Prolactin receptor (PRLR) acts as a pro-survival factor on sperm by inhibiting sperm maturation by inhibiting SRC kinase activation and stimulating AKT. PRL/PRLR signaling promotes the occurrence and progression of breast cancer.
Expression distribution of PRLR
PRLR is mainly expressed in endometrial stromal cells, mammary glandular cells, syncytiotrophoblast cells, prostate basal cells, and proximal renal tubular cells.
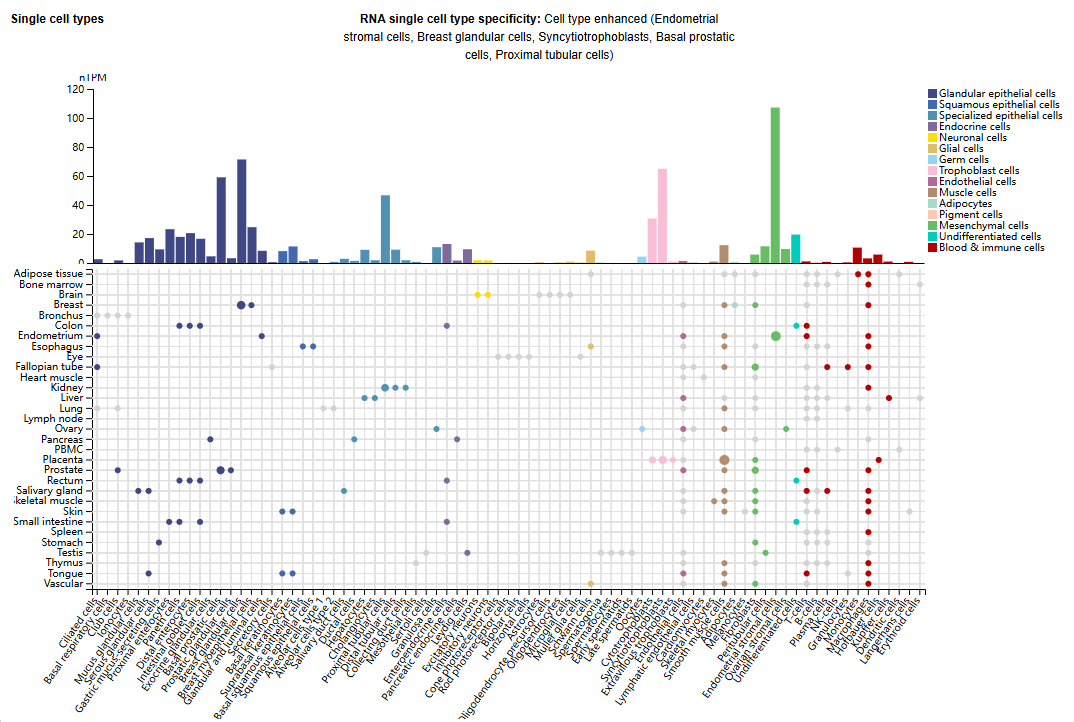
(Data source: unprot)
Structure of PRLR
PRLR is a type I membrane protein composed of 622 amino acids with three major domains: extracellular, transmembrane, and intracellular.
The extracellular domain: The two fibronectin type III (FnIII) domains, D1 and D2, are hallmark features of the type I cytokine receptor family. The WS motif in D2 acts as a molecular switch during ligand binding and activation of the PRLR. The prolactin binding interface primarily involves the D2 domain and the hinge region connecting D1 and D2.
The Transmembrane domain: PRLR has a single-pass transmembrane chain, which does not have kinase activity. The receptor chain relies on associated kinases to transduce phosphorylation-based signaling cascades.
The intracellular domain: Consists of two subdomains: Box 1 and Box 2. Box 1, a conserved proline-rich sequence (Pro-XX-Pro-X-Pro), is crucial for binding and activation of the Jak2 kinase. Box 2, a conserved hydrophobic region, also contributes to Jak2 recruitment and stabilization. The long receptor contains an intact Box 2, while the short receptor is missing or partially absent.
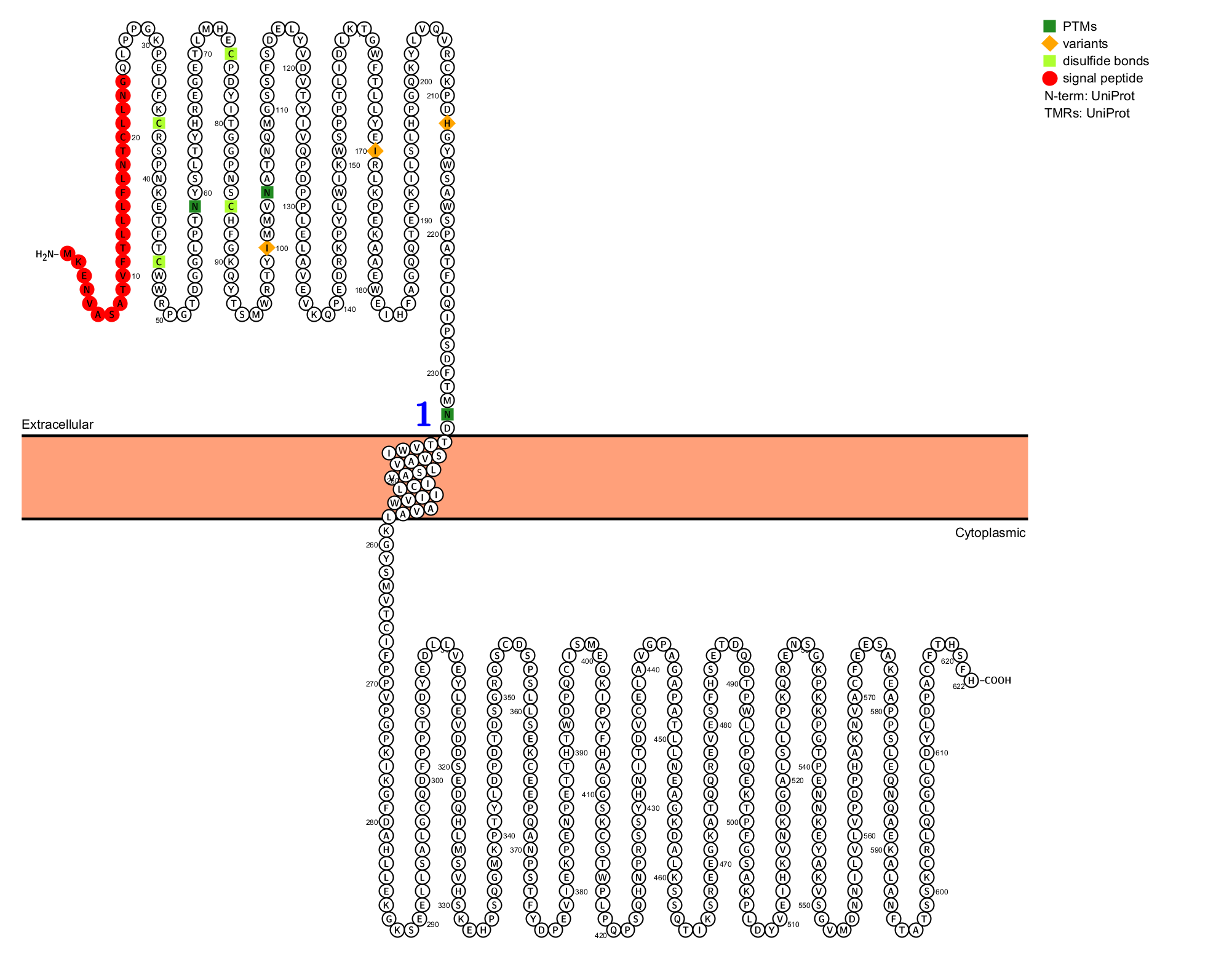
(Data source: uniprot)
Signaling pathway and regulation of PRLR
PRL binds to the PRLR dimer, leading to conformational changes and activation of the JAK/STAT pathway, MAPK, and SRC kinases. These signaling pathways are involved in lactation, immune response, cytoskeletal remodeling, and cell growth, proliferation, and survival.

(Data source: Kavarthapu R, et al. Front Endocrinol. 2022)
The signaling cascade induced by PRL/PRLR promotes tumor cell proliferation, survival, invasion, and metastasis, inducing therapeutic resistance (particularly resistance to tamoxifen). EGF released from the stromal microenvironment surrounding breast tumors activates a signaling cascade that overlaps with the PRLR signaling cascade when activated by PRL secreted by breast tumor cells. PRL stimulates HER2 and EGFR signaling pathways through JAK2. EGF/EGFR also indirectly activates STAT5 signaling through s-SRC. This crosstalk between receptors can increase breast tumor progression and endocrine resistance.
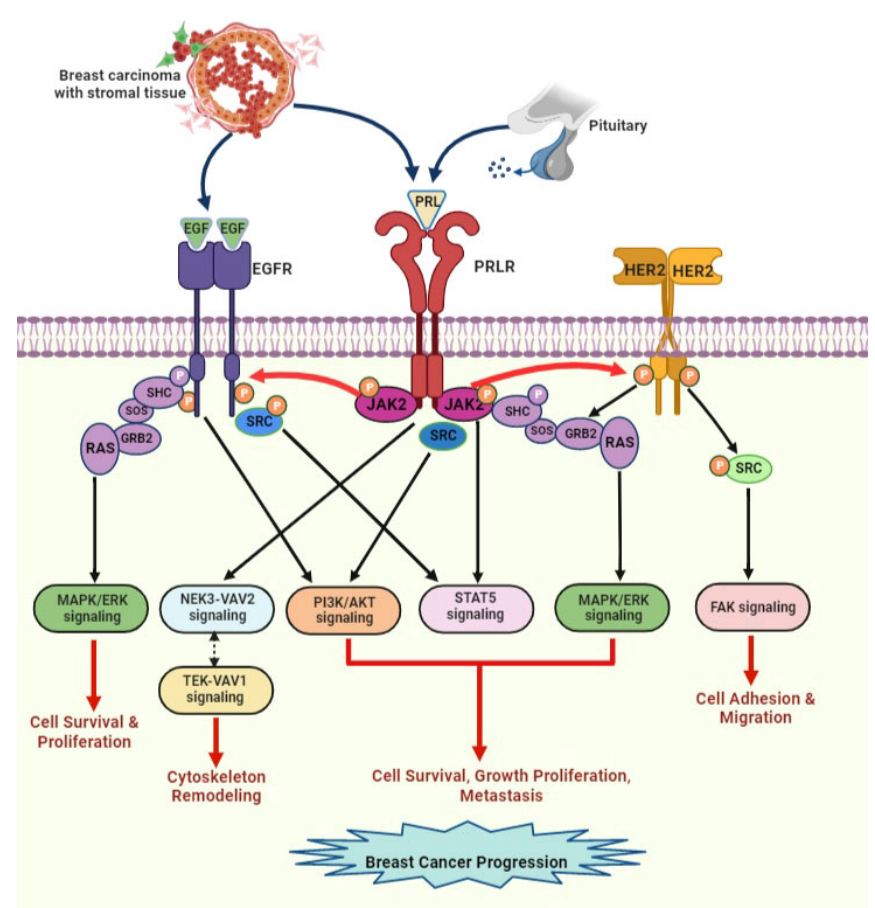
(Data source: Kavarthapu R, et al. Front Endocrinol. 2022)
PRLR targeted therapy
Rolinsatamab, a monoclonal antibody targeting PRLR developed by AbbVie for the treatment of solid tumors, was associated with significant toxicity in a Phase 1, dose-escalation study (NCT03145909). Although cytopenias were often dose-limiting, effusions and edema were also common and occurred later, suggesting cumulative toxicity. Although data were obtained from a small group of patients whose tumors varied in PRLR expression, no responses were observed. The study was terminated after 19 patients were treated.
BAY-1158061 (also known as HMI-115) is a monoclonal antibody targeting PRLR. Its global clinical development is currently led by Hope Medicine Inc. It was originally discovered by Bayer. HMI-115, a first-in-class monoclonal antibody for which Bayer holds global rights, has been designated a Breakthrough Therapy Designation (BTD) by the Center for Drug Evaluation (CDE) of the China National Medical Products Administration (NMPA) for the treatment of moderate to severe pain associated with endometriosis. HMI-115 is currently in a global Phase 2 clinical trial for both endometriosis and androgenic alopecia (CDE Clinical Trial Registration Number: CXSL2200442). In the first 102 patients included in the interim analysis, HMI-115 demonstrated statistically significant improvement in endometriosis-related pain. HMI-115 also demonstrated a favorable safety profile, with most patients reporting normal menstruation, no typical perimenopausal symptoms, and no significant changes in bone density or key sex hormone levels.

(Data source: hopemedinc official website)
