KLRC1 is an NKG2-A/NKG2-B type II integral membrane protein and an NKG2-A/B-activated natural killer receptor, also known as CD159a. Binding of CD94/NKG2A to its ligand HLA-E suppresses immune activity and helps prevent the development and progression of autoimmune diseases. The NKG2A-HLA-E pathway serves as a key checkpoint in anti-tumor responses, paving the way for novel immunotherapy strategies to improve outcomes for cancer patients.

(Data source: Li Y, et al. J Immunother Cancer. 2024)
Expression distribution of KLRC1
KLRC1 is primarily expressed on the surface of NK cells and T cells, and some T cells including Th2 cells, αβ CD8+ T cells and γδ CD8+ T cells (Vδ1 and Vδ2).
The non-classical major histocompatibility complex (MHC) class I molecule HLA-E is the main ligand for KLRC1, which is expressed at low levels on different cells and is upregulated in many cancer types.

(Data source: uniprot)
Structure of KLRC1 and its ligands
NKG2A is a member of the C-type lectin superfamily. Its gene is located in the natural killer (NK) complex region of chromosome 12 and consists of seven exons. NKG2A is a single-span transmembrane type II integral membrane glycoprotein containing cytoplasmic, transmembrane, and extracellular lectin-like domains. Its intracellular portion possesses two ITIMs that are involved in inhibitory signal transduction. NKG2A and a NK cell surface membrane protein CD94 encoded by the killer cell lectin-like receptor D1 form a disulfide-linked heterodimeric complex CD94/NKG2A.

(Data source: Li Y, et al. Front Immunol. 2014)
KLRC1–HLA-E immune checkpoint regulation
The interaction between HLA-E expressed on cancer cells and CD94/NKG2A has been shown to limit the anti-tumor responses of CD8+ tumor-infiltrating T cells and NK cells across various cancer types.
Peptide-loaded HLA-E on tumor cells interacts with NKG2A-CD94 on CD8+ T cells, leading to phosphorylation of NKG2A's cytoplasmic immunoreceptor tyrosine-based inhibitory motifs (ITMs), activating the intracellular phosphatase SHP1. This inhibits activation signals entering through T cell receptors (TCRs) containing immunoreceptor tyrosine-based activation motifs (ITAMs) or activating NKG2 family receptors such as NKG2D. This results in suppressed cytotoxic activity of CD8+ T cells. During immune checkpoint inhibition with anti-NKG2A antibodies, inhibitory signals in CD8+ T cells are shut down, allowing stimulation via NKG2D-MICA/B or costimulation via the CD3-TCR complex to induce tumor cell lysis.

(Data source: Creelan BC, et al. Nat Rev Clin Oncol. 2019)

(Data source: Li Y, et al. J Immunother Cancer. 2024)
Targeted therapy of KLRC1
The immunosuppressive effect of NKG2A in the tumor microenvironment suggests that blocking NKG2A may be a potential treatment to improve the immune response of NK cells and T cells. NKG2A blockade may enhance the anti-tumor immunity mediated by NK cells and CD8+ T cells. Currently, many antibody drugs targeting NKG2A are in clinical research.

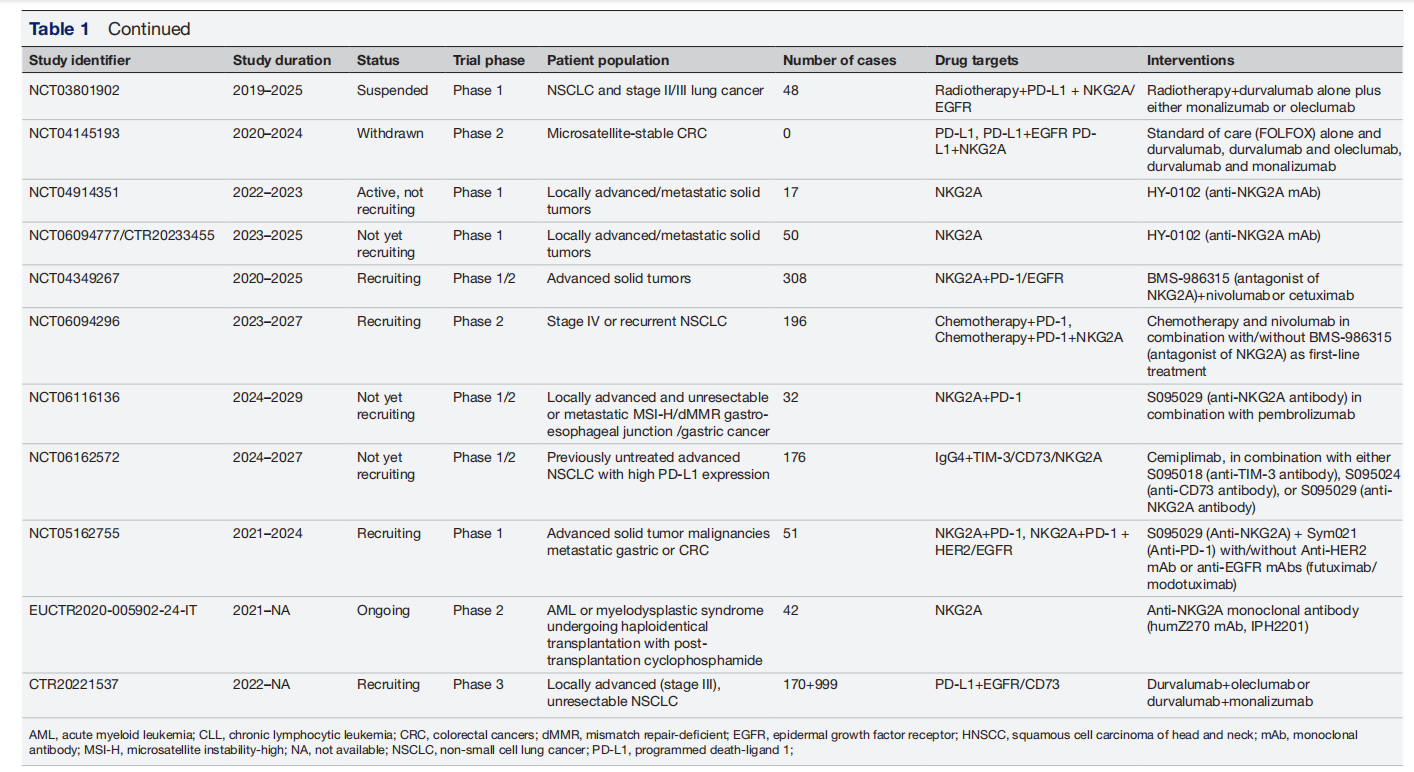
(Data source: Li Y, et al. J Immunother Cancer. 2024)
Monalizumab (IPH2201) is a potential first-in-class immune checkpoint inhibitor being developed by innate-pharma. It targets the NKG2A receptor expressed on tumor-infiltrating cytotoxic CD8+ T cells and NK cells. Monalizumab is a blocking antibody that prevents the suppression of CD8+ T cells and NK cells by HLA-E-expressing tumor cells. By simultaneously targeting both the innate and adaptive immune systems, monalizumab has the potential to re-establish a broad anti-tumor response. It is primarily intended for the treatment of non-small cell lung cancer (NSCLC). Monalizumab is currently under investigation in numerous clinical trials. PACIFIC-9 is a clinical trial evaluating the efficacy of durvalumab (anti-PD-L1) combined with monalizumab (anti-NKG2A) or AstraZeneca's oleclumab (anti-CD73) in patients with unresectable stage III non-small cell lung cancer (NSCLC) who have not progressed after concurrent platinum-based chemoradiotherapy.


(Data source: innate-pharma official website)
