The complement system is an important component of innate immunity and can be activated through three pathways: the classical pathway (CP), the alternative pathway (AP), and the lectin pathway (LP).

(Data source: Thomas D , et al. Front Immunol. 2023)
The lectin pathway (LP) was discovered relatively recently. Mannose-binding lectin (MBL), other lectins, and lipopolysaccharides are collectively referred to as LP pattern recognition molecules (PRMs), responsible for activating the LP into molecular patterns. MBL-associated serine proteases (MASPs) serve as effector molecules, while MBL-associated proteins (MAps) have regulatory functions. When PRMs bind to target surfaces, the associated serine proteases are activated and initiate the complement cascade by cleaving and activating subsequent components. Because a single active protease can cleave and activate numerous zymogen molecules, this proteolytic cascade has enormous potential for amplification. Indeed, if complement activation occurs uncontrolled, it can contribute to the development and progression of serious diseases.

(Data source: Daniel R , et al. Nat Rev Nephrol. 2016)
Serine proteases MASP
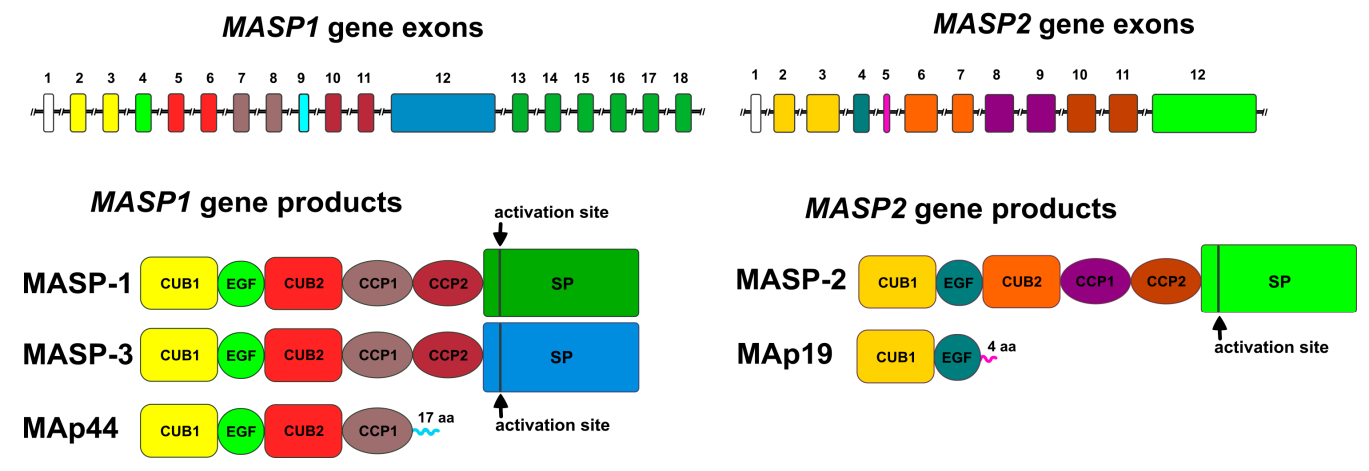
MBL-associated serine proteases (MASPs) are encoded by the MASP1 and MASP2 genes. They form different product proteins through alternative splicing or selective polyadenylation, serving as effector molecules for lectin pathway activation. Among them, MASP 1 and MASP 2 are responsible for triggering the activation of the lectin pathway, while the third component, MASP 3, is involved in the function of the alternative pathway (AP) of the complement system.
Aggregation of PRM/MASP1 and PRM/MASP2 complexes at the activation surface triggers LP activation; PRM/MASP3 complexes are cleaved by PCSK6, followed by pro-FD cleavage by MASP3 to activate the AP (red arrows indicate proteolytic cleavage, while blue arrows indicate non-enzymatic events. Brown indicates PRM, and gray oval indicates the activation surface.).
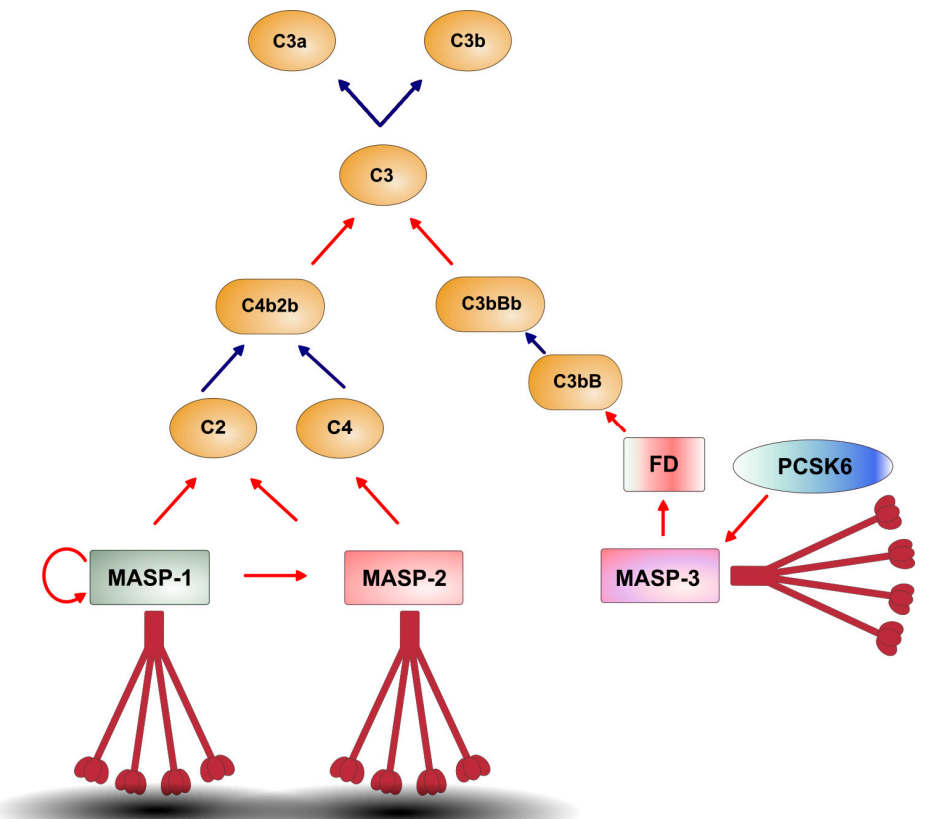
(Data source: Dobó J, et al. Int J Mol Sci. 2024)
MASP structure
All MASP proteins have the same structure: a heavy/α chain and a light/β chain connected by a cysteine disulfide bond. The heavy chain consists of two CUB domains and two complement regulatory protein (CCP) domains, connected by an epidermal growth factor (EGF) segment.

(Data source Alexandre R , et al. Structure. 2011)
MASP regulatory mechanisms
When complement encounters an immunogen, recognition molecules such as C1q, mannose-binding lectin (MBL), fibrinogen-like proteins (ficolins), and collectin-11 recognize antibody clusters or pathogen-associated molecular patterns (PAMPs) and activate associated serine proteases (MASPs). These proteases cleave C4 and C2 to form C3 convertases (C4b2b) of the classical pathway (CP) and lectin pathway (LP) on the triggering cell. This process activates more C3 to produce C3b, promoting an amplified cycle. Increased C3b concentrations favor the generation of C5 convertases, which activate C5 to release the anaphylatoxins C5a and C5b. These enzymes, after stepwise interaction with C6-C9 and membrane insertion, form membrane attack complexes (MACs), leading to target cell lysis, damage, or activation. The released anaphylatoxins act as immune mediators, particularly in the case of C5a, attracting and activating immune cells.

(Data source: Daniel R , et al. Nat Rev Nephrol. 2016)
Targeted therapy of MASPs
There are two main directions for developing inhibitors around MASP targets: one is targeting MASP2 (such as Narsoplimab). Although both MASP1 and MASP2 cleave C2, only MASP2 cleaves C4, allowing the formation of the C3 convertase C4b2a. In addition, inhibiting MASP-2 does not seem to interfere with the classical complement pathway, which is an important component of the acquired immune response after infection. This blocking drug is designed to prevent complement-mediated inflammation and endothelial damage while keeping the corresponding functions of other innate immune pathways unaffected; the other is targeting MASP 3 (such as Zaltenibart), because MASP3 is the only activating factor for the conversion of pro-FD to FD, and is a key regulator of FD cleavage of FB and the formation of the alternative pathway (AP) C3 convertase C3bBb. Inhibiting MASP 3 has potential therapeutic effects in inhibiting complement activation, inflammation and microvascular stasis.

(Data source: John D , et al. Transl Res. 2022)
Interestingly, the company currently developing targeted antibody therapies for MASP is mainly Omeros, and judging from its pipeline, the company is also very confident about the prospects of the MASP target.

(Data source: Omeros official website)
