Macrophage colony-stimulating factor 1 (CSF1) is a key cytokine that plays a central role in innate immunity, inflammation, tissue homeostasis (especially bone remodeling), reproduction, and various diseases (such as cancer and autoimmune diseases) by regulating the development, survival, differentiation, and function of monocytes and macrophages.
Expression distribution of CSF1
CSF1 is widely expressed in fibroblasts, epithelial cells (such as renal tubules and endometrium) and immune cells (such as macrophages), and is induced to be highly expressed in inflammation, abnormal bone metabolism and tumor microenvironment.
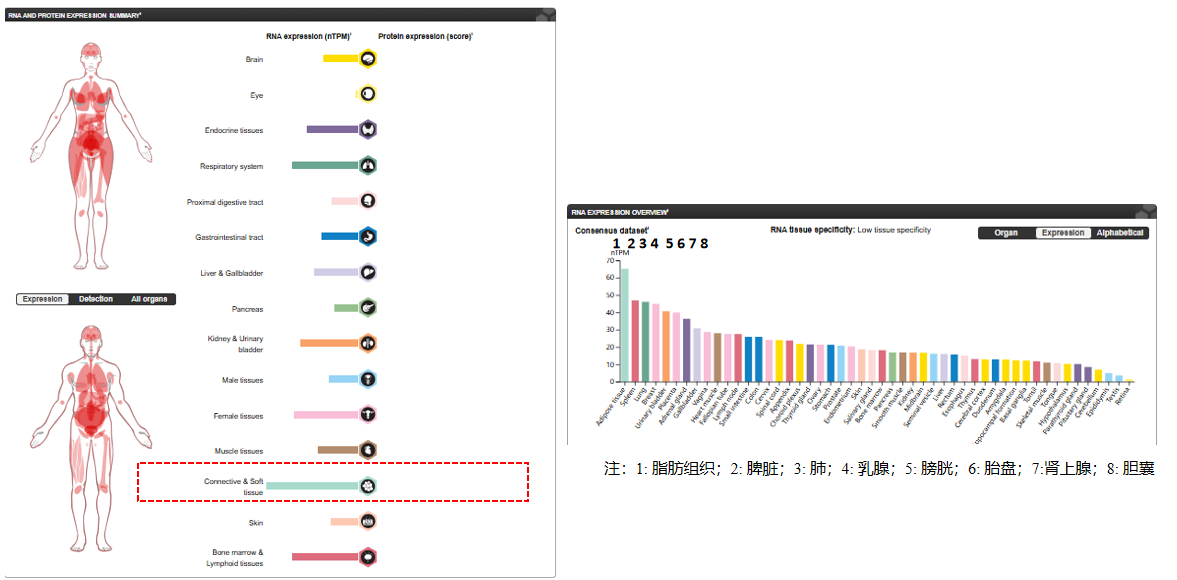
(Data source: Uniprot)
Function of CSF1
This cytokine plays a key role in regulating the survival, proliferation, and differentiation of hematopoietic precursor cells, particularly mononuclear phagocytes such as macrophages and monocytes. It promotes the release of proinflammatory chemokines, thus playing a crucial role in innate immunity and inflammatory processes. Furthermore, it is crucial in regulating osteoclast proliferation and differentiation, bone resorption, and maintaining normal bone development. This cytokine is also required for normal male and female fertility. Furthermore, it promotes the reorganization of the actin cytoskeleton, regulates membrane ruffle formation, cell adhesion, and cell migration, and plays a role in lipoprotein clearance.
Structure of CSF1
CSF1, encoded by the CSF1 gene, is a 554-aa homodimer with a molecular weight of approximately 60.1 kDa, linked by disulfide bonds. Its N-terminal domain is responsible for binding and activating the c-Fms receptor. It plays a central role in innate immunity, inflammation, tissue homeostasis, reproduction, and various diseases (such as cancer and autoimmune diseases) by regulating the development, survival, differentiation, and function of monocytes and macrophages. Its structural diversity (glycosylation and different isoforms) contributes to the complexity of its functional regulation.
Isoform diversity: Through differential splicing and post-translational processing, a variety of isoforms are produced, mainly including secretory glycoprotein (sM-CSF), secretory proteoglycan (spM-CSF) and membrane-bound (mM-CSF or csf).
Disulfide bond: The monomer contains four highly conserved cysteine residues, forming two pairs of intramolecular disulfide bonds; the two monomers form an intermolecular disulfide bond through Cys157 (or nearby positions) to form a homodimer.
Receptor binding domain: Its receptor is c-Fms (encoded by the CSF1R gene), which belongs to the type III receptor tyrosine kinase family. M-CSF dimers bind to the extracellular regions of two c-Fms receptor molecules through their N-terminal domain (comprising the first approximately 150 amino acids), inducing receptor dimerization, autophosphorylation, and activation of downstream signaling pathways (such as PI3K/Akt, MAPK, and JAK/STAT).
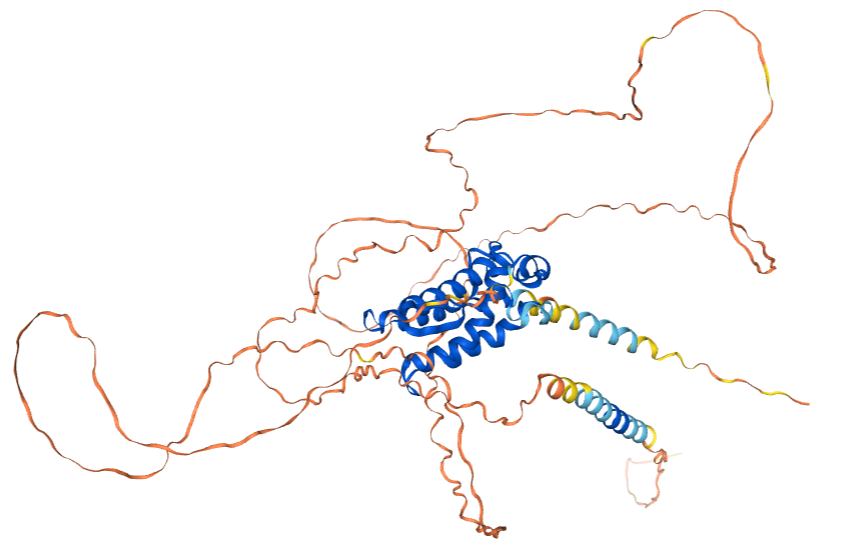
(Data source : AlphaFold)
CSF1 signaling pathway
CSF1 binds to CSF1R (a receptor tyrosine kinase), inducing receptor dimerization and autophosphorylation of tyrosine residues, initiating downstream signaling . 16 IL-34 can also activate CSF1R , but its expression pattern differs from that of CSF1 . Upon phosphorylation, CSF1R recruits adaptor proteins, activating the following major pathways:
PI3K/AKT/ NF-κB pathway: CSF1R phosphorylation activates PI3K, producing PIP3, which in turn activates AKT and mTOR, thereby promoting cell survival, metabolic regulation, and anti-apoptosis (such as macrophage survival).
RAS/RAF/ERK pathway: Activates RAS through the GRB2/SOS complex, cascade-activates RAF-MEK-ERK , and then drives cell proliferation and differentiation (such as monocyte differentiation into macrophages).
JAK/STAT pathway: CSF1R indirectly activates JAK kinase, phosphorylating STAT proteins (such as STAT3/5 ) , thereby regulating the expression of inflammatory genes (such as IL-6 and TNF-α).
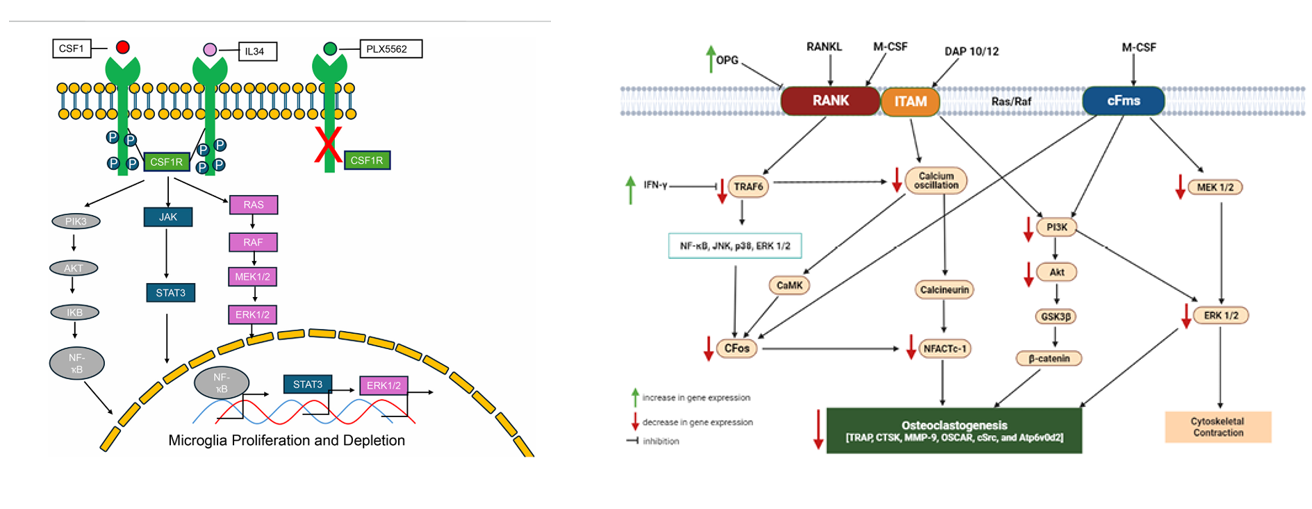
(Data source: Left: Jie Cao, et al. Front Pharmacol. 2025; left: Innocent U Okagu, et al. Int J Mol Sci. 2022)
CSF1 and disease
CSF1 (colony-stimulating factor 1, also known as M-CSF) and its receptor, CSF1R (c-FMS), are key signaling axes regulating macrophage survival, proliferation , differentiation, and function. This target is closely associated with the pathological processes of various diseases, primarily tumors, inflammatory disorders, and neurological diseases .
Tenosynovial Giant Cell Tumors ( TGCT ) : Most TGCT cases ( especially diffuse D -TGCT ) have chromosomal translocations, such as t(1;2)(p13;q35) , which lead to the fusion of the CSF1 gene (located at 1p13 ) and the COL6A3 gene (located at 2q35 ) , causing the CSF1 gene to translocate. The translocation causes the deletion of the 3' untranslated region of CSF1, relieves transcriptional inhibition, and leads to abnormally high expression of CSF1 protein. The CSF1 secreted by tumor cells forms a concentration gradient, recruiting inflammatory cells such as macrophages and osteoclasts that express CSF1R, forming the main body of the tumor.
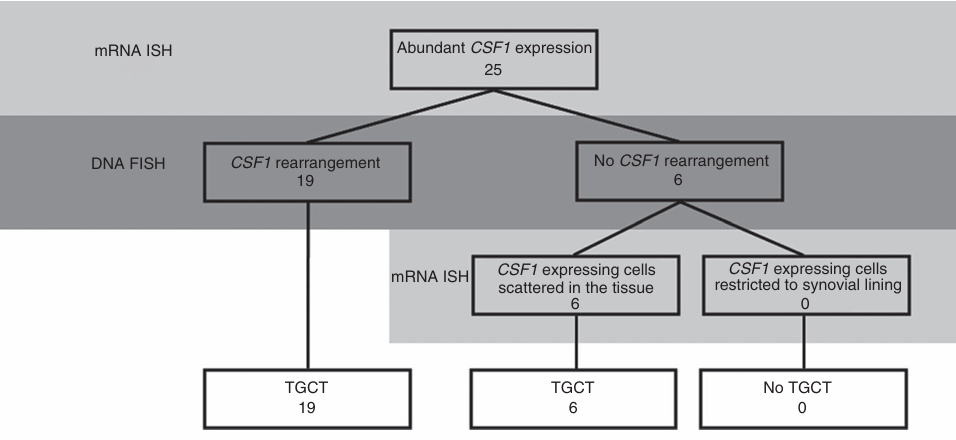
(Data source: Mastboom MJL, et al. Histopathology. 2019)
Thyroid cancer: BRAF mutation induces thyroid cancer cells to secrete CSF-1 , recruit tumor-associated macrophages ( TAMs ), promote tumor angiogenesis and the formation of an immunosuppressive microenvironment.
Breast cancer: CSF-1 activates macrophages in the TMEM doorway (tumor metastasis microenvironment portal), prompting them to secrete VEGF-A , destroying vascular endothelial junctions and promoting the bloodstream metastasis of tumor cells.
Colorectal cancer: After radiotherapy, tumor cells secrete more CSF-1 , inducing TAM polarization to M2 type (immunosuppressive type), upregulating PD-L1 expression, inhibiting CD8⁺ T cell function, and leading to resistance to radiotherapy and chemotherapy.
Targeted therapy for CSF1
Lacnotuzumab , developed by Novartis Pharma AG, is a monoclonal antibody targeting CSF1. Its primary mechanism of action is as a CSF1 inhibitor, modulating immune cell function in the tumor microenvironment by blocking CSF1-mediated signaling pathways. It is used to treat locally advanced melanoma. It was approved for Phase 2 clinical trials in the United States on September 1, 2008.
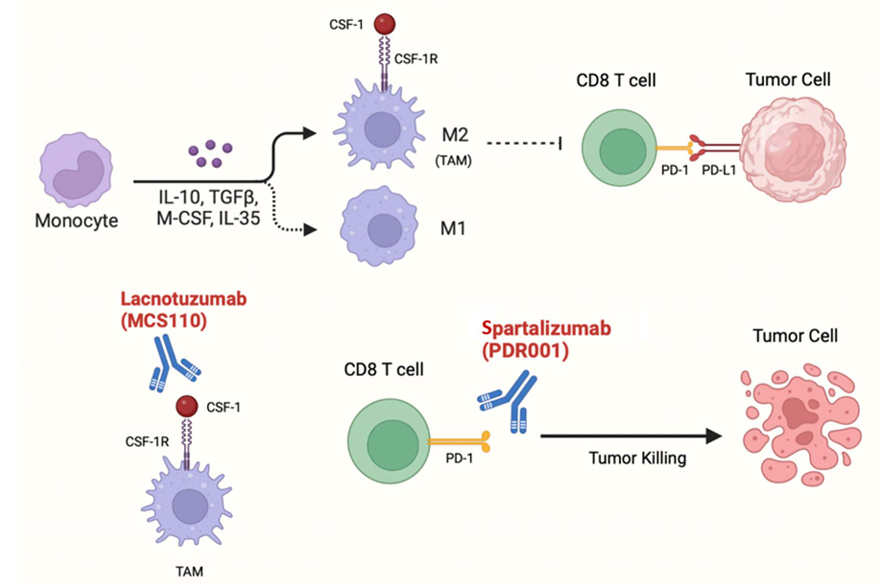
(Data source: Ahmed J , et al. J Immunother Precis Oncol. 2024)
Anti -CD115 humanized mAb is a monoclonal antibody targeting CSF1, developed by Tasly Pharmaceutical Group Co., Ltd. Its primary mechanism of action is as a CSF1 inhibitor, and it is used to treat tumors. It is currently in the preclinical stage.
PD-360324 is a monoclonal antibody developed by Pfizer that targets CSF1. Its primary mechanism of action, similar to lacnotuzumab , is a CSF1 inhibitor, used to treat discoid lupus erythematosus and rheumatoid arthritis. It was approved for Phase 2 clinical trials in the United States on March 15, 2013.
UniTI-101 , a bispecific antibody developed by Marengo Therapeutics , targets CSF1 and CCR2. Its primary mechanism of action is as a CSF1 inhibitor and a CCR2 antagonist, intended for the treatment of tumors. It is currently in the preclinical termination stage.
