Alpha-synuclein (SNCA) is a neuronal protein that plays multiple roles in synaptic activity, including regulating synaptic vesicle trafficking and subsequent neurotransmitter release. It is also known as the non-A4 component of the amyloid precursor (NACP) and PARK1. Aggregated α-synuclein, through intercellular transmission and the spread of neuroinflammation, has emerged as a key mechanism in Parkinson's disease neuropathology.

(Data source: Stocchi F, et al. Nat Rev Neurol. 2024)
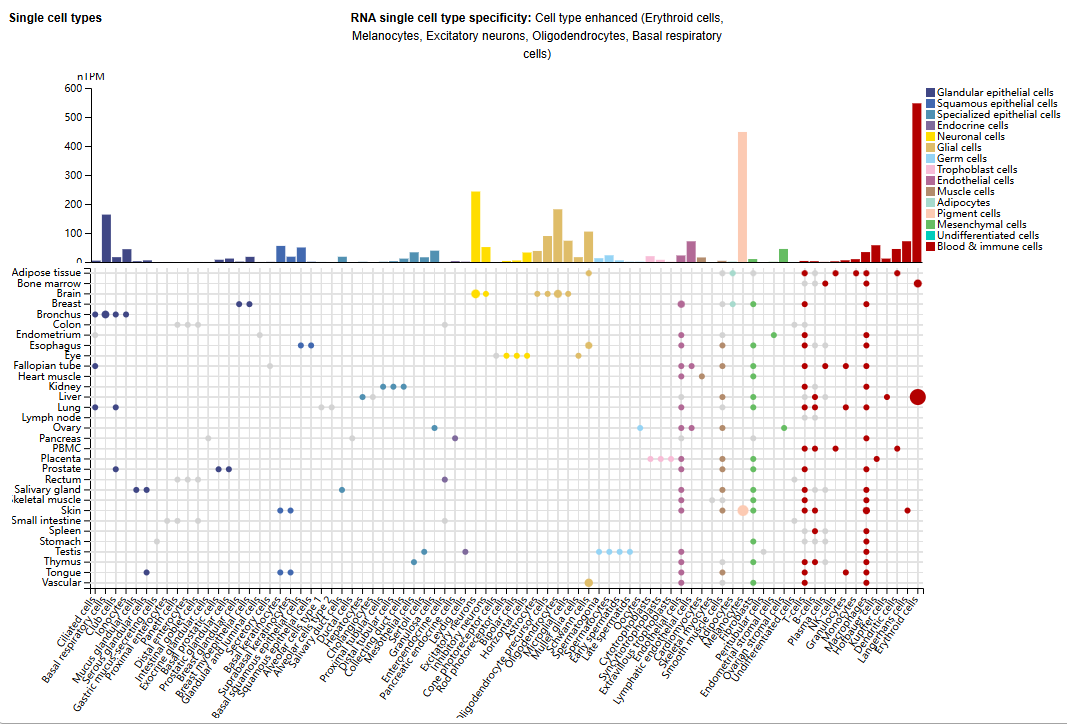
SNCA expression distribution
SNCA is mainly expressed in the brain, bone marrow and lymphoid tissues, and is expressed in erythroid cells, melanocytes, excitatory neurons, oligodendrocytes and basal respiratory cells.

(Data source: Uniprot)
Structure and function of SNCA
SNCA is an intracellular protein with a molecular weight of 14 kDa and composed of 140 amino acids. The primary αSyn amino acid sequence consists of three regions: the N-terminus, the NAC (non-amyloid β component), and the C-terminus, each of which exhibits distinct molecular and biological properties. In healthy neurons and other cell types, αSyn primarily exists as an unfolded monomer. Due to its lack of a native three-dimensional structure, monomeric αSyn is considered an intrinsically disordered protein. Consequently, it possesses structural flexibility and sensitivity to modification, making it prone to misfolding and aggregation.
The N-terminus is an alpha-helical structure that serves as an anchor for lipid membrane binding. The basic structural elements of these interactions include seven incomplete 11-residue repeats that form the core consensus motif "KTKEGV." The N-terminus contains four 11-residue repeats, with the remaining three located in the NAC region.

(Data source: Alphafold)
Native unfolded αSyn monomers may exist as α-helices, tetramers, or membrane-bound multimers. Numerous factors, including intrinsic protein properties, biological and chemical stimuli, the αSyn gene (SNCA), and post-translational modifications (PTMs), can trigger αSyn misfolding and aggregation. Aggregated, toxic αSyn significantly impairs synaptic function by interfering with dopamine synthesis, reuptake, and release; affecting synaptic vesicle cycling; generating reactive oxygen species; reducing axonal transport; and ultimately leading to neuronal apoptosis.

(Data source: Saramowicz K, et al. Int J Mol Sci. 2023)
SNCA in Parkinson's disease (PD)
Parkinson's disease (PD) is a common, age-related neurodegenerative disorder characterized neuropathologically by the presence of intracellular inclusions known as Lewy bodies and Lewy neurites. These protein aggregates, composed in part of misfolded α-synuclein, are characteristic of PD neuropathology. Increased SNCA expression is associated with the pathogenesis of PD. SNCA mutations, including A53T, A30P, and E64K, have been shown to promote α-synuclein misfolding and aggregation.
Autophagy in microglia plays a crucial neuroprotective role by regulating α-synuclein homeostasis and inflammasome activation. α-Synuclein activates the NF-κB pathway through TLR2 and TLR4, upregulating p62 mRNA levels. As a selective autophagy receptor, p62 binds to internalized α-synuclein and recruits the autophagic machinery. The p62-α-synuclein complex is sequestered by autophagosomes and degraded by lysosomes. Activation of the NF-κB pathway by TLR-α-synuclein interactions increases NLRP3 mRNA levels, initiating inflammasome activation. Components of the inflammasome complex are recognized by the autophagic machinery and degraded by autophagosomes and lysosomes, thereby preventing excessive inflammasome formation in the brain.

(Data source: Choi I, et al. Sci Adv. 2022)
Targeted therapy for SNCA
α-Syn aggregation itself is a therapeutic target. From the moment the α-syn gene is encoded in DNA to the moment α-syn aggregates into fibrils, interventions can be strategically addressed and designed at every stage. Potential disease-modifying therapies include aggregation inhibitors that block α-synuclein misfolding and aggregation, as well as monoclonal antibodies that prevent protein accumulation and enhance protein clearance.

(Data source: Kuo G, et al. Mol Neurodegener. 2025)
Amlenetug is a monoclonal antibody targeting SNCA for the treatment of multiple system atrophy (MSA). It recognizes and binds to all major forms of extracellular α-synuclein, thereby preventing its uptake and inhibiting its aggregation. Amlenetug has an active Fc region, potentially enhancing immune-mediated clearance of the α-syn/mAb complex via microglia-mediated uptake. Amlenetug was developed by Lundbeck under a joint research and licensing agreement between Lundbeck and Genmab a/S.
On November 27, 2024, Lundbeck announced the clinical development of amlenetug (Lu AF82422) for the treatment of MSA with the initiation of MASCOT, a randomized, double-blind, Phase III trial. The trial aims to evaluate the efficacy, safety, and tolerability of amlenetug in patients with multiple system atrophy (MSA). Amlenetug will be administered by intravenous infusion every four weeks.
Cinpanemab (BIIB054), developed by Biogen , targets the N- terminus of α-syn and has been shown in preclinical studies to inhibit the spread and aggregation of α-syn. However, despite an acceptable safety profile in a Phase 1 trial, a Phase 2 study showed no difference between the treatment and placebo groups in motor function, non-motor function, activities of daily living, quality of life, and progression of imaging biomarkers at 52 weeks, leading to the discontinuation of development.
Prasinezumab (PRX002/RG7935) is a monoclonal antibody targeting α-syn C for the treatment of Parkinson's disease (PD). In a Phase 1 trial, it demonstrated safety and tolerability, binding to peripheral α-syn, and successfully achieving a dose-dependent increase in prasinezumab levels in the cerebrospinal fluid . Prasinezumab is currently in a Phase 2b clinical trial to further evaluate its efficacy in patients with more advanced PD.

(Data source: Kuo G, et al. Mol Neurodegener. 2025)
