Matrix metalloproteinase-9 (MMP9) can specifically degrade type IV collagen and plays an important role in local proteolysis of the extracellular matrix and leukocyte migration. Due to its proteolytic activity, it plays a key role in tumorigenesis by regulating cancer cell migration, epithelial-mesenchymal transition and survival, inducing immune responses, angiogenesis, and the formation of the tumor microenvironment.
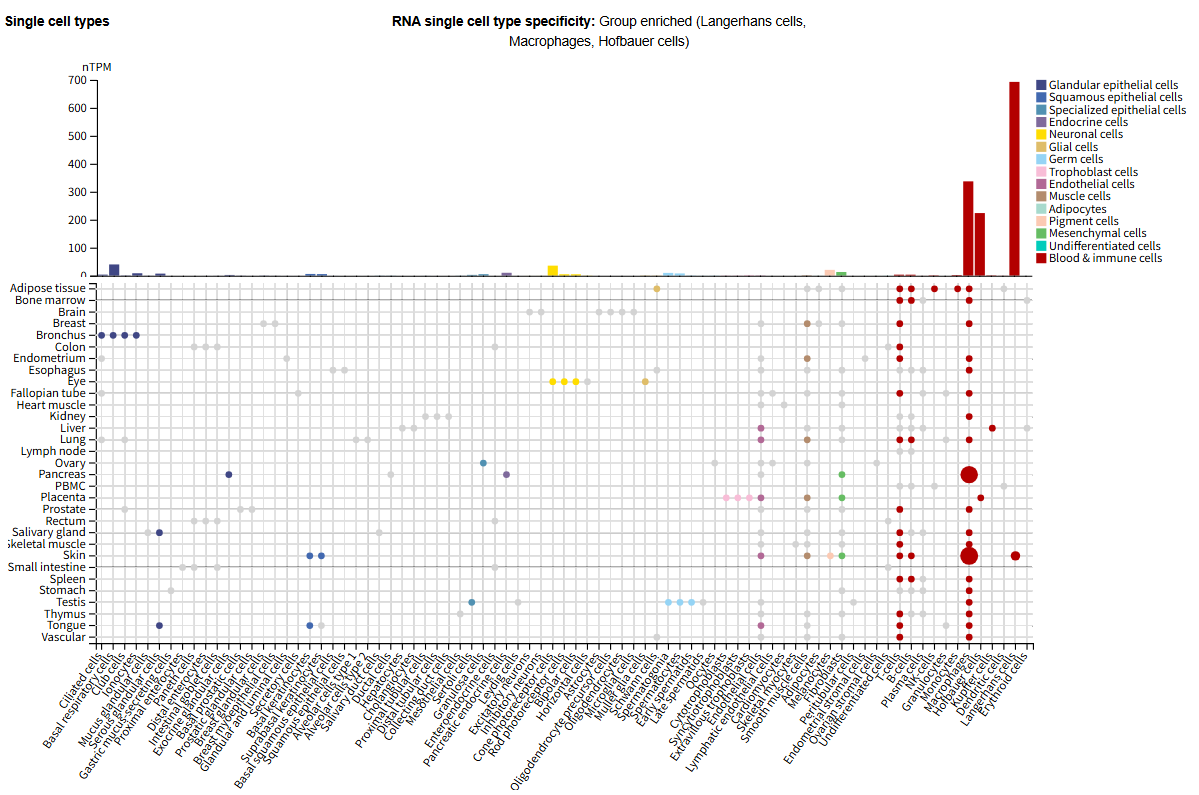
Expression distribution of MMP9
MMP9 is mainly expressed in Langerhans cells, macrophages, and Hofbauer cells. Under normal physiological conditions, neutrophils are the main source of MMP9 and play an important role in the body's natural immune response.

(Data source: Uniprot)
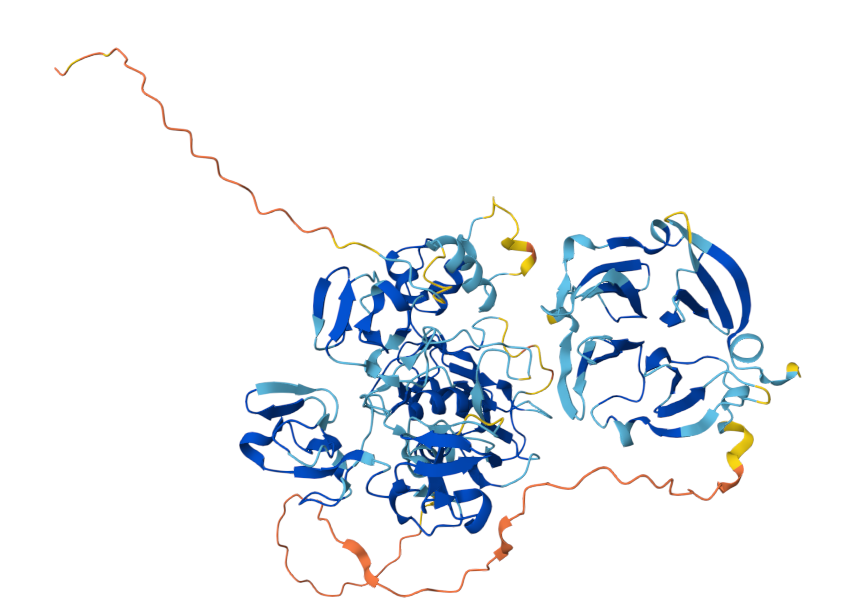
The structure of MMP9
MMP-9 is a secreted protein that is synthesized as a proenzyme and then transported to the extracellular environment as pro-MMP9-9 enzyme. Activated MMP-9 is generated by protease-mediated cleavage of the MMP-9 proenzyme. MMP-3 is an example of a protease that activates MMP-9 by removing the N-terminal propeptide region. Removal of the N-terminal propeptide significantly disrupts the latency of the MMP.

(Data source: Wang Y, et al. Biomed Pharmacother. 2024)
The MMP-9 gene is located on chromosome 20q13.12 and contains 13 exons and 12 introns. The protein is composed of the following domains: a signal peptide domain, a propeptide region, a catalytic domain, a hemopexin-like domain, and a hinge region.
Signal peptide: It guides the protein into the endoplasmic reticulum during synthesis and is subsequently removed and does not exist in the mature protein.
Propeptide Domain: Contains a highly conserved " cysteine switch " sequence (PRCGVPDV). This domain maintains the enzyme's inactive state (proenzyme). The cysteine residues within it coordinate with the catalytic zinc ion, preventing substrate access to the active center. When this domain is disrupted by external proteases (such as trypsin and plasmin) or chemical modifications (such as organomercurial reagents), MMP9 becomes activated. Catalytic Domain: Contains the highly conserved zinc binding site (HEXXHXXGXXH motif) of the MMP family, which is the core of catalytic activity. A structural zinc ion and calcium ion stabilize the three-dimensional structure of the catalytic domain. It is responsible for the hydrolytic cleavage of substrate proteins.
Hinge Region (Linker Peptide): A flexible peptide linking the catalytic and hemopexin domains. This region allows for relative motion between the two domains and is crucial for recognition of certain macromolecular substrates and specificity.
Hemopexin-like domain (PEX Domain): A β-propeller-like structure composed of four blades. This domain is not directly involved in catalysis but recognizes specific substrates (such as native type IV collagen). It binds to tissue inhibitors of TIMPs (particularly TIMP-1) and interacts with cell surface receptors (such as CD44) to mediate cell localization and function.
(Data source: Alphafold)
Signal transduction of MMP9
The expression of MMP9 is regulated by multiple transcription factors, such as activator protein 1 (AP-1), nuclear factor-κB (NF-κB), and specificity protein 1 (SP-1). These transcription factors can bind to the MMP9 gene promoter, initiating transcription and promoting its expression. During inflammation, NF-κB activation can induce the expression of MMP9, thereby participating in inflammation-related tissue remodeling and cell migration. MMP9 degrades extracellular matrix components, releasing a variety of bioactive molecules such as growth factors, cytokines, and chemokines. These molecules can activate downstream signaling pathways such as MAPK/ERK, PI3K/AKT, and FAK, thereby regulating cell proliferation, migration, invasion, and survival.

(Data source: Wang Y, et al. Biomed Pharmacother. 2024)
Targeted therapy of MMP9
MMP9 is an attractive target for anticancer therapy. However, on the one hand, the expression, synthesis and activation regulation mechanisms of MMP9 are very complex, which determine the specific and often contradictory effects of this enzyme. On the other hand, the high homology with other members of the MMP family makes the development of effective and safe MMP9 inhibitors as anticancer drugs extremely difficult.
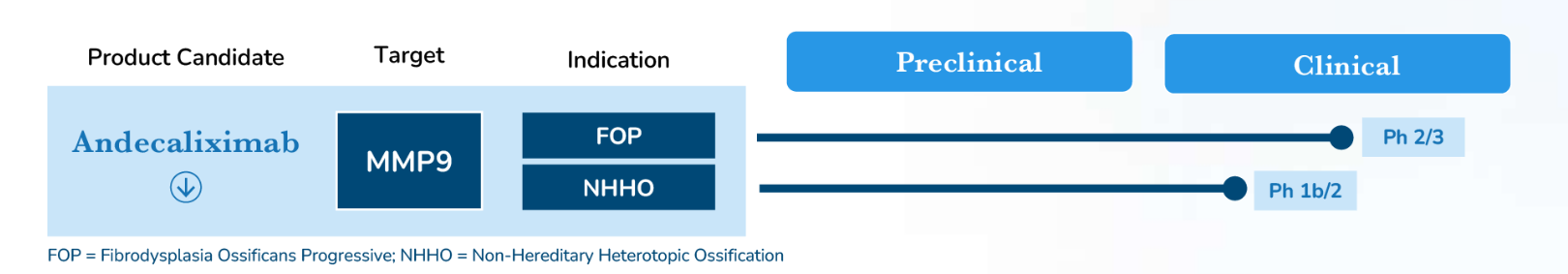
Andecaliximab is an IgG4 monoclonal antibody targeting MMP9, currently under clinical development for the treatment of fibrosus ossification progressive (FOP) and non-hereditary heterotopic ossification (NHHO).
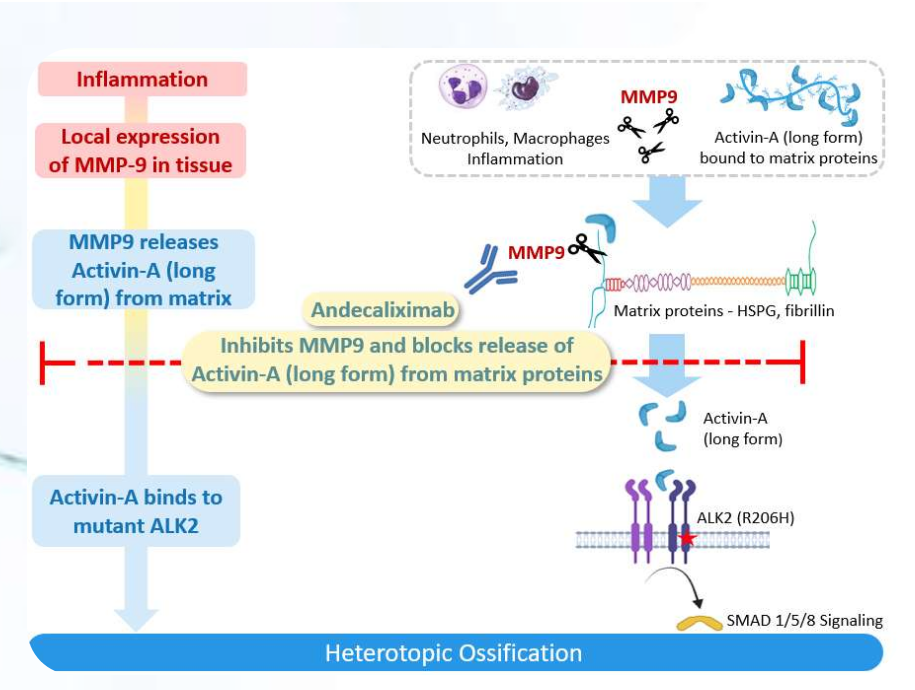
In FOP, MMP9 is a key enzyme that releases the long form of Activin-A in inflamed tissues. Activin-A then binds to the mutant ALK2 receptor, leading to the formation of HO. Studies in mouse FOP models have shown that blocking MMP9 activity significantly inhibits HO formation. Andecaliximab is a potent and specific inhibitor of MMP9.
In NHHO, MMP9 is highly expressed at sites of trauma and inflammation and is a key enzyme in the release of bone morphogenetic protein (BMP) ligands into local tissues. BMP ligands (e.g., BMP 2, 4, and 6) then bind to wild-type ALK2 receptors, leading to HO formation. Studies conducted by āshibio in a mouse model of trauma-induced HO have demonstrated that MMP9 inhibition leads to a significant reduction in HO.
(Data source: ashibio official website)
