Transmembrane serine/threonine kinases TGFBR1 and TGFBR2 are non-pleiotropic receptors for TGF-β cytokines (TGFB1, TGFB2, and TGFB3). They transduce TGFB1, TGFB2, and TGFB3 signals from the cell surface to the cytoplasm, thereby regulating numerous physiological and pathological processes, including cell cycle arrest in epithelial and hematopoietic cells, control of mesenchymal cell proliferation and differentiation, wound healing, extracellular matrix production, immunosuppression, and carcinogenesis.
Expression distribution of TGFBR1 and TGFBR2
TGFBR1 is widely expressed in various tissues and cells, with high expression in glial cells and ductal cells, and low expression in immune cells. TGFBR2 is mainly expressed in endothelial cells and neutrophils, and low expression in various immune cells.


(Data source: uniprot)
Structure and receptors of TGFBR1 and TGFBR2
TGFBR1 and TGFBR2 are both type I transmembrane proteins, containing multiple functional domains from their extracellular to intracellular regions: an N-terminal signal peptide, an extracellular ligand-binding domain, a single transmembrane domain, and a C-terminal intracellular serine/threonine kinase domain. The extracellular regions of both receptors are relatively compact, containing several conserved cysteine residues. These residues stabilize the spatial conformation of the extracellular domains by forming disulfide bonds, which are crucial for the specific recognition of receptors and ligands.
TGFBR2 kinase activity is constitutively active in the basal state, while TGFBR1 kinase activity is entirely dependent on TGFBR2 activation. TGFBR2 phosphorylates specific serine and threonine residues in the GS region of the TGFBR1 kinase domain, thereby relieving autoinhibition and fully activating the kinase function of TGFBR1.
In addition to TGFBR1 and TGFBR2, the TGF-β receptor system also includes TGFBR3 (also known as betaglycan), which is a co-receptor that does not have kinase activity itself, but can enhance the binding efficiency of TGF-β ligands (especially TGF-β2) to signal receptors.

(Data source: Wieteska Ł, et al. Nat Commun. 2025)
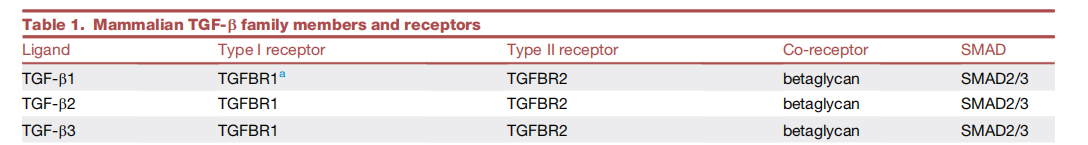
(Data source: Massagué J, et al. Cell. 2023)
Signaling pathways and regulation of TGFBR1 and TGFBR2
A receptor complex formed by the symmetrical binding of two TGFBR1 and two TGFBR2 molecules phosphorylates and activates TGFBR1 via persistently active TGFBR2. Activated TGFBR1 phosphorylates SMAD2, causing it to dissociate from the receptor and bind to SMAD4. Subsequently, the SMAD2-SMAD4 complex translocates to the nucleus, where it regulates the transcription of TGF-β-related genes. This constitutes the classic SMAD-dependent TGF-β signaling cascade. It also participates in non-classical, SMAD-independent TGF-β signaling pathways , such as the MAPK pathway, the PI3K/Akt pathway, and the Rho-like GTPase pathway.

(Data source: Li L, et al. Biomedicines. 2024)
Targeted therapy of TGFBR1 and TGFBR2
Targeted therapies against the TGF-β signaling pathway mainly include inhibiting TGF-β receptor binding and inhibiting TGF-β receptor kinase , such as antibody inhibitors targeting TGFBR2.

(Data source: Massagué J, et al. Cell. 2023)
INCA33890 is an investigational TGFβR2×PD-1 bispecific antibody developed in collaboration with Merus using its Biclonics technology platform. It aims to block TGF-β-mediated signaling in T cells that co-express TGF-β and PD-1. TGF-β is known to promote cancer immune escape and predict poor response to PD-L1 targeted therapy. INCA33890 aims to protect tissues where TGF-β signaling is crucial for normal function, avoiding the known toxicities of broadly blocking the TGF-β pathway. INCA33890 provides a promising targeted therapy strategy for patients with advanced or metastatic solid tumors, including microsatellite stable colorectal cancer. In October 2025, Incyte announced Phase 1 results of its TGFβR2×PD-1 bispecific antibody for advanced colorectal cancer and its KRAS G12D inhibitor for advanced pancreatic ductal adenocarcinoma , demonstrating a favorable safety profile and evidence of clinical benefit in patients with previously treated pancreatic ductal adenocarcinoma (PDAC).

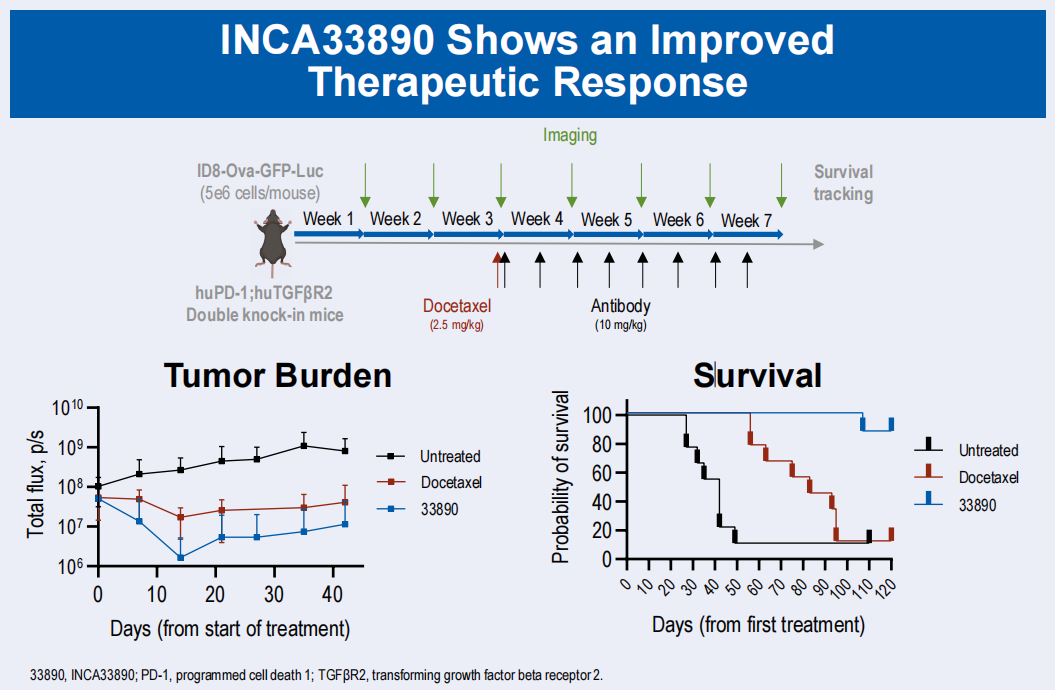
(Data source: incytemi official website)
Bintrafusp alfa is an antibody targeting PD-L1 and TGFBR2. It is a first-in-class bifunctional fusion protein composed of the extracellular domain of the TGF-βRII, which fuses with a human IgG1 antibody that blocks PD-L1, acting as a TGF-β “trap.” By co-localizing and simultaneously inhibiting these pathways, bintrafusp alfa has the potential to enhance immune cell invasion of tumors, limit metastasis, and improve the response to anticancer therapies. Bintrafusp alfa inhibits TGF-β activity through matrix modulation, possessing the potential to suppress angiogenesis and potentially restore normal vascular homeostasis, thereby enhancing drug delivery and T cell infiltration into the tumor microenvironment (TME).

(Data source: Li L, et al. Front Oncol. 2022)
