Galectin-3 (LGALS3), also known as MAC2, Gal-3, and IgE-binding protein, participates in acute inflammatory responses, including neutrophil activation and adhesion, monocyte-macrophage chemotaxis, opsonization of apoptotic neutrophils, and mast cell activation. Together with TRIM16, it coordinates membrane damage recognition and the mobilization of core autophagy regulators ATG16L1 and BECN1 in response to damaged endothelial cells. It plays a crucial role in fibrosis and inflammation in the heart, liver, kidney, and lungs, respiratory infections caused by COVID-19, and neuroinflammatory diseases.
(Data source: Bouffette S, et al. Trends Pharmacol Sci. 2023)
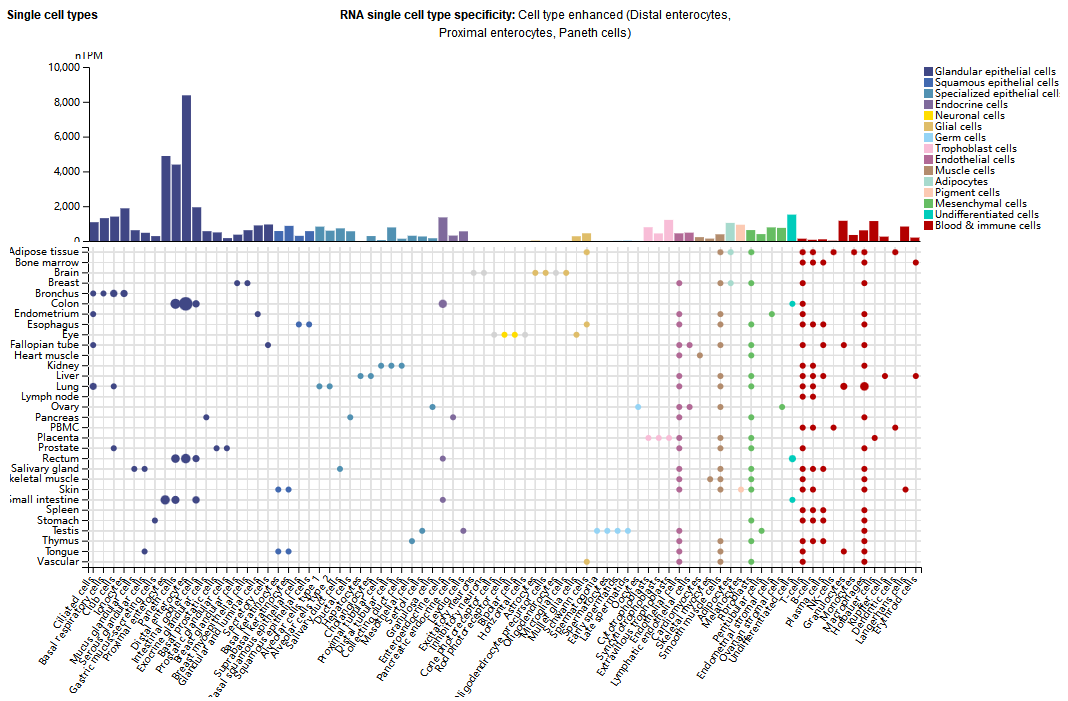
Expression distribution of LGALS3
LGALS3 is widely expressed in human tissue cells, including immune cells (macrophages, monocytes, dendritic cells, eosinophils, mast cells, natural killer cells, and activated T and B cells), epithelial cells (distal enterocytes, proximal enterocytes), endothelial cells, and sensory neurons.
(Data source: uniprot)
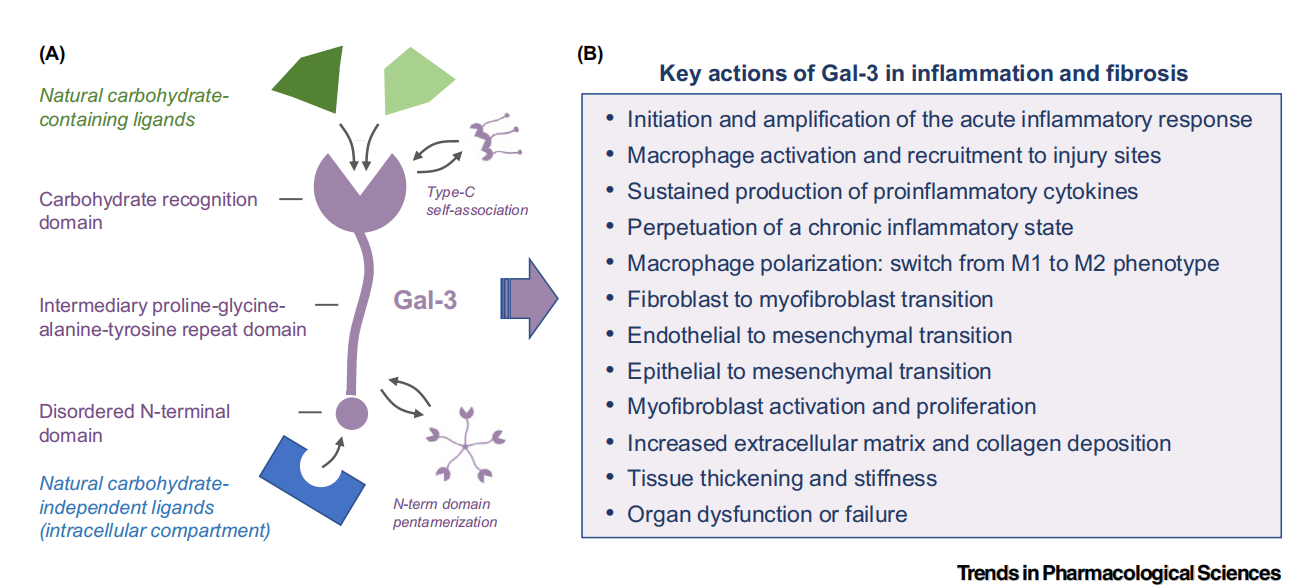
Structure of LGALS3 and its receptor
LGALS3 is a secreted protein that can also be localized in the cytoplasm and nucleus. It consists of 250 amino acids and has a molecular weight of approximately 35 kDa. Gal-3 is the only galectin that can form a pentamer.
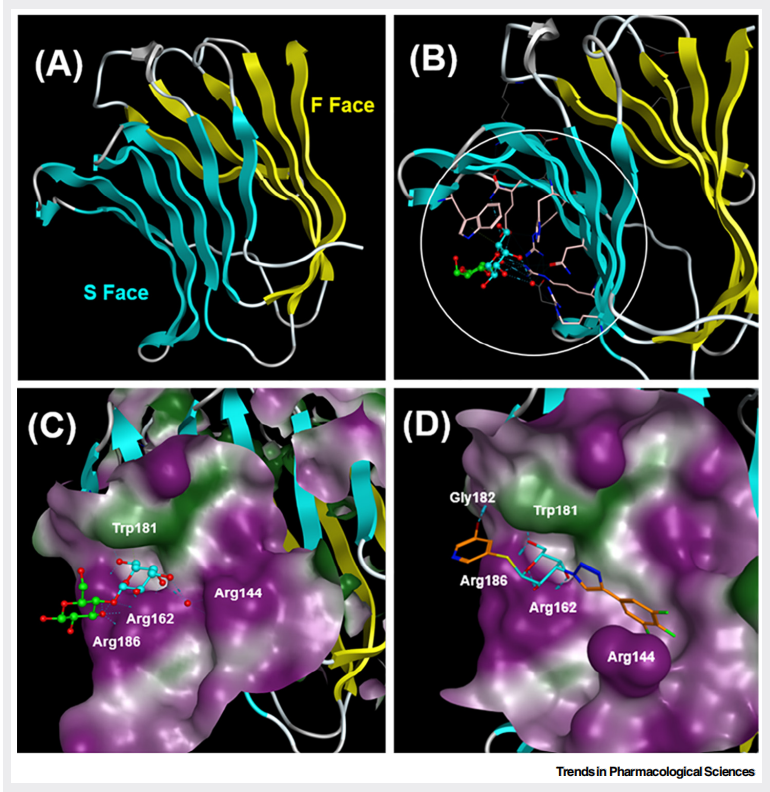
The C-terminal domain is a highly conserved carbohydrate recognition domain. This is the core site for binding to β-galactoside-containing carbohydrate ligands. The carbohydrate recognition domain (CRD) is a β-sandwich fold consisting of approximately 130 amino acids, containing a shallow, solvent-exposed groove for carbohydrate binding. The S-face, consisting of a six-stranded β-sheet, is the primary site of carbohydrate binding. The F-face, consisting of a five-stranded β-sheet, is the site of action for newly discovered allosteric inhibitors (such as PTX-013).
The middle domain is a repeating sequence rich in proline, glycine, alanine, and tyrosine. This region is similar to collagen and plays an important role in protein stability and functional regulation.
The N-terminal domain is a short disordered domain that is the most unique part of LGALS3 and mediates its multimerization and sugar-independent interactions with other proteins.
It can also form oligomers through its N-terminal domain via a process similar to liquid-liquid phase separation or through self-association through its C-terminal domain, a process known as C-type self-association.
(Data source: Bouffette S, et al. Trends Pharmacol Sci. 2023)
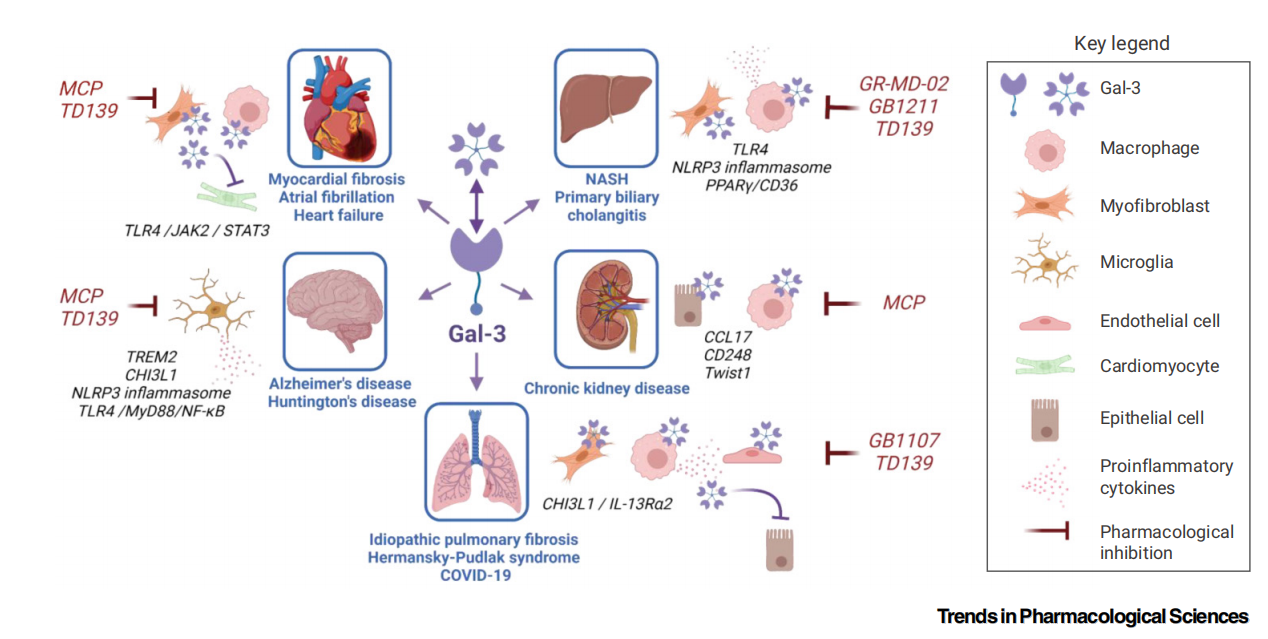
The role of LGALS3 in disease
Role in inflammation: Activates inflammatory signaling pathways (such as MyD88/NF-κB) through Gal-3-TLR4 interaction. Interacts with the NLRP3 inflammasome axis to promote the maturation and secretion of IL-1β/IL-18. Forms "galectin factors," which form heterodimers with chemokines (such as CXCL12) and cytokines (such as IFN-γ), regulating immune cell migration and function.
Role in fibrosis: LGALS3 is a core molecule that drives fibrosis by activating macrophages, amplifying chronic inflammatory signals, and directing the inflammatory response toward uncontrolled tissue repair. It directly promotes the transformation of fibroblasts into myofibroblasts and drives mesenchymal transition of epithelial and endothelial cells, leading to an increase in cells that produce excessive collagen, thereby exacerbating tissue sclerosis and loss of organ function. LGALS3 has become a key fibrosis biomarker and therapeutic target. Inhibiting its function has been shown to effectively alleviate the pathological process in models of fibrosis in various organs, including the heart, liver, lungs, and kidneys.
(Data source: Bouffette S, et al. Trends Pharmacol Sci. 2023)
Gal-3 is a core molecule driving neuroinflammation: In neurodegenerative diseases, Gal-3 is expressed and released in large quantities by activated microglia, amplifying the inflammatory response and exacerbating neuronal damage by interacting with key pattern recognition receptors (such as TLR4 and TREM2).
Plays a harmful role in Alzheimer's disease: In AD, Gal-3 is a hallmark protein of Aβ plaque-associated microglia. As a ligand of TREM2, it deleteriously regulates inflammatory responses and promotes pathological progression. Gene knockout or inhibition of Gal-3 can significantly alleviate pathological and cognitive deficits in AD model mice.

(Data source: García-Revilla J, et al . Cell Death Dis. 2022)
LGALS3-targeted therapy
TB-006 is a humanized monoclonal antibody targeting Galectin-3 developed by TrueBinding. It may be a revolutionary drug for the treatment of Alzheimer's disease and is currently in Phase 2 clinical trials.

In addition to amyloid plaques, another key protein, lectin-3 (Gal3), has also been implicated in the pathogenesis of Alzheimer's disease (AD). Abnormally elevated levels of Gal3 are found in the brains of AD patients, where it binds to amyloid-β (Aβ) and promotes its aggregation. Acting as a molecular scaffold, Gal3 facilitates the self-assembly of Aβ into toxic oligomers and plaques. These Aβ deposits on the surface of neurons, disrupting synaptic signaling and contributing to cognitive decline in AD. TB006 selectively binds to Gal3, inhibiting its adhesion function, thereby preventing Aβ aggregation and promoting the dissolution of existing plaques. This mechanism restores synaptic integrity, potentially alleviating neurotoxicity and improving cognitive function in Alzheimer's patients.

TB001 is another monoclonal antibody targeting Gal3 that is currently in preclinical research. In two transgenic (APPSwe, 5xFAD) and Abeta injection-induced Alzheimer's disease mouse models, after only two weeks of treatment with mTB001 (an alternative to TB006), total Abeta plaques and other neurodegenerative biomarkers were significantly reduced. Treatment with TB001 significantly improved spatial memory cognitive function that depends on the hippocampus.
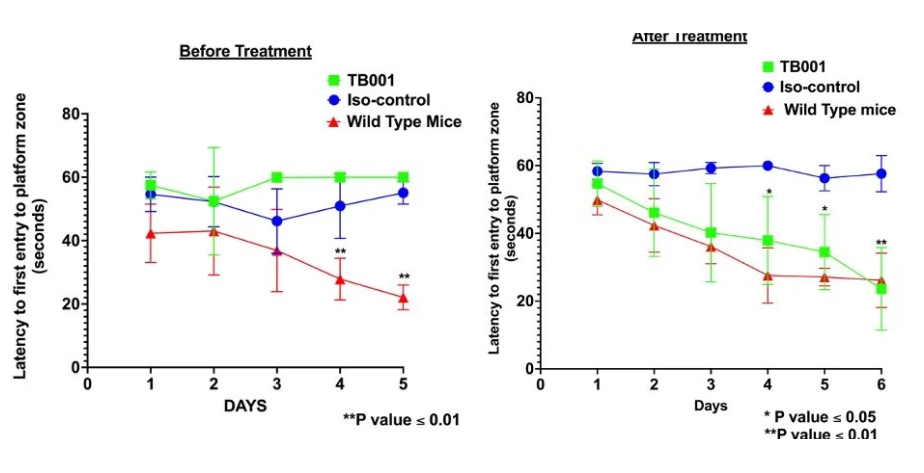
(Data source: TrueBinding official website)
