Vascular endothelial growth factor receptor 1 (VEGFR1), also known as FLT1 or FRT, is a tyrosine protein kinase. As a cell surface receptor for VEGFA, VEGFB, and PGF, it plays a crucial role in embryonic angiogenesis, cell survival, cell migration, macrophage function, chemotaxis, and cancer cell invasion. It is not only a vital regulator of vascular system development and function maintenance but also a key therapeutic target for various cancers, ophthalmic diseases, and inflammatory diseases.
VEGFR1 expression
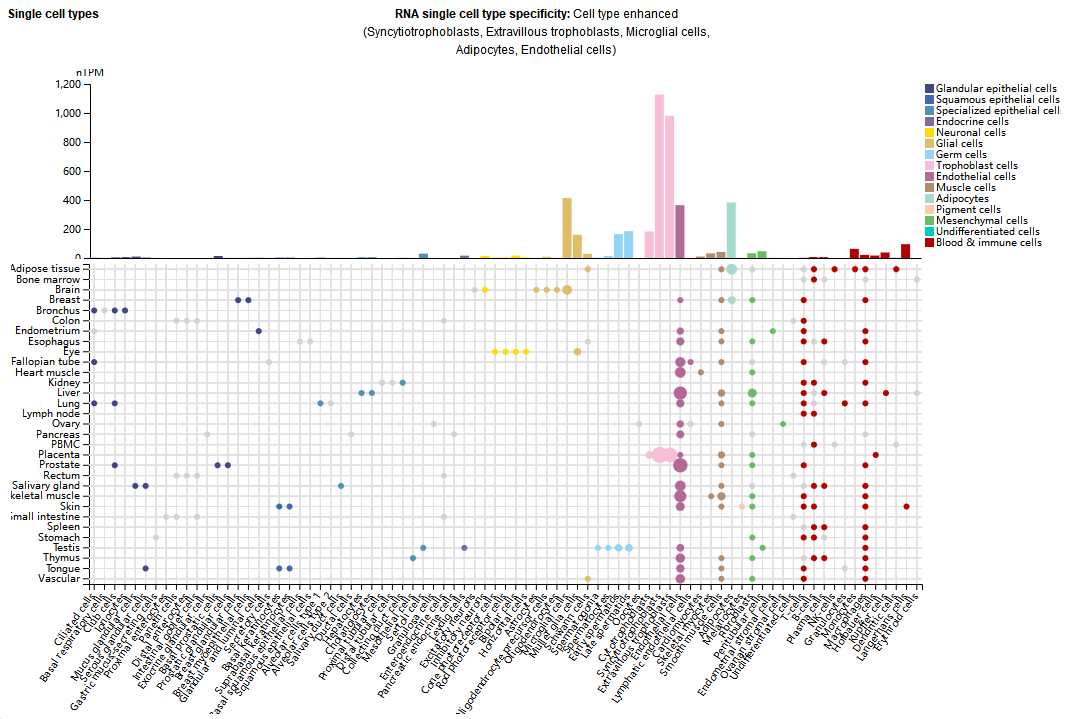
VEGFR1 is expressed in various cell types, primarily in syncytiotrophoblast cells, extravillous trophoblast cells, microglia, adipocytes, endothelial cells, photoreceptor cells, and retinal pigment epithelial cells.
(Data source: uniprot)
Structure of VEGFR1 and its ligands
VEGFR1 is a transmembrane protein composed of 1338 amino acids with a molecular weight of approximately 149 kDa. It consists of an extracellular region, a transmembrane region, and an intracellular region.
Extracellular region: Contains 7 immunoglobulin-like domains, which are responsible for binding to ligands with high affinity.
Transmembrane region: A hydrophobic α-helix that anchors the receptor to the cell membrane.
Intracellular region: Contains a splitting tyrosine kinase domain and a C-terminal tail. The tail contains multiple phosphorylated tyrosine residues and is key to signal transduction.

(Data source: Markovic-Mueller S, et al. Structure. 2017)
There are five VEGFR ligands, of which VEGF-A can bind to both VEGFR1 and VEGFR2, while PlGF binds only to VEGFR1. Splicing produces isoforms of VEGF-A and PlGF. Furthermore, soluble/secretory VEGFR1 and VEGFR2 can be produced through alternative splicing or proteolytic cleavage, retaining their extracellular ligand-binding domains. VEGF-A and PlGF ligands, as well as VEGFR1 and VEGFR2 receptors, can form both homodimers and heterodimers. The synergistic effect of PlGF and VEGF-A is due to their shared receptor VEGFR1 and their ability to heterodimerize.
VEGFR1 has an extremely high affinity for its major ligands VEGF-A and PIGF (placental growth factor) (approximately 10 times higher than VEGFR2's affinity for VEGF-A), but its tyrosine kinase activity is relatively weak. This characteristic determines its unique behavior in signal transduction.

(Data source: Uemura A, et al. Prog Retin Eye Res. 2021)
VEGFR1 signaling pathway and regulation
Specific ligands can bind to and activate VEGFR1, transmitting signals through its own kinase domain. However, VEGFR1 can also act as a decoy receptor for VEGFR2 ligands, thereby indirectly affecting VEGFR2 activity.
VEGFR1 binds to VEGF-A or PIGF, activating various downstream signal transduction pathways. PKC/MEK/MAPK leads to endothelial cell proliferation, the PKB pathway promotes endothelial cell survival, NF-κB promotes macrophage migration, and the eNOS pathway regulates vascular permeability. These pathways promote many pathological processes in endothelial cells and pericytes in the choroid and retina.
In retinal pigment cells: angiogenesis occurs through the Bruch membrane into the retinal pigment epithelium, resulting in the loss of retinal pigment cells; in photoreceptor cells: photoreceptor integrity is lost, rod cells die, and cone cell fragments are lost; in Müller cells: Müller cells are activated; in microglia: recruitment, accumulation, and activation of microglia and other retinal macrophages lead to the release of pro-inflammatory cytokines such as platelet-derived growth factor-A, soluble intracellular adhesion molecule-1, CC chemokine ligand 2, and interleukin-8, thereby resulting in the formation of hyperreflective foci.

(Data source: Uemura A, et al. Prog Retin Eye Res. 2021)
VEGFR1 targeted therapy
Icrucumab is a monoclonal antibody that targets VEGFR1. It inhibits tumor survival-related cellular activities by blocking the VEGF/VEGFR-1 interaction. The most recent stage of research is Phase 2 clinical trials, and there are currently no new research updates.
AL201-AB is a bispecific antibody developed by AngioLab that targets VEGFR1 and VEGFR2 for the treatment of eye diseases and tumors, and is currently in the preclinical research stage.


(Data source: AngioLab official website)
