Inflammatory chemokines CXCLs (CXCL9, CXCL10, CXCL11, etc.) are primarily induced by interferon (IFN)-γ and share a dedicated chemokine receptor, CXC chemokine receptor 3 (CXCR3). These inflammatory chemokines CXCLs have the prototypical function of guiding the spatiotemporal migration of activated T cells and natural killer cells and inhibiting angiogenesis. Therefore, they are believed to play a role in infection, acute inflammation, autoinflammation and autoimmunity, and cancer.
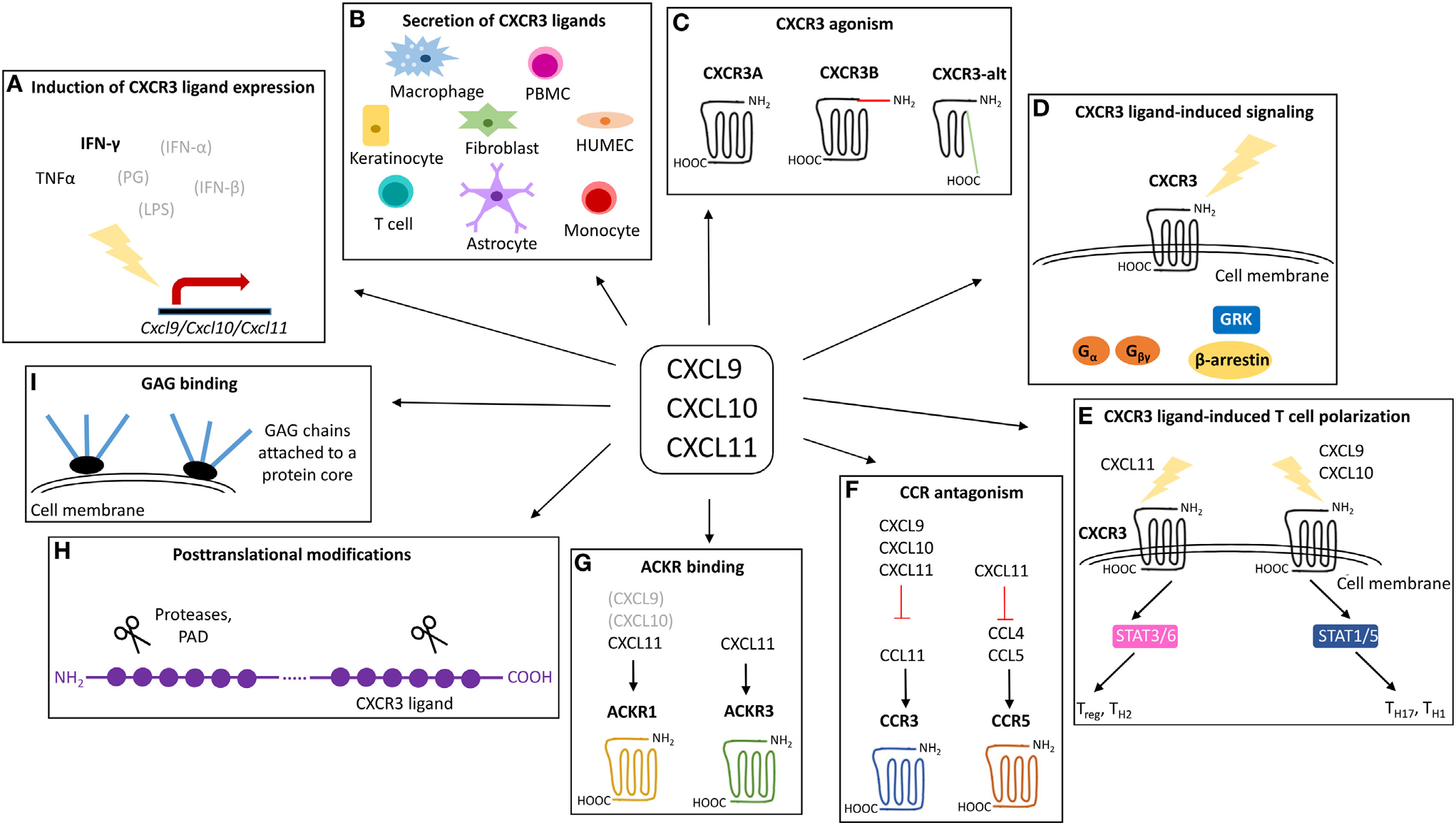
(Data source: Mieke M , et al. Front Immunol. 2018)
CXCL family
The human CXCL chemokine family comprises numerous members: CXCL1-3, CXCL5-8, CXCL9-13, and CXCL16. Most of these members share a conserved glutamic acid-leucine-arginine ("ELR") amino acid motif, comprising three antiparallel β-sheets (pink) and a carboxyl-terminal α-helix (orange), interconnected by 30s, 40s, and 50s loops. A flexible NH2-terminal domain is followed by an N-loop and 310 helix. The three-dimensional structure of the mature secreted protein is stabilized by two disulfide bonds formed by four conserved Cys residues (blue).
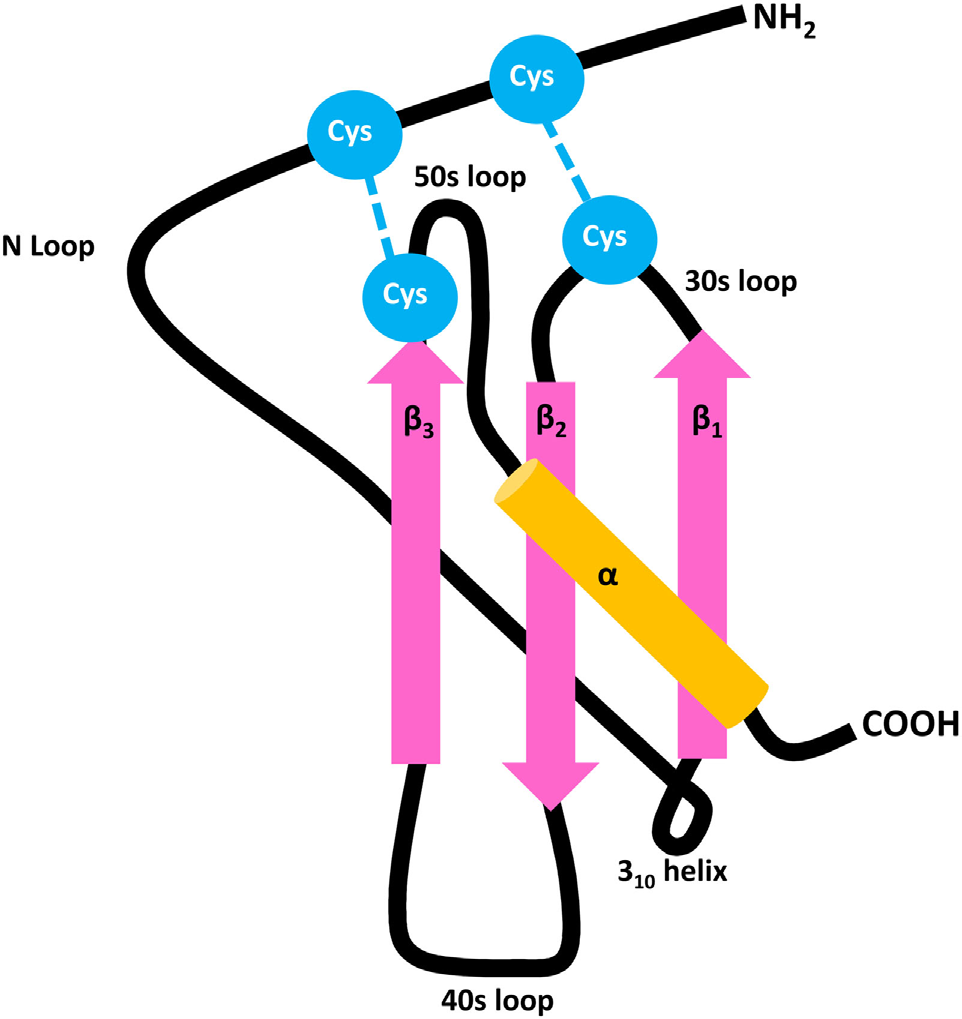
(Data source: Mieke M , et al. Front Immunol. 2018)
CXCL6 and CXCL8 signal through CXCR1; CXCL chemokines with ELR+ motifs activate CXCR2, and activation of these receptors leads to neutrophil chemotaxis; in addition, the CXCR2 ligand is the only ligand for CXCL12 and CXCR4; most CXC chemokines lacking the ELR motif interact with CXCR3, and these CXCR3 ligands can distinguish between the platelet-associated agonists CXCL4 and CXCL4L1 on the one hand and interferon (IFN)-γ on the other. CXCL9, CXCL10, and CXCL11 serve as major inducers; the CXCLI2/CXCR4 axis activates the RAS and MAPK pathways, promoting tumor cell proliferation and enhancing CXCR4 expression through a positive feedback loop; the interaction between CXCL13 and CXCR5 plays a key role in B cell maturation and differentiation, contributing to the production of natural antibodies; CXCL16 interacts with CXCR6 to activate the PI3K-AktPKB pathway, regulating cell activation, proliferation, homing, and migration; CXCL11 and CXCL12 act with CXCR7 through GPCR signaling pathways to regulate cell migration and angiogenesis.
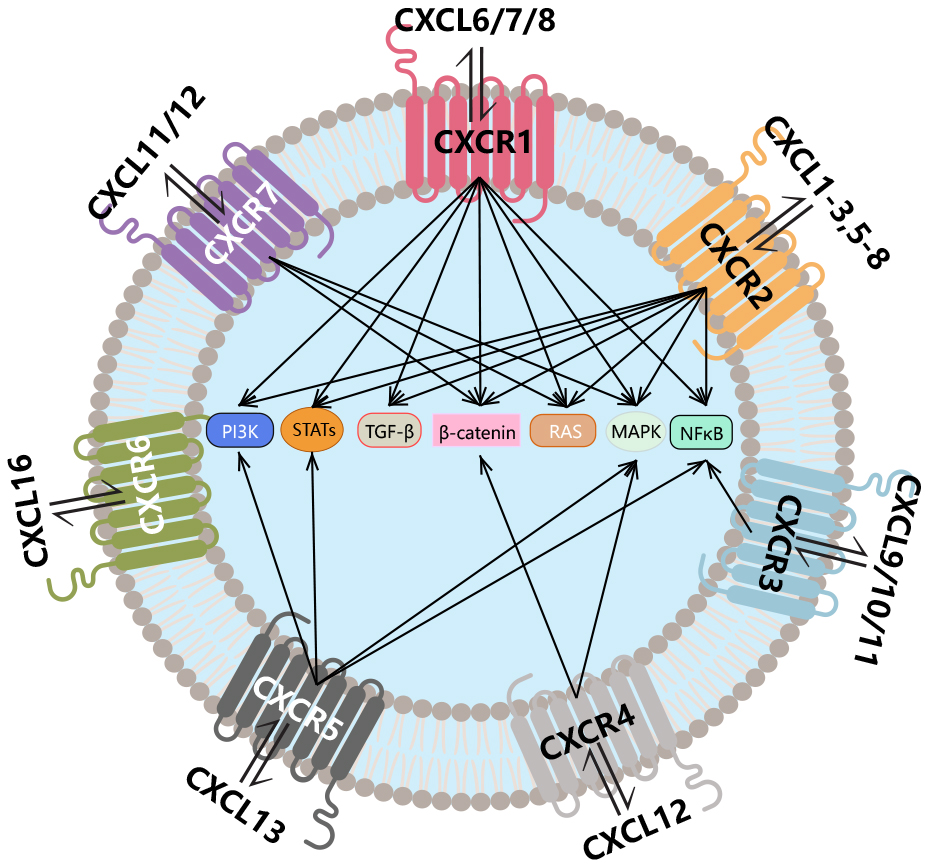
(Data source: Jin X , et al. Front Biosci. 2025)
CXCL10 molecule
The gene encoding CXCL10 is located on chromosome 4, at band q21. Translation produces a 12 kDa protein, which, after enzymatic cleavage under various environmental conditions, is secreted as a 10 kDa protein. Four conserved cysteine residues are located at the N-terminus. The structure of CXCL10 exhibits a typical chemokine fold, consisting of a three-stranded β-sheet capped by an α-helix. Many receptor-binding residues are located in related loops stabilized by disulfide bonds, and the protein forms complexes with different conformations.

(Data source: Mieke M , et al. Front Immunol. 2018)
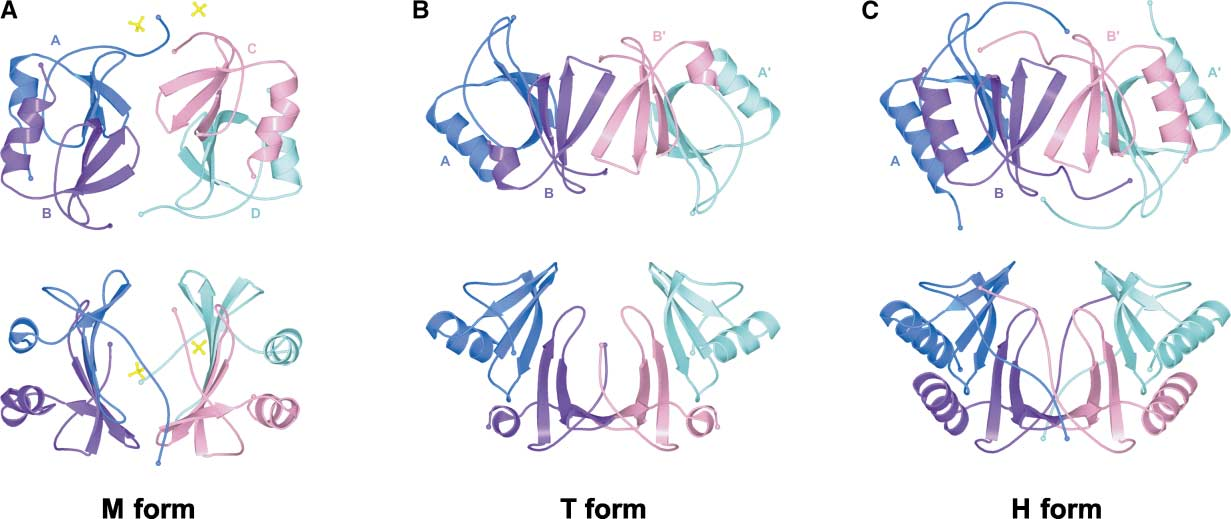
(Data source: G Jawahar S , et al. Structure. 2003)
CXCL10 regulation
when immune cells are stimulated by interferon (IFN)-γ or tumor necrosis factor-α (TNF-α). The CXCL10/CXCR3 axis acts in two main directions:
Paracrine signals are used for immune activation (primarily for the migration, differentiation, and activation of immune cells for paracrine signaling. Through this axis, immune responsiveness recruits CTLs, NK cells, and macrophages, activates TLRs and RNA helicases, leading to the release of IFN-α/β by tissue cells and endothelial cells, followed by the secretion of chemokines such as CXCL10; CXCL10 recruits NK and CD4+ Th1 cells into target tissues, releasing IFN-γ in an antigen-specific manner; IFN-γ produced by recruited CD8+ CTLs further stimulates tissue cells to produce more CXCL10. Increased chemokine release and inflammatory responses are amplified, leading to further recruitment of Th1 and CD8+ TIL cells expressing CXCR3.).

(Data source: Xiaoming W , et al. Front Oncol. 2022)
Autocrine signaling is used for the proliferation and metastasis of cancer cells (cancer cells are prone to metastasis due to the activity of tumor-derived CXCR3 ligands, primarily through CXCR3A. Tumor-derived chemokines are also responsible for the recruitment of Th cells, Tregs, and MDSCs, which play a role in creating a tumor-friendly microenvironment.).
CXCL10 -targeted therapy
CXCL10 is involved in the body's response to various viral (such as influenza, hepatitis, HIV) and bacterial infections. Elevated CXCL10 levels are associated with chronic inflammation, immune dysfunction, and autoimmune diseases (such as systemic lupus erythematosus and multiple sclerosis). It plays a complex role in cancer, influencing tumor development, the infiltration and metastasis of immune cells into tumors, and is considered an important biomarker for multiple conditions, making it an attractive target for potential therapeutic strategies aimed at modulating immune responses in disease.

(Data source: Noha M , et al. Viruses. 2022)
Eldelumab (BMS-936557, formerly known as MDX-110067), a fully human monoclonal antibody targeting the CXCR3/CXCL10 signaling axis, is the primary candidate for development. Eldelumab was evaluated in a Phase IIa, multicenter, double-blind, randomized, placebo-controlled trial for the treatment of moderate to severe ulcerative colitis. However, no significant differences were observed between the treatment and placebo groups on the primary or secondary endpoints. Consequently, development of this pipeline drug has been discontinued.

(Data source: Elisabetta B , et al. Biomedicines. 2023)
Inhibition of the CXCR3-CXCL10 pathway appears safe but has failed to demonstrate overall efficacy, suggesting that inhibition of CXCR3-CXCL10 alone is insufficient to induce clinical responses. Further defining patients based on clinical characteristics, or more relevant immune phenotypes, may allow treatment to be targeted to patient subpopulations more likely to achieve therapeutic effects.
