and block its binding to TrkA on the surface of nociceptors, ultimately achieving a non-addictive and potent analgesic effect. It has the potential to replace opioids and NSAIDs in indications such as chronic pain and chemotherapy-induced pain. Currently, the drug is in Phase II clinical trials.
SSS40 is a monoclonal antibody targeting NGF developed by 3SBIO Inc. for the treatment of cancer pain, and is currently in Phase 1/2 clinical trials.
TNM009 is a recombinant, fully human anti-nerve growth factor (NGF) monoclonal antibody developed by Trinomab for analgesia. In March 2023, the CDE approved the Investigational New Drug (IND) application for TNM009. NGF antibodies inhibit the NGF-TrkA signaling axis only in the peripheral nervous system, without interfering with the release of neurotransmitters in the central nervous system; therefore, TNM009 theoretically has no significant risk of addiction. Furthermore, thanks to its large molecular weight monoclonal antibody characteristics, NGF antibodies require less frequent administration, a feature particularly suitable for patients with chronic pain requiring long-term management, such as bone metastasis cancer pain and chemotherapy-induced neuropathic pain. NGF antibodies hold promise for combination therapy with low-dose opioids, enhancing analgesic effects through multi-pathway synergy; this strategy can reduce the cumulative dosage of opioids, decrease addiction and dependence, and delay the pain sensitization process through complementary mechanisms. In addition, due to the low immunogenicity of fully human monoclonal antibodies, TNM009 is theoretically less likely to experience reduced efficacy due to immunogenicity.

(Data source: Trinomab website)
