The B cell receptor (BCR) CD22 is the most highly expressed sialic acid-binding immunoglobulin-like lectin (SIGLEC) on B cells. Also known as Siglec-2, it plays a role in various aspects of B cell biology, including differentiation, antigen presentation, and migration within the bone marrow. It acts as an inhibitory coreceptor on the B cell surface, inhibiting B cell receptor-induced signaling. Mechanistically, the immunoreceptor's tyrosine-based inhibitory motif domain is phosphorylated by the Src kinase LYN, which in turn leads to the recruitment of protein tyrosine phosphatase 1/PTPN6, resulting in negative regulation of BCR signaling.
Expression distribution of CD22
CD22 is primarily expressed in B cells, with smaller amounts also found in oligodendrocytes, granulocytes, granulosa cells, ovarian stromal cells, and secretory cells. It is also expressed in most B-cell malignancies, including acute lymphoblastic leukemia (B-ALL), non-Hodgkin lymphoma (NHL), and hairy cell leukemia (HCL).
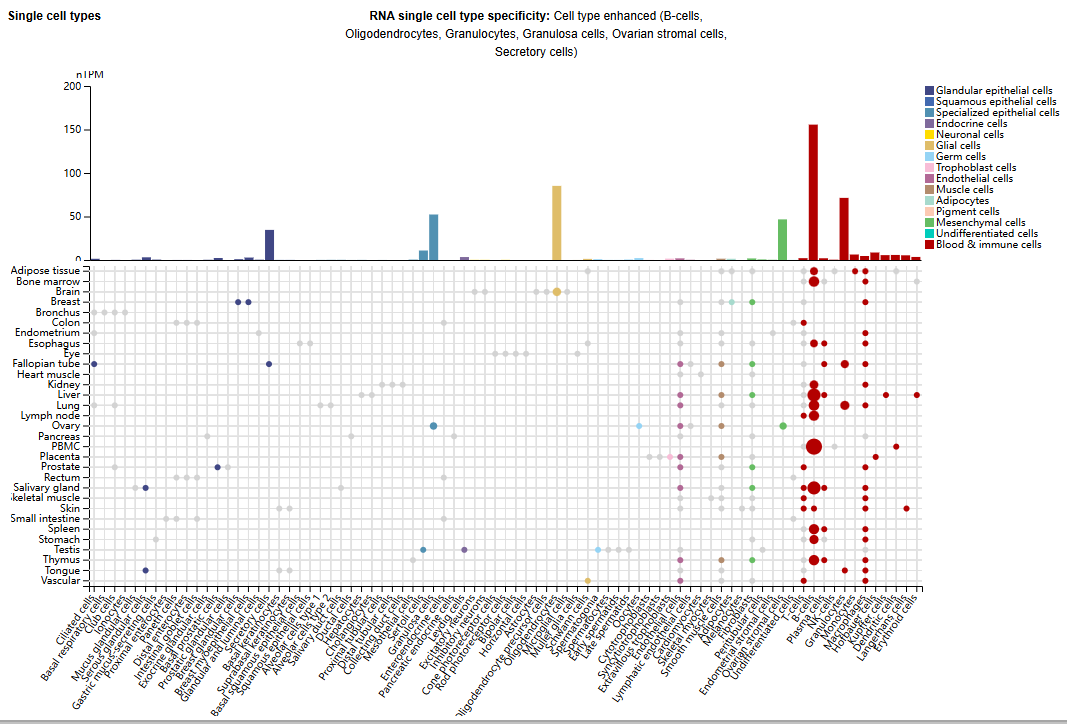
(Data source: Uniprot)
The structure of CD22
CD22 is a transmembrane protein belonging to the siglec family and the immunoglobulin superfamily. It consists of seven extracellular immunoglobulin G-like domains (six C- type and one V- type immunoglobulin domain) and a 141-amino acid cytoplasmic tail. The intracellular domain contains an immunoreceptor tyrosine-based inhibitory motif (ITIM). Upon tyrosine phosphorylation, CD22 recruits phosphatases such as SHP-1, SHP-2, and SHIP-1. CD22 can bind in a " cis " configuration to α - 2,6-linked sialic acid residues on B cell surface molecules (such as CD22 itself, CD45, and IgM). It can also bind in a " trans " configuration to ligands on other cells as an adhesion molecule.

(Data source: Ereño-Orbea J, et al. Nat Commun. 2017)
CD22 signaling pathway and regulation
Trans-interactions: CD22 binds to other cell surface sialylated ligands, regulating B cell migration and BCR signaling thresholds. When CD22 cross-links with the BCR, its intracellular immunoreceptor tyrosine inhibitory motif (ITIM) becomes phosphorylated, recruiting the tyrosine phosphatase SHP-1, leading to dephosphorylation of downstream signaling proteins and ultimately inhibiting B cell activation.
Cis interaction: After binding to its own sialic acid glycans, CD22 cannot bind to trans ligands (such as ligands on APC cells), weakening its inhibitory ability.
The ligand-binding region of CD22 is associated with the initiation of BCR inhibitory signals, and the binding state of its α2,6-sialylated ligand can regulate the inhibitory function of CD22: when α2,6-sialyltransferase expression is downregulated or sialidase expression is upregulated, the shielding effect of CD22 is released, thereby enhancing BCR signal transduction.

(Data source: Lanza F, et al. Cancers. 2020)
CD22 -targeted therapy
Currently, drugs targeting CD22 mainly fall into three categories: monoclonal antibodies, antibody conjugates (ADCs), and CAR-T therapies. Many of these drugs have been approved or are in clinical development.
Monoclonal antibodies
Suciraslimab is the world's first anti-CD22 monoclonal antibody developed by SinoMab BioScience Limited. for the treatment of rheumatoid arthritis (RA) and other immune and neuroimmune diseases. Suciraslimab utilizes a novel mechanism of action, different from currently available therapies, with a dual mechanism of action that simultaneously inhibits Aβ-induced inflammation and promotes Aβ clearance.
On April 26, 2023, the company announced that suciralimab met its primary endpoint in a Phase III clinical trial conducted in China. This multicenter, randomized, double-blind, placebo-controlled study was designed to verify its efficacy and safety in patients with moderately to severely active RA who were refractory to methotrexate (MTX). The Phase III extension study, which enrolled 93 patients, was completed in December 2024, with results expected in the third quarter of 2025. The application was accepted by the National Medical Products Administration (NMPA) in September 2023, and clinical and GMP inspections have been completed (January 2024). Approval is expected within 2025, becoming the company's first commercialized product. Following its successful launch in RA, the company will further advance the clinical development of suciralimab in SLE, MCI due to Alzheimer's disease , and Alzheimer's disease itself, expanding its therapeutic area to address additional unmet clinical needs.
Another drug candidate from SinoMab BioScience Limited, SM06, is a second-generation humanized anti-CD22 antibody derived from suciraslimab, with a similar mechanism of action. Internal in vitro studies have demonstrated that SM06 has the potential to enhance immunomodulatory efficacy. This compound is in the IND-enabling stage and is currently undergoing clinical development optimization.
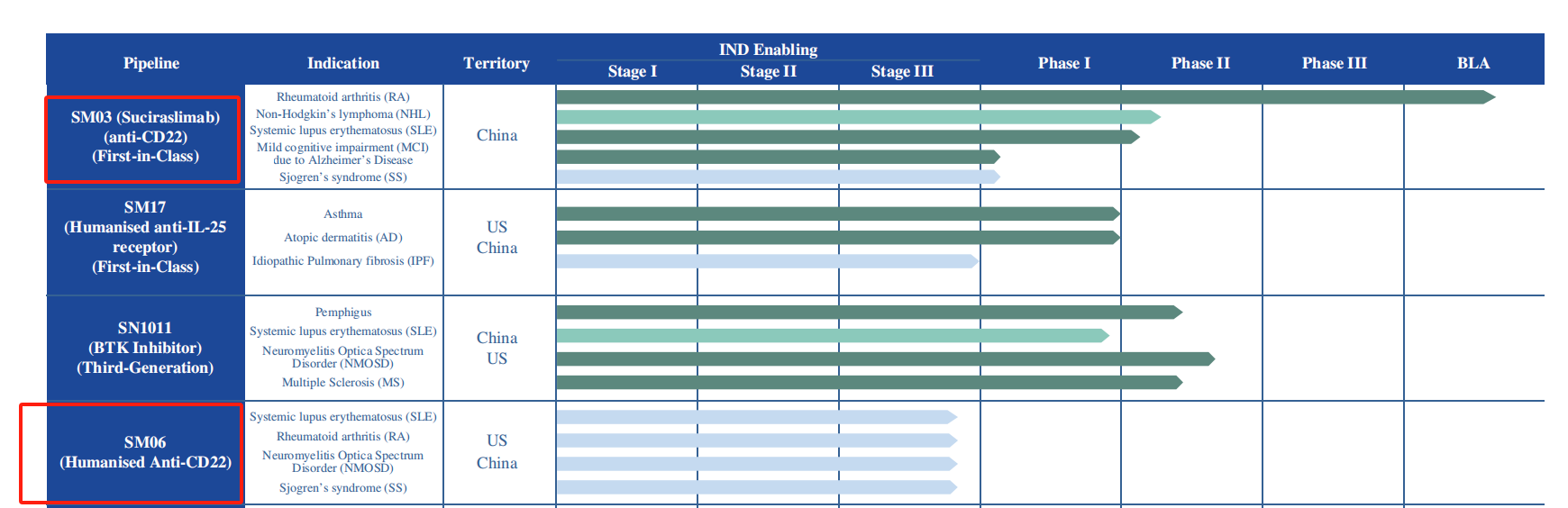
(Data source: SinoMab BioScience official website)
Antibody-drug conjugates (ADCs)
As an endocytic receptor, ligation of CD22 with its ligand triggers rapid endocytosis, enabling the application of CD22 ADCs . Inotuzumab Ozogamicin (INO), a CD22-targeting antibody-drug conjugate, was developed by Celltech (a UK biotechnology company) and Wyeth (a pharmaceutical company subsequently acquired by Pfizer in 2009). It was approved for marketing in the US in 2017 and in China in 2021. It comprises the humanized anti-CD22 monoclonal IgG4 antibody G544 and the chemically linked DNA-damaging payload Calich-DMH. It recognizes the IgG-like domain 1 of CD22 and exerts potent cytotoxic effects on tumor cells, resulting in significant tumor mass regression in two lymphoma xenograft models.

(Data source: Rubinstein JD, et al. Front Immunol. 2023)

(Data source: Short NJ, et al. J Hematol Oncol. 2024)
Pinatuzumab vedotin (targeting CD22-vc-MMAE) was developed by Roche and its development was terminated in a Phase II study for relapsed or refractory diffuse large B-cell lymphoma (DLBCL) because its efficacy was inferior to that of polatuzumab vedotin (targeting CD79b) with the same target.
Moxetumomab pasudotox is a CD22-targeting ADC developed by AstraZeneca . It consists of the binding portion of an anti-CD22 antibody fused to a toxin. Upon binding to CD22, the molecule is internalized, processed, and releases its modified protein toxin, inhibiting protein translation and leading to apoptotic cell death. Moxetumomab pasudotox was granted orphan drug designation by the FDA in 2018 for the treatment of hairy cell leukemia ( HCL ).
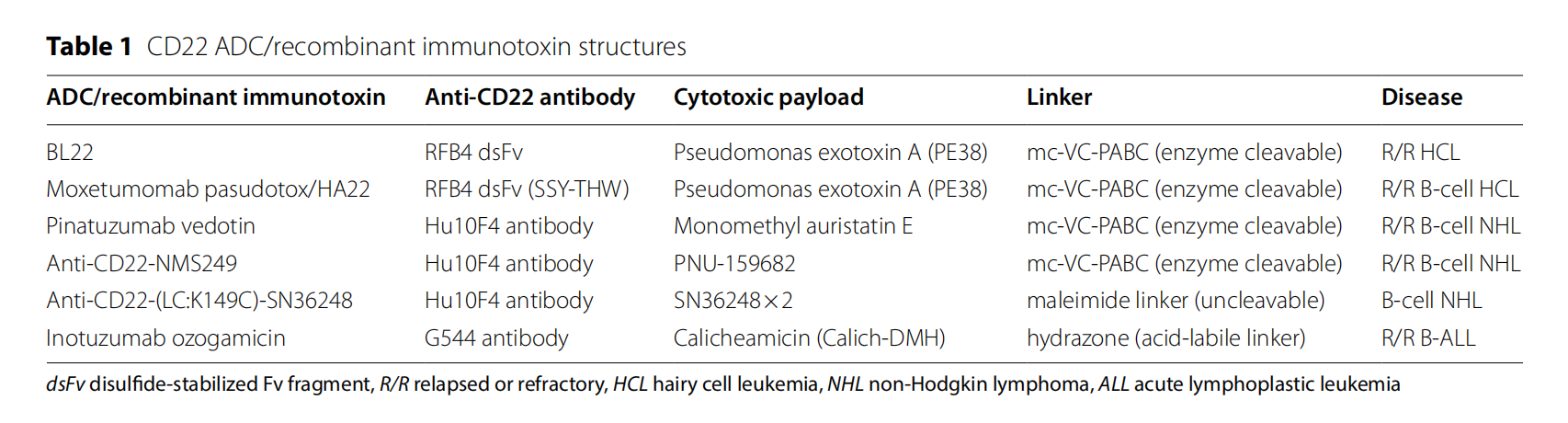
CAR-T therapy
Unlike ADCs, CARs consist of an extracellular antigen-recognizing single-chain variable fragment (scFv), hinge and transmembrane domains, and an intracellular signaling domain. Research into CD22 CAR-T cell therapy began with scFvs derived from two recombinant immunotoxins, HA22 and BL22. Second-generation CAR-T cells incorporating the m971 scFv exhibited superior anti-tumor activity compared to cells harboring scFvs derived from BL22 or HA22. The linker between the heavy and light chains also influences the targeting ability of CD22 CAR-T cells. The short linker promotes immune synapse formation and spontaneous aggregation of CARs without the need for antigen stimulation, thereby inducing tonic activation and improving CAR-T cell function.


The core advantage of CD19/CD22 dual-target CAR-T cell therapy is that it significantly reduces the recurrence rate of tumors due to antigen escape and re-induces remission in patients who have failed previous single-target treatment. At the same time, the overall efficacy and safety are better than single-target strategies.
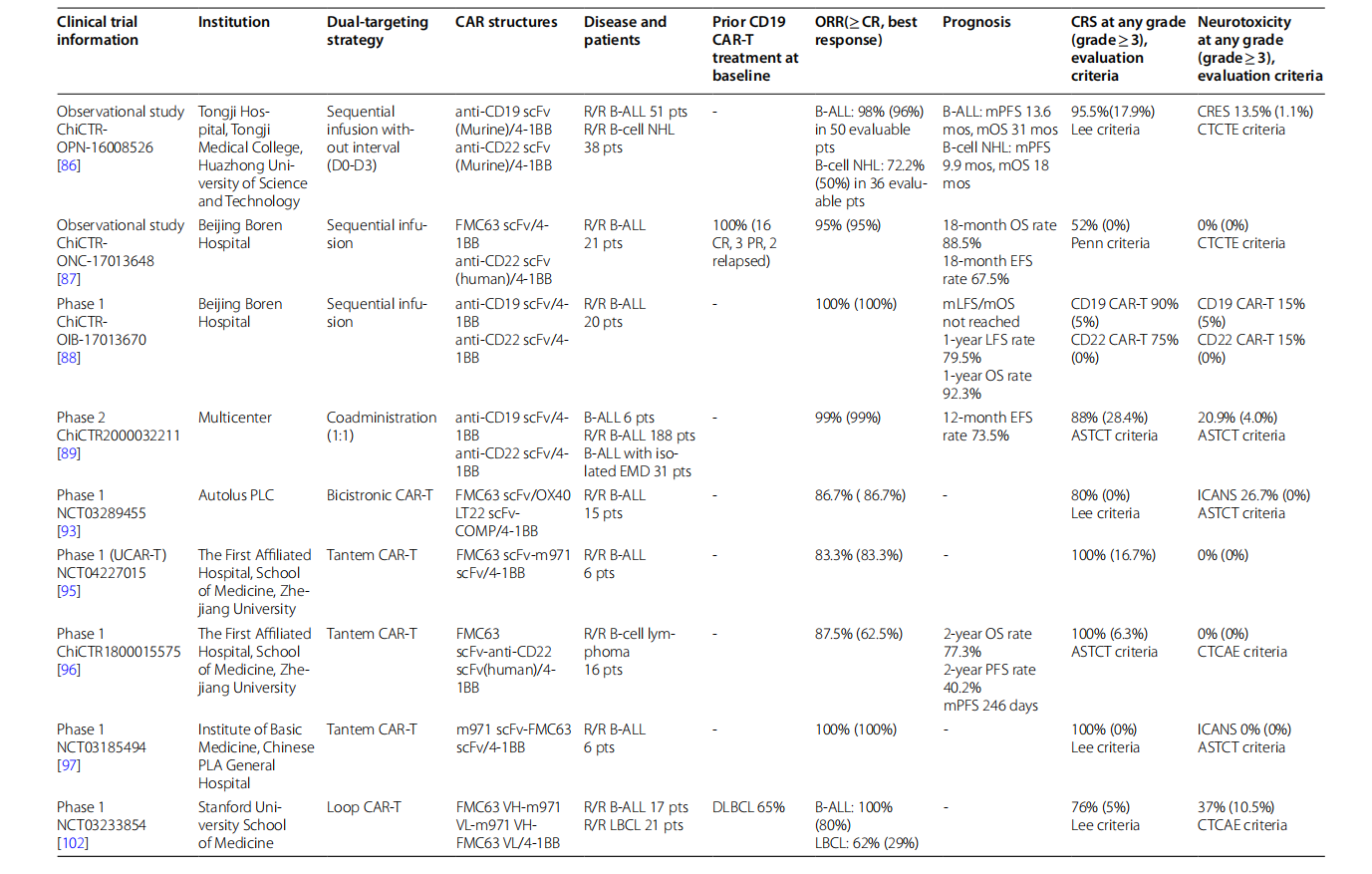
(Data source: Xu J, et al. Exp Hematol Oncol. 2023)
