The tumor antigen 5T4/WAIF1 (Wnt activation inhibitory factor 1; also known as trophoblast glycoprotein TPBG) is a cell surface protein targeted in multiple cancer immunotherapy clinical trials. TPBG may act as an inhibitor of Wnt/β-catenin signaling by indirectly interacting with LRP6 and preventing Wnt3a-dependent LRP6 internalization.
Expression distribution of TPBG
TPBG is mainly expressed in basal respiratory cells, extracellular trophoblasts, and mesothelial cells. TPBG is widely expressed in placental trophoblast cells during embryonic development and is involved in embryo implantation and placenta formation.

TPBG is highly expressed in a variety of solid tumors (with a positive rate as high as 80%), and its expression level is directly correlated with tumor aggressiveness and poor prognosis. These include non-small cell lung cancer, renal cancer, pancreatic cancer, prostate cancer, breast cancer, colorectal cancer, gastric cancer, ovarian cancer, and cervical cancer. The limited expression of 5T4 in normal tissues makes it an attractive target for cancer immunotherapy.
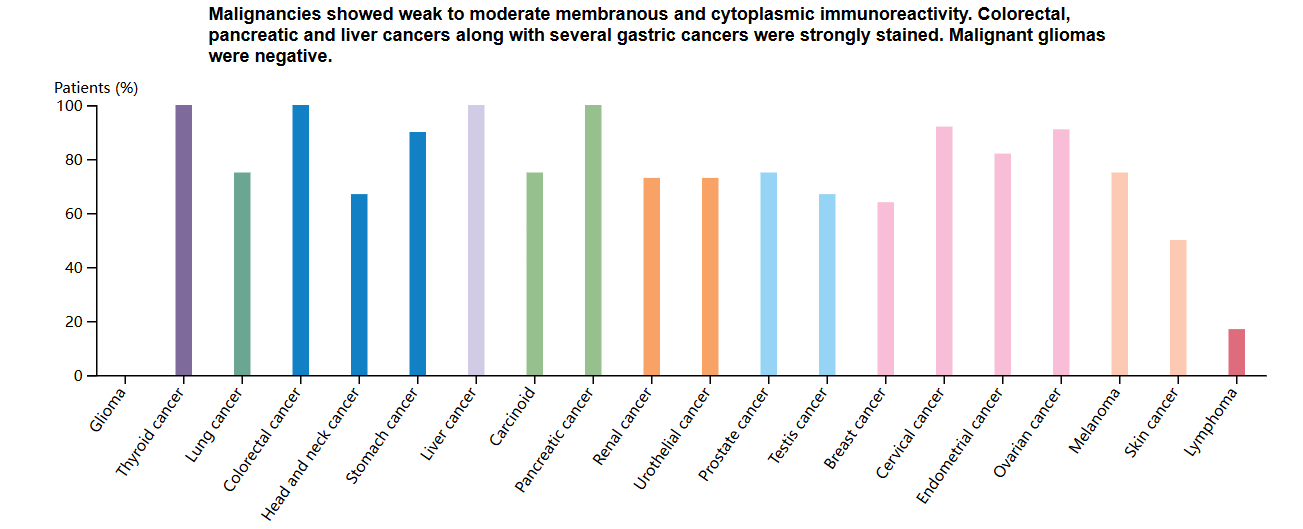
(Data source: Uniprot)
The structure of TPBG and its receptor
TPBG is a type I transmembrane protein composed of 420 amino acids, including an N-terminal extracellular region, a transmembrane region, and a C-terminal intracellular region.
The extracellular domain contains seven LRR ( leucine-rich repeat ) motifs that mediate protein-protein interactions and participate in cell adhesion and signal transduction . The cytoplasmic tail contains a PDZ domain that mediates cytoskeletal interactions.

(Data source Tsuboi A. Front Mol Neurosci. 2020)
Features of TPBG
TPBG exerts a bidirectional regulatory effect on the Wnt pathway
Inhibits the canonical Wnt/β-catenin pathway: Induces LRP5/6 endocytosis through DKK1 protein, blocks the formation of Frizzled-LRP5/6 complex , leads to β-catenin degradation, and inhibits cell differentiation and adhesion.
Activation of the non-canonical Wnt pathway: Activation of Rho/Rac GTPases through the DKK1-JNK axis promotes cytoskeleton reorganization and enhanced motility, driving tumor metastasis . This dual regulation enables tumor cells to lose their differentiation ability while gaining strong migration, which is an important mechanism of metastasis.
Epithelial-mesenchymal transition (EMT)
Epithelial-mesenchymal transition (EMT) plays an important role in tumor invasion and metastasis. TPBG induces downregulation of E-cadherin expression, enhances cytoskeletal actin reorganization, weakens cell adhesion, and promotes cell morphology changes and metastasis.
These three pathways do not operate independently. TPBG-induced EMT can enhance CXCR4 expression, while the Wnt non-canonical pathway cooperates with CXCL12 to promote metastasis, forming a pro-cancer positive feedback loop .
Regulation of the CXCL12/CXCR4 biological axis
TPBG co-localizes with CXCR4 on the cell membrane, enhancing the chemotactic response mediated by CXCL12; in the absence of TPBG, CXCL12 instead binds to CXCR7, activating the Ras-Raf-MAPK and PI3K-Akt pro-survival pathways.
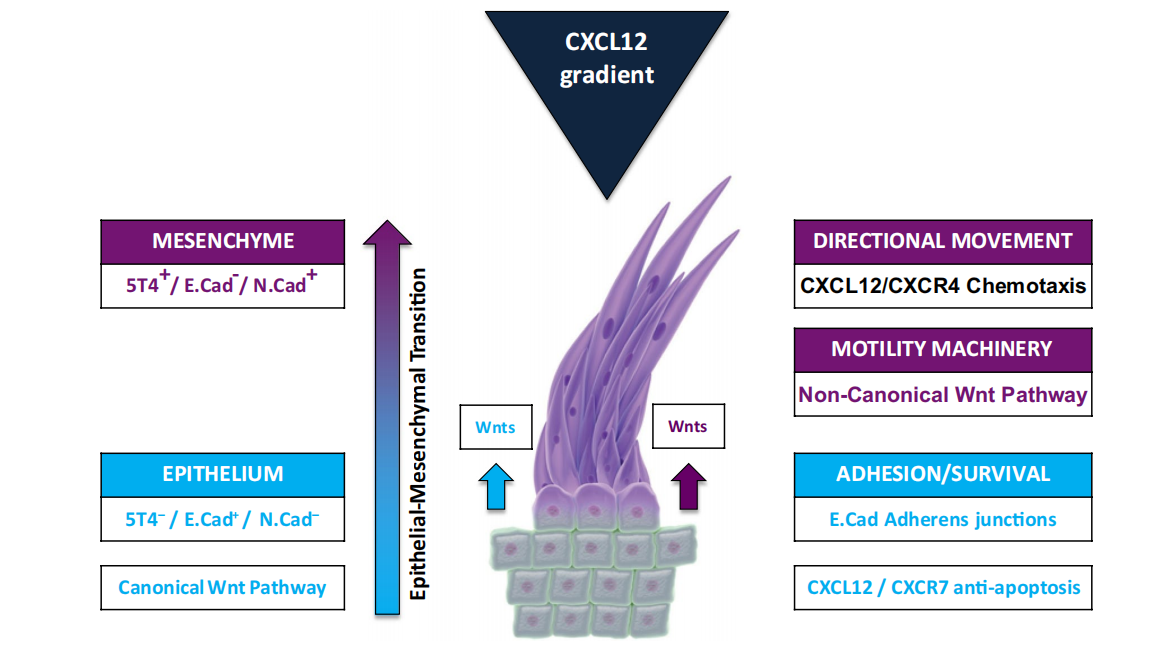
(Data source: Stern PL, Cancer Immunol Immunother. 2017)
Targeted therapy with TPBG
Due to the tumor-specific expression of TPBG, a variety of targeted therapies have entered clinical development, including antibody-drug conjugates (ADCs), tumor vaccines, bispecific antibodies, and cell therapies, among which ADC drugs are the most developed.
Naptumomab estafenatox ( naptumomab ) is a tumor-targeted immunotherapy that targets TPBG , enhancing the immune system's ability to recognize and kill tumors. Developed by Active Biotechs' partner, NeoTX, naptumomab is intended for the treatment of solid tumors. Naptumomab, a fusion protein known as a tumor-targeting superantigen (TTS), comprises a Fab fragment of an antibody that binds to the tumor antigen 5T4, which is expressed on many different types of solid tumors. The antibody portion of naptumomab binds to a bacterial superantigen that activates T cells with specific T cell receptors. Naptumomab works by activating T cells of the immune system and directing them to the 5T4 protein on tumors. This results in the accumulation of large numbers of activated T cells within the tumor, where they kill tumor cells.

This antibody is currently undergoing two clinical studies: an open-label, multicenter, dose-finding Phase Ib/II study combining naptumomab with a checkpoint inhibitor (durvalumab). Results showed that naptumomab combined with durvalumab was well tolerated at the recommended Phase II dose, with limited toxicity. Treatment responses, including complete responses, were observed in patients who would not have expected responses to checkpoint inhibitors alone. Overtreatment with the B-cell therapy obinutuzumab may reduce the development of anti-naptumomab antibodies. An expansion cohort of this study is planned for patients with ovarian cancer.
An open-label, Phase IIa study conducted in the United States evaluated the combination of nivolumab and docetaxel in patients with metastatic or advanced non-small cell lung cancer (NSCLC) who had previously received checkpoint inhibitor therapy. Results were presented at the American Society of Clinical Oncology (ASCO) in June 2024. The combination of nivolumab and docetaxel showed preliminary activity, but did not increase the overall response rate (primary endpoint) compared with docetaxel alone. The safety profile of this combination was acceptable in these heavily pretreated NSCLC patients.

(Data source: Active Biotech official website)
ACR246 , a best-in-class ADC developed by Hangzhou Adcoris and potentially first to market, targets the 5T4 fetal antigen and utilizes a patented topoisomerase I inhibitor. It received clinical trial approval in China in January 2024, with a first-in-human (FIH) trial planned to initiate in the first quarter of 2024. ACR246 is at the forefront of oncology innovation, designed with a stable linker that is cleavable in the tumor microenvironment (TME). It has demonstrated significant anti-tumor activity, safety, pharmacokinetics, and tolerability in preclinical studies involving rodent and non-human primate models. The specificity of 5T4, which is primarily expressed in various solid tumors and is expressed at very low levels in normal adult tissues, highlights the potential of ACR246 for a broad range of tumor applications and marks a new era in precision oncology.

(Data source: adcoris official website)
JK-06 , an ADC being developed by Salubrisbio, is in a Phase 1 dose-escalation study in patients with unresectable locally advanced or metastatic cancer.
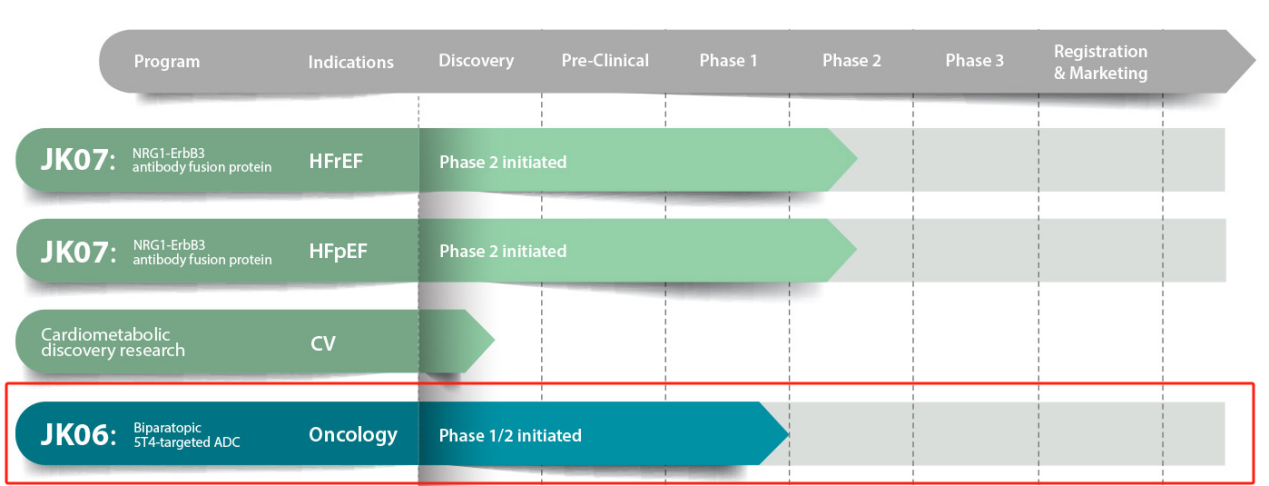

(Data source: Salubrisbio official website)
TUB-030 is an ADC developed by Tubulis targeting 5T4, a cancer-associated antigen expressed in various solid tumors. It consists of an IgG1 antibody targeting 5T4 and the topoisomerase I inhibitor isavucamabine linked via the company's patented P5 conjugation technology, resulting in a drug-to-antibody ratio (DAR) of 8. P5 conjugation is a novel cysteine-selective conjugation chemistry that enables the generation of an ADC with unprecedented linker stability and superior biophysical properties.
Preclinical pharmacokinetic analysis demonstrated that TUB-030 effectively delivers its payload to tumor sites while minimizing off-target toxicities. The drug candidate demonstrated potent and durable responses in multiple preclinical solid tumor models, even at low target expression levels, and possesses an excellent safety profile. TUB-030 is currently being studied in a multicenter Phase I/IIa study (5-STAR 1-01, NCT06657222) designed to evaluate the safety, tolerability, pharmacokinetics, and efficacy of TUB-030 as a monotherapy in patients with advanced solid tumors.

(Data source: tubulis official website)
ALG.APV-527 is a bispecific antibody that targets the immune co-stimulatory receptor 4-1BB and the tumor-associated antigen 5T4. APV-527 directs the stimulation of CD8+ T and NK cells in 5T4+ tumors and is designed to minimize the toxicities observed with other 4-1BB therapies. The Phase I study of ALG.APV-527 (NCT05934539) is a first-in-human, open-label, multicenter trial that includes up to six cohorts (0.1 to 15 mg/kg) with a 3+3 escalation of ALG doses. APV-527 is administered intravenously every two weeks as monotherapy for adult patients with advanced solid tumors. Inclusion criteria are limited to patients whose tumor types may express the 5T4 antigen. ALG.APV-527 has demonstrated favorable tolerability, safety, and biological activity.
ALG.APV-527 was detectable in all patients , and its serum concentration was consistent with the administered dose and predicted exposure; biomarker analysis showed that in tumor biopsies, the expression of targets (4-1BB and 5T4), treatment led to an increase in soluble 4-1BB in the blood, and an increase in the overall number of CD8 T cells in the tumor, confirming the biological activity of ALG.APV-527. In the study, the longest-lasting disease stability occurred in a breast cancer patient who had progressive disease at enrollment and later achieved disease stability, which lasted for more than 11 months in the study. This patient successfully upgraded the dose level twice. One colon cancer patient maintained disease stability for six months in the study. One prostate cancer patient had stable disease, which lasted for more than four months in the study.

(Data source: Aptevo official website)
