CD59 is a membrane-bound protein that functions as a membrane attack complex inhibitory factor (MACIF). It acts by binding to C8 and C9 complement components of the membrane attack complex (MAC), thereby preventing the binding of multiple copies of C9 required for complete formation of the permeability pore. This inhibitor appears to be species-specific and is involved in signaling between T cell activation and protein tyrosine kinases. CD59 is the only membrane-bound inhibitor of the MAC on human cells and serves as a last line of defense against activation of the terminal complement pathway.
(Data source: Couves EC, et al. Nat Commun. 2023)
CD59 distribution:
CD59 is mainly expressed on the cell membrane and exists in various types of cells, mainly in red blood cells, monocytes, endothelial cells, neurons and other cells, and participates in various physiological and immune processes.
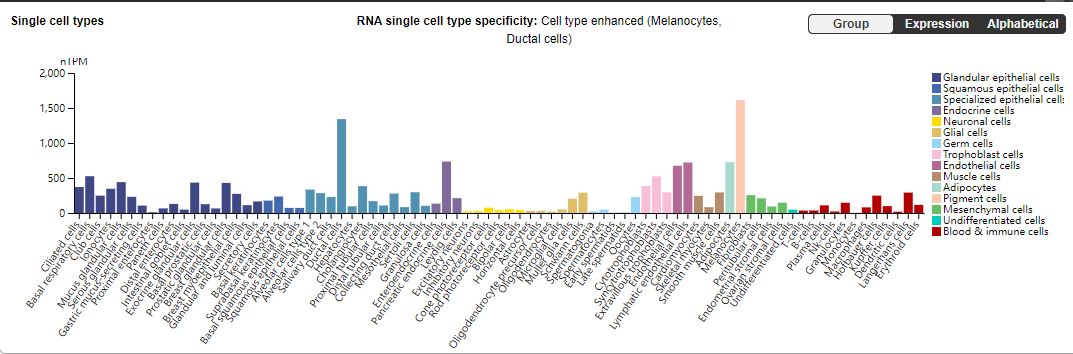
(Data source: the uniprot)
CD59 structure:
CD59 is a glycosylphosphatidylinositol-anchored membrane protein encoded by the gene CD59. CD59 is a relatively flat, disc-shaped molecule with a highly compact spatial structure, consisting of three tandem β-pleated sheets, five protruding loops, and a short α-helix. This unique structure enables CD59 to effectively inhibit complement activation, thereby protecting cells from complement-mediated attack.
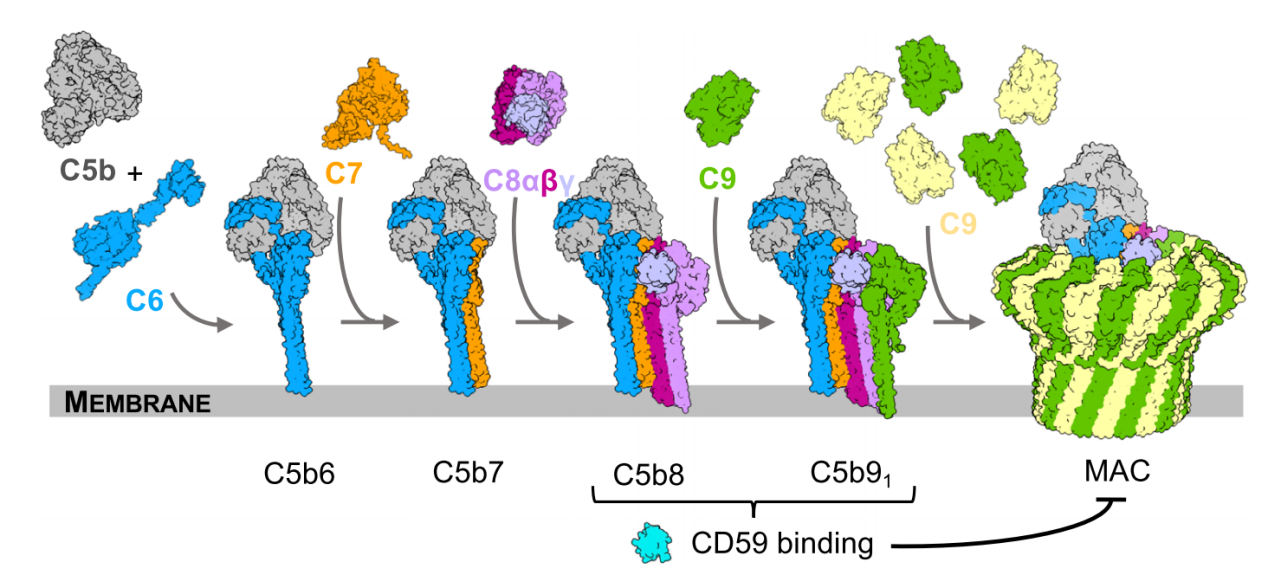
Structural basis for CD59 inhibition of the membrane attack complex: CD59 binds to complement proteins C8 and C9 on the cell membrane, preventing the insertion and polymerization of the membrane attack complex (MAC) pore, thereby inhibiting MAC formation. The structural basis for CD59-C8 inhibition of the membrane attack complex includes CD59 binding to the leading β-strand of TMH2 of C8, forming a continuous antiparallel β-sheet. The C-terminal β-strand of CD59 forms a continuous antiparallel β-sheet with the MACPF structure of C8. The aromatic residues of CD59 fill the gap in the TMH2 structure of C8, affecting the structural bending and membrane insertion position of C8.
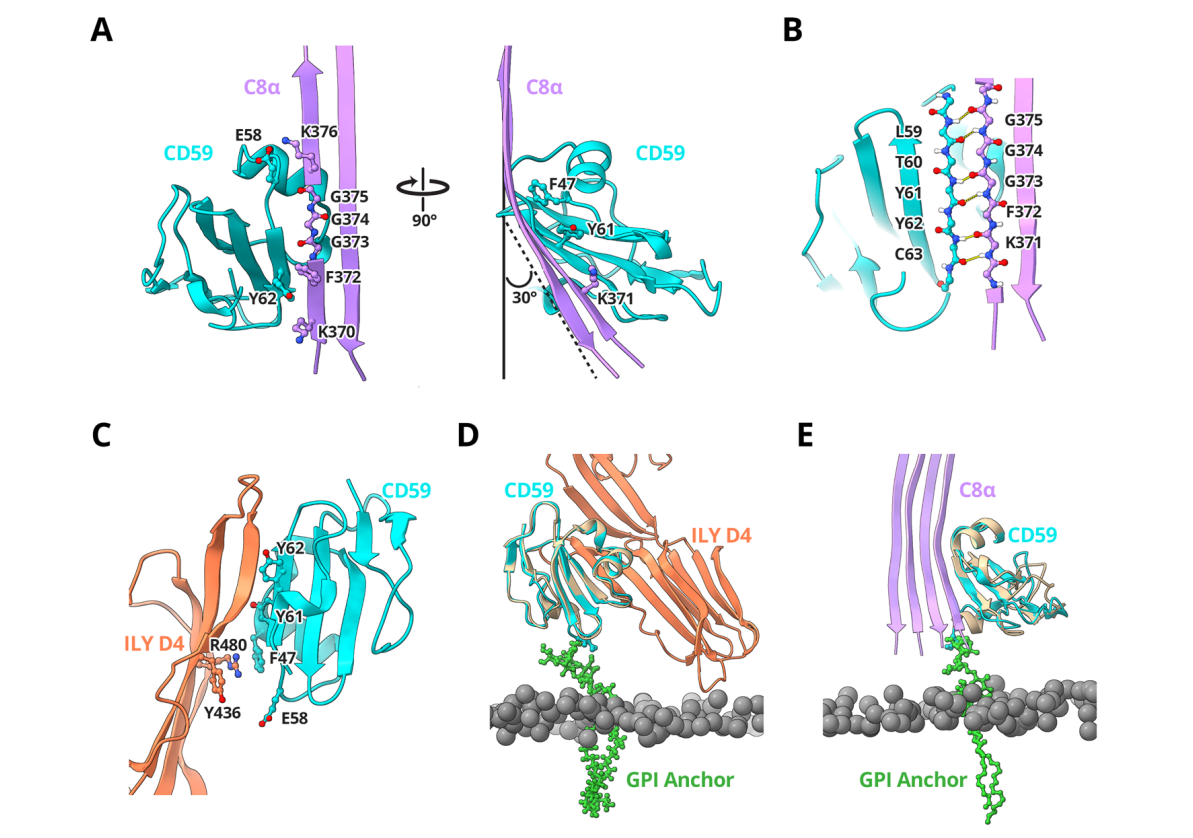
(Data source: Couves EC, et al. Nat Commun. 2023)
When bound to C8, CD59 captures C9's extended β-hairpin, preventing it from crossing the lipid bilayer, thereby preventing membrane insertion and polymerization. The mechanism of CD59 binding to C9 is similar to that of CD59 binding to C8, capturing C9's extended β-hairpin and preventing it from crossing the lipid bilayer. Blocking C9 insertion limits the structural transition of subsequent monomers and indirectly halts MAC polymerization.

(Data source: Couves EC, et al. Nat Commun. 2023)
The relationship between CD59 and disease:
CD59 and paroxysmal nocturnal hemoglobinuria (PNH)
Paroxysmal nocturnal hemoglobinuria (PNH) is a rare hematologic disorder characterized by hemolysis, coagulation, and bone marrow failure. Deficiency of CD55 and CD59 leads to abnormal complement activation in PNH erythrocytes. The classical, lectin, and alternative pathways of complement activation lead to the formation of C3 convertase, which cleaves C3 into C3a and C3b, which in turn leads to the formation of C5 convertase, which cleaves C5 into C5b, thereby activating the terminal complement component and forming the membrane attack complex (MAC). In normal red blood cells (RBCs), the GPI-anchored surface proteins CD55 and CD59 inhibit complement activation by blocking C3 convertase and the membrane attack complex, respectively. In the absence of CD55 and CD59, GPI-deficient PNH RBCs exhibit uncontrolled complement activation and lysis. This suggests that CD59 plays a crucial role in the pathogenesis of PNH and could serve as a valuable diagnostic target for PNH.

(Data source: Colden MA, et al. Front Immunol. 2022)
CD59 and diabetes
CD59 plays multiple roles in the development of diabetes. Studies have shown that CD59 is inactivated in diabetic patients due to glycation caused by sustained high blood sugar, which leads to the activation of intracellular signaling pathways and the release of pro-inflammatory and pro-coagulant cytokines, as well as growth factors, thereby impairing the function of multiple proteins. In addition, downregulation of CD59 expression in pancreatic islets is associated with diabetes, which may affect insulin secretion. Downregulation of CD59 significantly reduces the ability to secrete insulin, thereby affecting the regulation of blood sugar levels. Downregulation of CD59 further reduces the fusion events between insulin granules and cell membranes by affecting the interaction of SNARE protein complexes associated with insulin release, thereby reducing insulin release.

(Data source: Blom AM. J Intern Med. 2017)
CD59 and tumor cells
CD59 is highly expressed in various cancer cell lines and tumor tissues, affecting the function and phenotype of immune cells in the tumor microenvironment. The relationship between tumor cells and immune cells is closely related to CD59. CD59 expression on the surface of tumor cells can protect tumor cells from immune attack by preventing the formation of the membrane attack complex (MAC). Overexpression of CD59 can effectively enhance the resistance of tumor cells to lysis by the complement system, thereby protecting tumor cells from attack by the immune system. In addition, silencing of CD59 can enhance complement-mediated tumor cell apoptosis, thereby inhibiting tumor growth. Immune cells such as NK cells, macrophages, and T cells may regulate their activity through interaction with CD59, affecting the immune escape and growth of tumor cells. Targeting CD59 is a very promising tumor immunotherapy strategy.
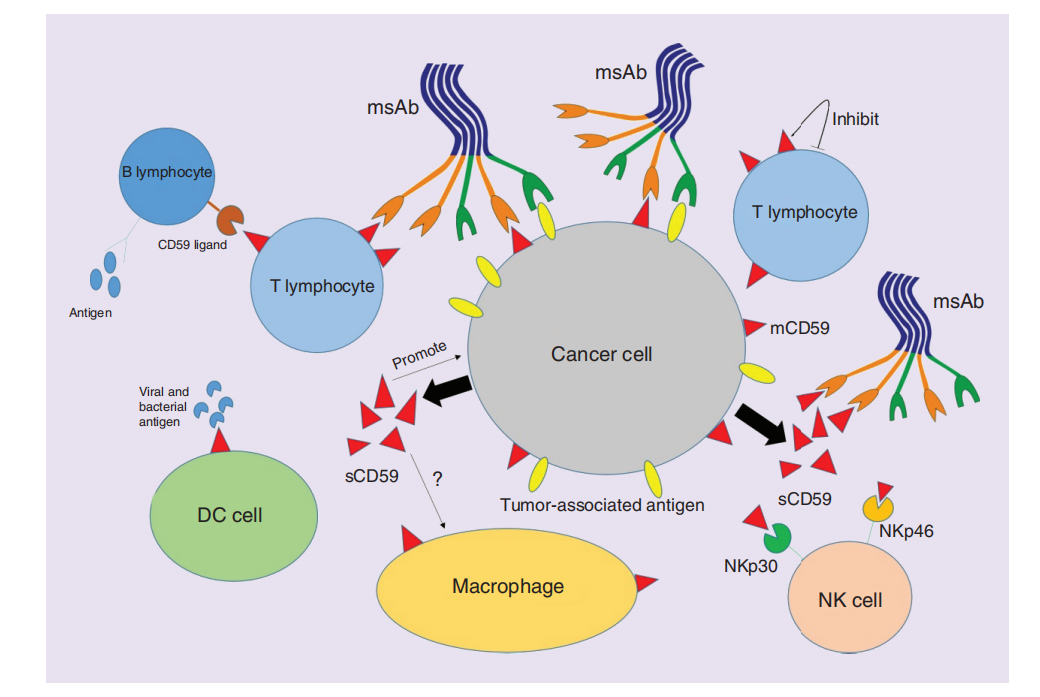
(Data source: Zhang R, et al. Future Oncol. 2018)
