Killer cell lectin-like receptor subfamily B member 1 (KLRB1), also known as CD161, plays an inhibitory role in natural killer (NK) cell cytotoxicity. As a lectin, it binds to the terminal carbohydrate Gal-alpha (1 , 3) Gal epitope and the N-acetylgalactosamine epitope. It also acts as a ligand for CLEC2D/LLT1, inhibiting NK cell-mediated cytotoxicity and interferon-γ secretion in target cells.
Expression distribution of KLRB1
KLRB1 (CD161) is primarily expressed in NK cells and is also expressed in some T cells. Among T cells, CD161 was initially reported to be expressed on memory/effector CD4+ and CD8+ αβT cells, γδT cells, and NKT cells. CD161 expression is maintained across all functional lineages of the Th17 lineage, making it a consistent and stable marker for these pathogenic populations.
LLT1 is not expressed on immature dendritic cells (DCs), but its expression is induced on TLR-activated DCs and plasmacytoid DCs (pDCs). On B cells, LLT1 expression is induced upon BCR crosslinking with CD40 and TLR stimulation. LPS and CpG stimulation strongly induce LLT1 expression in monocyte-derived DCs and CpG-activated pDCs.

(Data source: Braud VM, et al. Front Immunol. 2022)
Structure of KLRB1 and its ligands
CD161, a type II transmembrane protein of the C-type mitochondrial family encoded by KLRB1, is a 40 kDa dimer composed of two subunits.
Extracellular domain: Contains a C-type lectin-like domain, which is the key region for its recognition and binding to the ligand LLT1. Unlike some other lectins, KLRB1 has a mutation in the calcium binding site, making its binding independent of calcium ions.
Transmembrane region: Contains a positively charged lysine residue, which helps it bind to negatively charged transmembrane adaptor proteins (such as FCεRIγ) to form a signal transduction complex.
Intracellular domain: It’s very short and lacks the classic signaling motif, indicating that KLRB1 cannot directly transmit signals downstream and must rely on its partner proteins to complete signal transduction.
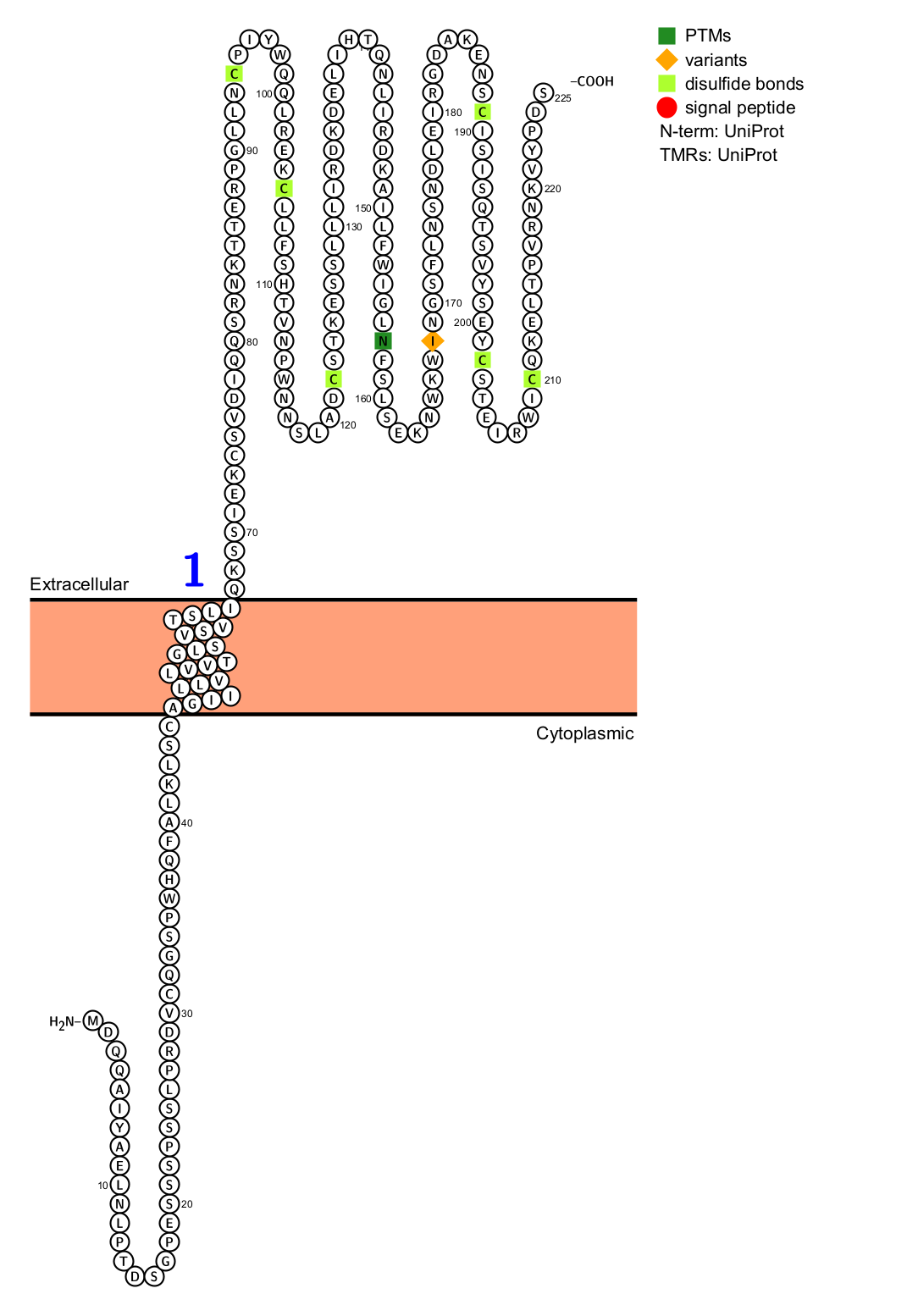
(Data source: uniprot)
The role of CD161 in immune cells
CD161 is a key surface marker of Th17 cells, Tc17 cells, mucosal-associated invariant T cells (MAIT), and some γδ T cells. These cells rapidly produce proinflammatory cytokines and tend to home to inflamed tissues, driving pathological processes. CD161⁺CD4⁺ T cells are the primary source of multiple proinflammatory cytokines, including IL-17A, IL-17F, IL-22, and IFN-γ. CD161⁺CD8⁺ T cells possess cytotoxicity and produce multiple cytokines, such as IL-17, IFN-γ, and GM-CSF. They exhibit unique tissue homing properties, enabling them to penetrate tissues such as the blood-brain barrier.
Targeting CD161⁺ T cells can selectively eliminate pathogenic cells and inhibit the simultaneous production of multiple cytokines that promote inflammation in autoimmune diseases.
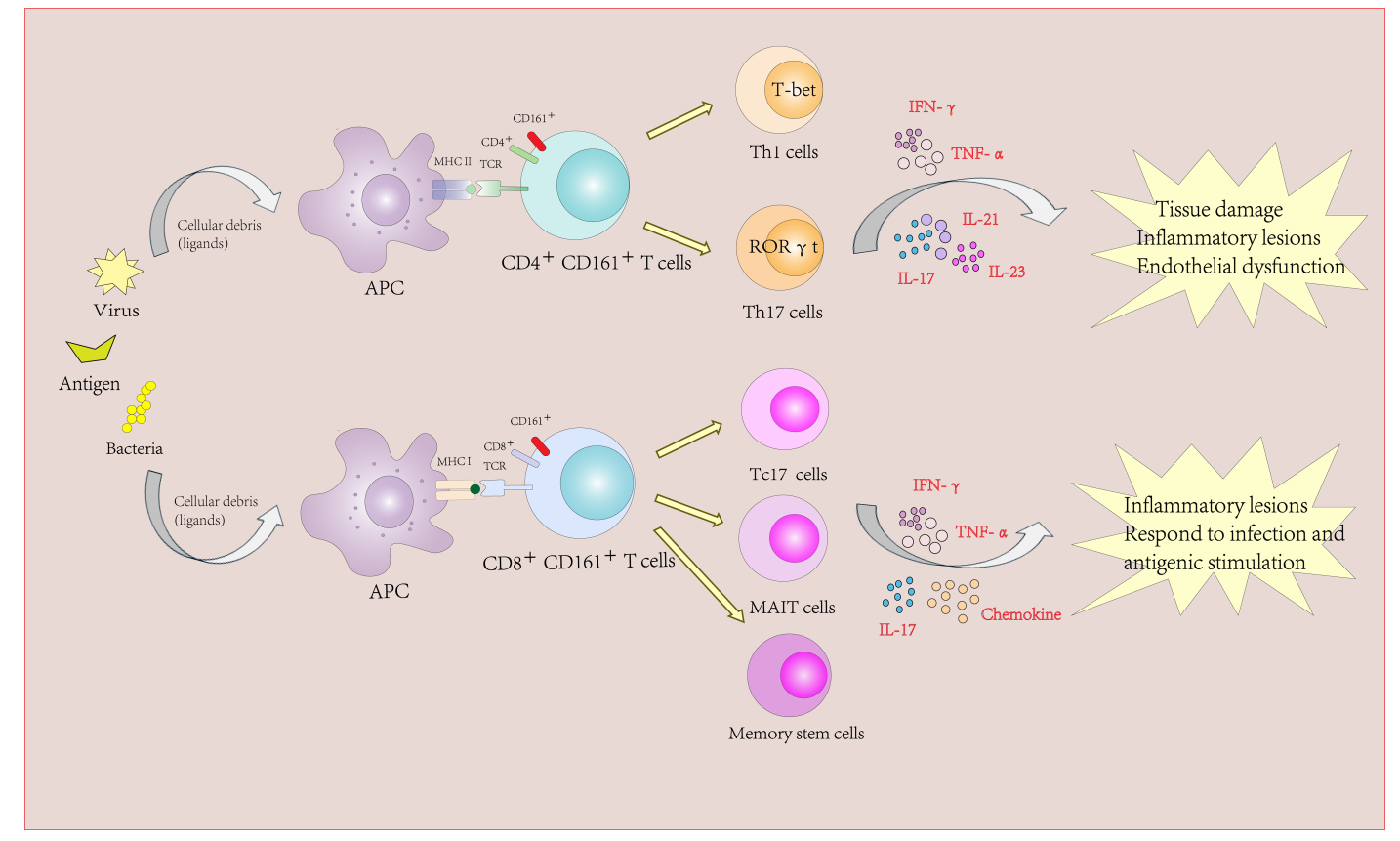
(Data source: Tong B, et al. Front Immunol. 2025)
LLT1/CD161 interaction in cancer immune surveillance
LLT1 is expressed under inflammatory conditions and CD161 is widely expressed in NK/ILC and T cell populations, suggesting that the LLT1/CD161 interaction plays a role in cancer immune surveillance.
Inhibiting NK cell function and promoting tumor immune escape: LLT1 is expressed on the surface of various tumor cells, such as B-cell lymphomas, gliomas, prostate cancer, and triple-negative breast cancer. Binding to CD161, LLT1 inhibits NK cell cytotoxicity and cytokine secretion. Blocking this interaction can restore NK cell killing capacity. This constitutes a tumor immune escape mechanism, particularly in LLT1-expressing lymphomas and solid tumors.
When LLT1 is expressed on B cells in tertiary lymphoid structures (TLS), it is a marker of effective antitumor immunity and is associated with better prognosis.
When CD161 is expressed on tumor-infiltrating T cells, it typically identifies a functionally active, non-exhausted T cell subset.
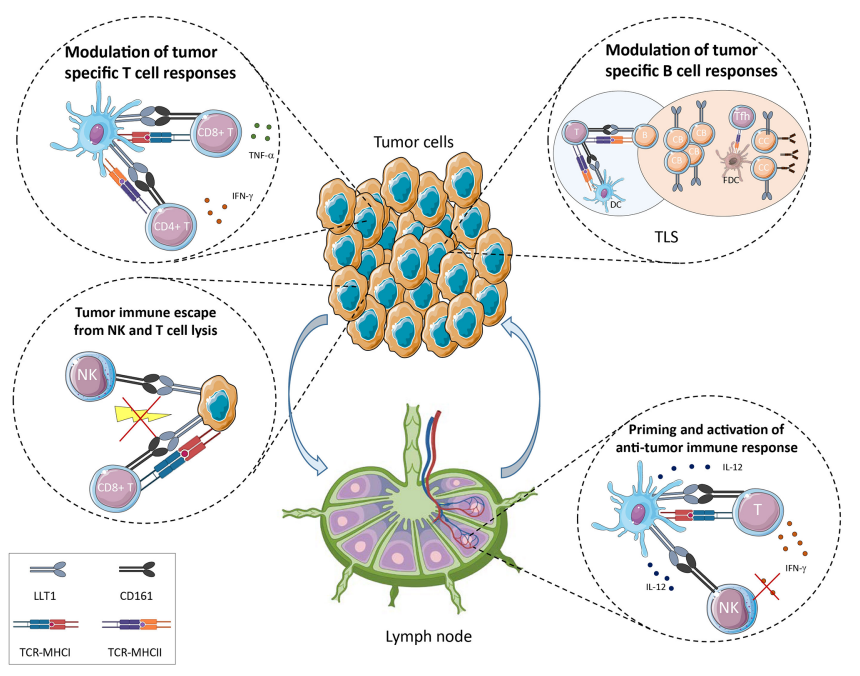
(Data source Braud VM, et al. Front Immunol. 2022)
Targeted therapy for KLRB1
Rezorstobart (IMT-009), a monoclonal antibody targeting KLRB1, is being developed by Immunitas Therapeutics and is currently in clinical trials for patients with various difficult-to-treat tumors, including MSS colorectal cancer, head and neck cancer, and non-small cell lung cancer. Preclinical data suggest that blocking CD161 with IMT-009 enhances anti-tumor activity. IMT-009 is being evaluated in a Phase 1/2a clinical trial as a monotherapy and in combination with solid tumors and hematologic malignancies. The Phase 1 study aims to assess the safety, tolerability, pharmacokinetic biomarkers, and preliminary efficacy of IMT-009 and to determine the recommended Phase 2 dose (RP2D).
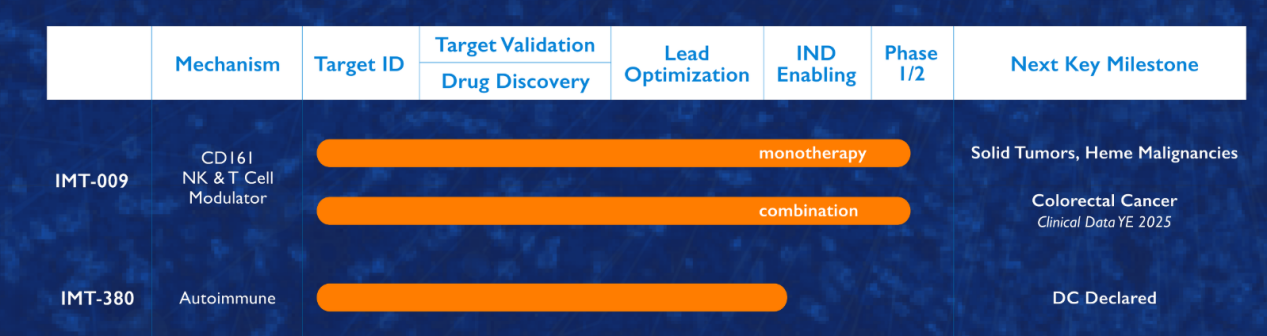
IMT-380 is another KLRB1-targeting monoclonal antibody developed by the company and has been focused on enhancing antibody-dependent cellular cytotoxicity and antibody-dependent cellular phagocytosis. Anti-CD161 therapy is a promising strategy to restore immune balance, enhance therapeutic durability, and provide a safer and more targeted alternative to broad immunosuppression. IMT-380 is the first antibody to selectively eliminate CD161⁺ cells, thereby benefiting in autoimmune diseases.
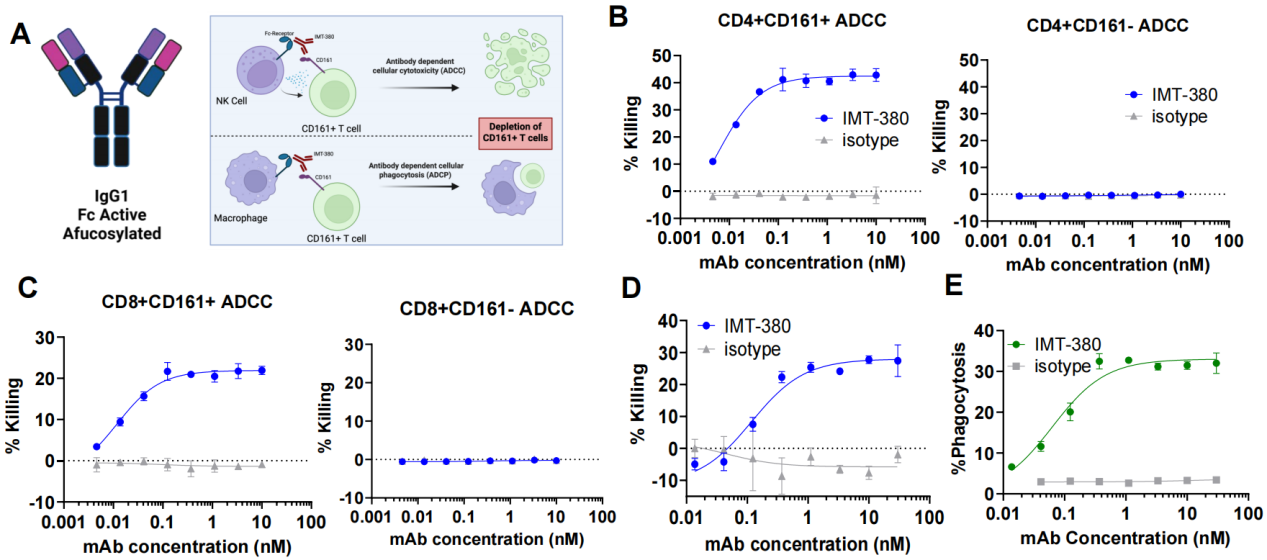
(Data source: Immunitas Therapeutics official website)
