Tenascin (TNC) is a cytosmal matrix-rich glycoprotein. Extracellular matrix proteins participate in guiding neuronal and axonal migration during development and are also associated with synaptic plasticity and neuronal regeneration. It promotes neurite growth in cortical neurons growing on an astrocyte monolayer. This protein is a ligand for integrins α-8/β-1, α-9/β-1, α-V/β-3, and α-V/β-6. In tumors, it stimulates angiogenesis by promoting endothelial cell elongation, migration, and budding. TNC serves as an adjunct biomarker for the diagnosis and prognosis of cardiovascular disease patients and plays a crucial role in atherosclerosis.
TNC expression distribution
TNCs are mainly expressed in smooth muscle cells, parietal cells, secretory cells, glial cells, basal cells, and supporting cells of secretory glands.

(Data source: uniprot)
The structure and receptors of TNC
TNC is a secreted protein composed of 2201 amino acids, which, from the N-terminus to the C-terminus, contain: a tendinogenic assembly domain (TA), an epidermal growth factor-like (EGF-L) repeat sequence, a fibronectin type III (FNIII) domain, and a fibronectin globulin (FG) domain. The FNIII domain consists of 8 fixed FNIII domains and 9 variable FNIII domains.
TNCs possess a variety of biological functions through binding to and interacting with multiple genes. Heat shock protein 33 (HSP33) is responsible for the accumulation of intracellular TNCs. The EGF-like repeat domain of TNCs regulates cell adhesion and cell movement, serving as an anti-adhesion region for fibroblasts, neurons, and glial cells. This domain also regulates neuronal migration and axonal pathways during development. Due to the alternative splicing of fibronectin type III (FNIII)-like repeat sequences, TNCs have many isoforms with different functions and sizes.
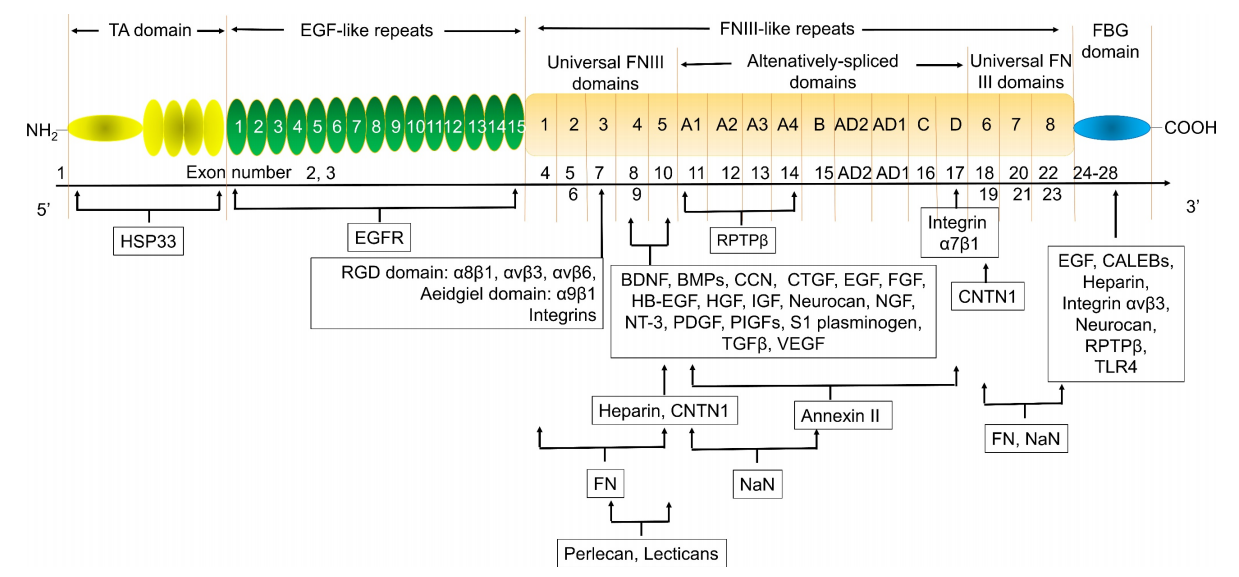
(Data source: Chen W, et al. Int J Biol Sci. 2024)
(Data source: Alphafold)
The role of TNC in vascular regulation
TNCs interact with various cell surface receptors and extracellular matrix components. They transmit signals to intracellular effectors (such as MAPK) via integrins, TLR4, and Wnt receptors, leading to changes in gene transcription and consequently altering the expression of proteins associated with proliferation, adhesion, and cell survival/apoptosis responses.
TN-C exhibits moderate downregulation of genes related to nuclear division, cell division, and cell migration in lymphatic endothelial cells, suggesting an inhibitory effect on these cellular processes. TN-C is an important inducer of the neuroinflammatory cascade and the resulting stroke pathology. Following subarachnoid hemorrhage, its expression is upregulated in cerebral arteries and brain tissue, including astrocytes, neurons, and cerebral capillary endothelial cells. TN-C plays a crucial role in regulating angiogenesis, endothelial cell function, and development.
Different domains or isoforms of TNC may exert different, or even opposite, effects, either promoting or resisting atherosclerosis.
Many TNC domains/subtypes promote atherosclerosis, such as the A1 domain, A2 domain, EGF-like domain, FBG domain, and TnfnIII 1-5 domain. TNCs stimulate not only the expression of pro-inflammatory cytokines, such as IL-6, IL-8, and TNFα, but also the expression of anti-inflammatory cytokines, such as IL-4 and IL-13, indicating that TNCs have a dual pro-inflammatory and anti-inflammatory effect. TNCs derived from bone marrow and systemically possess anti-atherosclerotic activity, while TNCs derived from macrophages, platelets, and smooth muscle cells promote atherosclerosis.

(Data source: Aithabathula RV, et al. Antioxidants (Basel). 2025)
Targeted therapy for TNC
There are many monoclonal antibodies targeting the TNC domain in preclinical and clinical studies. TNC (especially its C-terminal domain TNC-C) is highly enriched in active plaque areas but hardly expressed in normal tissues, which provides a direction for achieving precise targeted drug delivery.
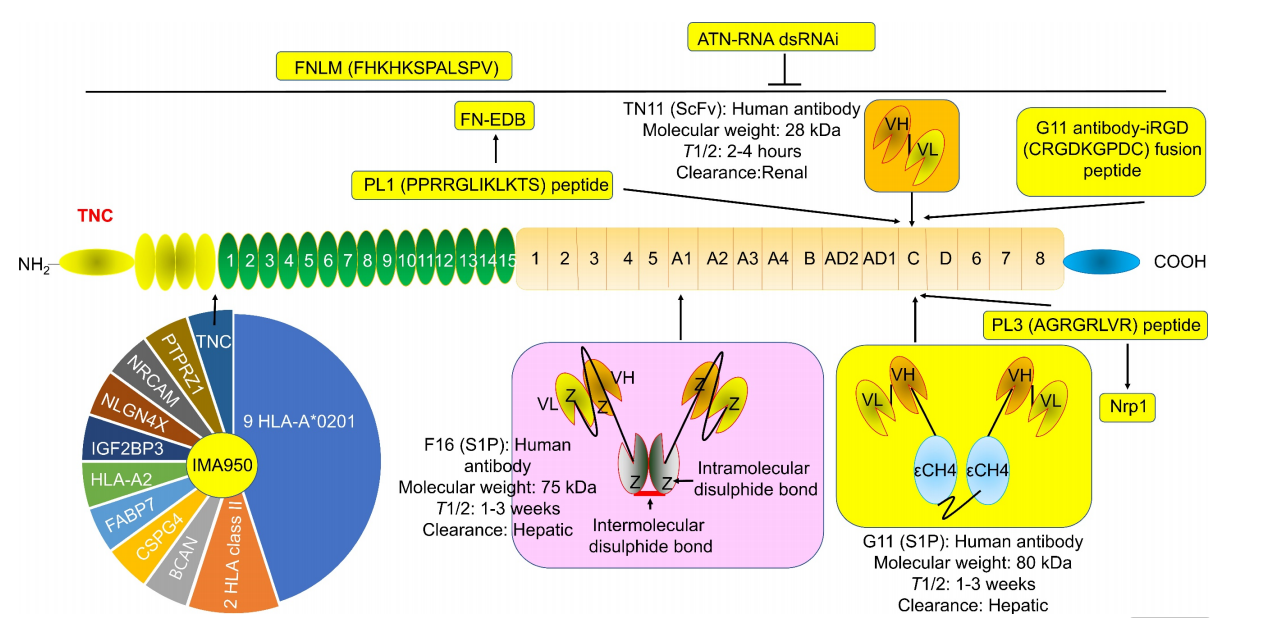
(Data source: Chen W, et al. Int J Biol Sci. 2024)
Tenatumomab (ST2146) is a monoclonal antibody targeting tumor nuclei (TNCs), but the amount of drug reaching tumor lesions was extremely low, leading to the termination of the clinical trial. The results showed significant inter-individual variability in response, potentially including the production of anti-drug antibodies (ADAs). Subsequently, by linking three chelating agents to ST2146, an improved version of ST2146 was generated. ST2146-DOTA (ST8198AA1) showed the best efficacy, with an immune response rate exceeding 50%. Radiolabeling with 111I was then used for pre-treatment imaging and dosimetry, achieving a radiolabeling efficiency of 77.0%, but no information regarding its anti-tumor efficacy was provided.
TNC-targeted therapy can also be combined with anti-inflammatory, lipid-lowering, or repair-promoting drugs (such as statins and IL-2). Most recombinant fully humanized single-chain scFv antibodies targeting TNC, “F16”and “P12”(recognizing TNC's FNIII-A1 and FNIII-D, respectively), have been engineered to significantly enhance their binding affinity to TNC. When fused with IL2 to form the immune cytokine F16-IL2, it was found to accumulate in a breast MDA-MB231 tumor transplantation model. F16-IL2 may exert its anti-tumor effect by enhancing the recruitment and infiltration of immune cells, particularly natural killer (NK) cells, suggesting that TNC is an ideal tumor delivery target for IL2.


(Data source: Dhaouadi S, et al. Matrix Biol. 2024)
