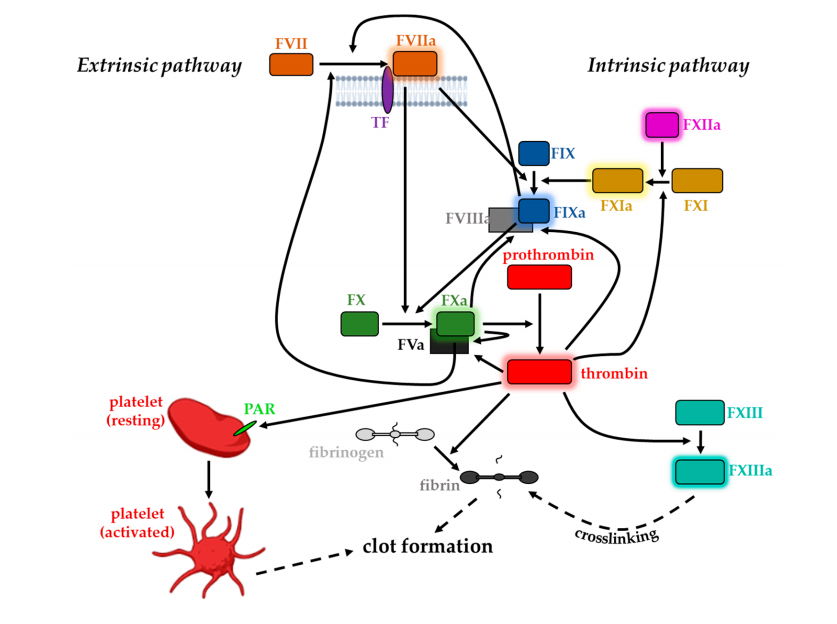
Tissue factor (CD142), also known as coagulation factor III, F3, or CD142, initiates the blood clotting process by forming a complex with circulating factor VII or VIIa. The CD142/VIIa complex activates factor IX or X through specific limited proteolytic activity. CD142 plays a role in normal hemostasis, initiating the formation and propagation of the prothrombinase cascade on the cell surface. CD142 is the promoter of the extrinsic coagulation pathway, participating in angiogenesis, cell adhesion, motility, and cell survival; it is also involved in tumor growth, angiogenesis, and metastasis.
(Data source: Hassan N, et al. Cancers (Basel). 2023)
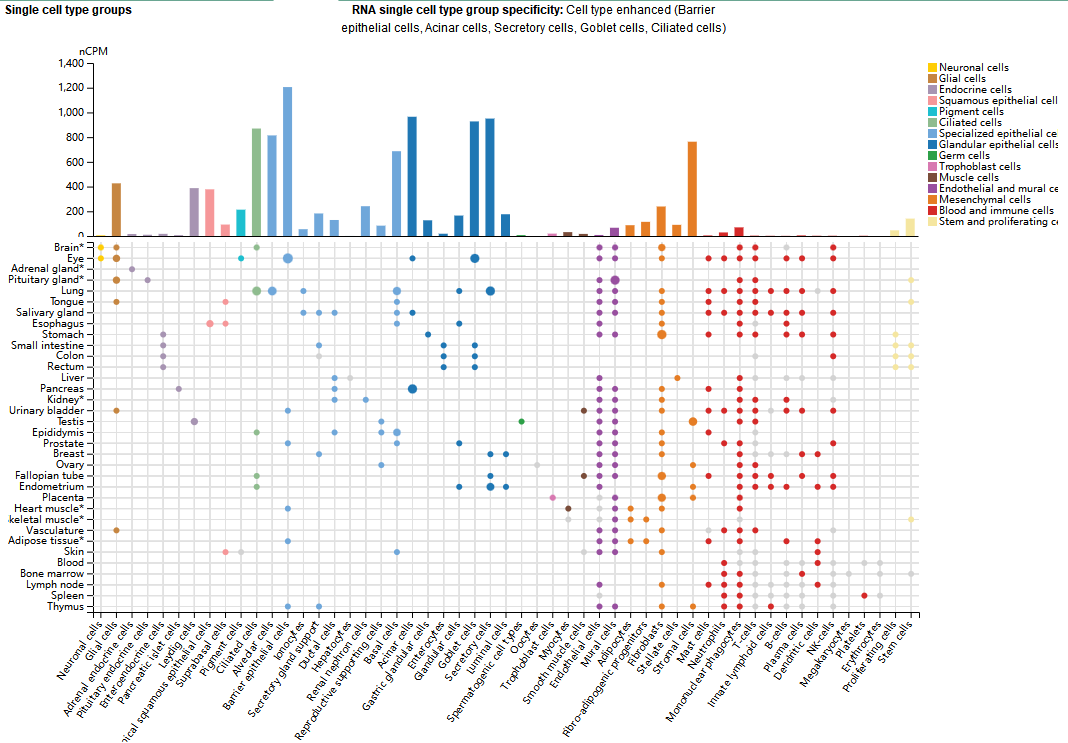
CD142 expression distribution
CD142 expression varies across tissues; it is most highly expressed in the brain, lungs, skin epithelial cells, heart, and testes. At the cellular level, it is primarily expressed in epithelial cells, acinar cells, secretory cells, goblet cells, and ciliated cells. CD142 is also expressed in various solid tumors and hematologic malignancies, regulating tumor development.

(Data source: uniprot)
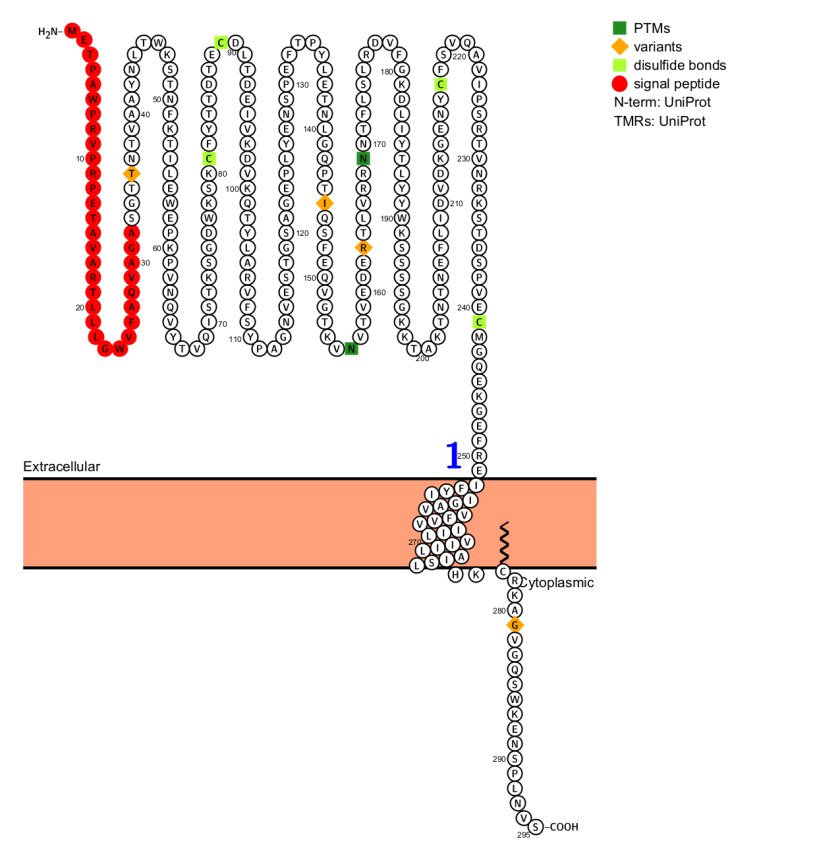
Structure and composition of CD142
CD142 is a transmembrane glycoprotein receptor for coagulation factors VIIa and X, with a molecular weight of 47 kDa and 295 amino acids. It consists of an extracellular domain (1-219 aa), a transmembrane domain (220-242 aa), and an intracellular C-terminal domain (243-263 aa). The intracellular region of CD142 contains two potential phosphorylation sites, indicating that this protein is involved in intracellular activities. When the extracellular domain of CD142 binds to factor VIIa and acts as its catalytic cofactor, the plasma coagulation protease cascade begins, producing thrombin and forming fibrin. Thromboplasm factor VIIa attaches to the extracellular domain of CD142 with subnanomolar affinity, allosterically activating the protease and efficiently cleaving protein substrates, primarily factor X. CD142 shares structural similarities with cytokine receptor family II (especially the IFN-γreceptor), with the similar region being the intracellular C-terminal domain.
(Data source: protest)
CD142 exists in three forms: exon 1A tissue factor (TF-A), asTF, and flTF. asTF lacks a transmembrane domain and is therefore secreted only. Although asTF is not particularly procoagulant, it is involved in non-hemostatic functions. Unlike flTF, asTF binds to integrins without the aid of FVII. The TF-A variant is a novel transcript of the TF gene, generated through alternative splicing of the first intron.

(Data source: Ahmadi SE, et al. Biomark Res. 2023)
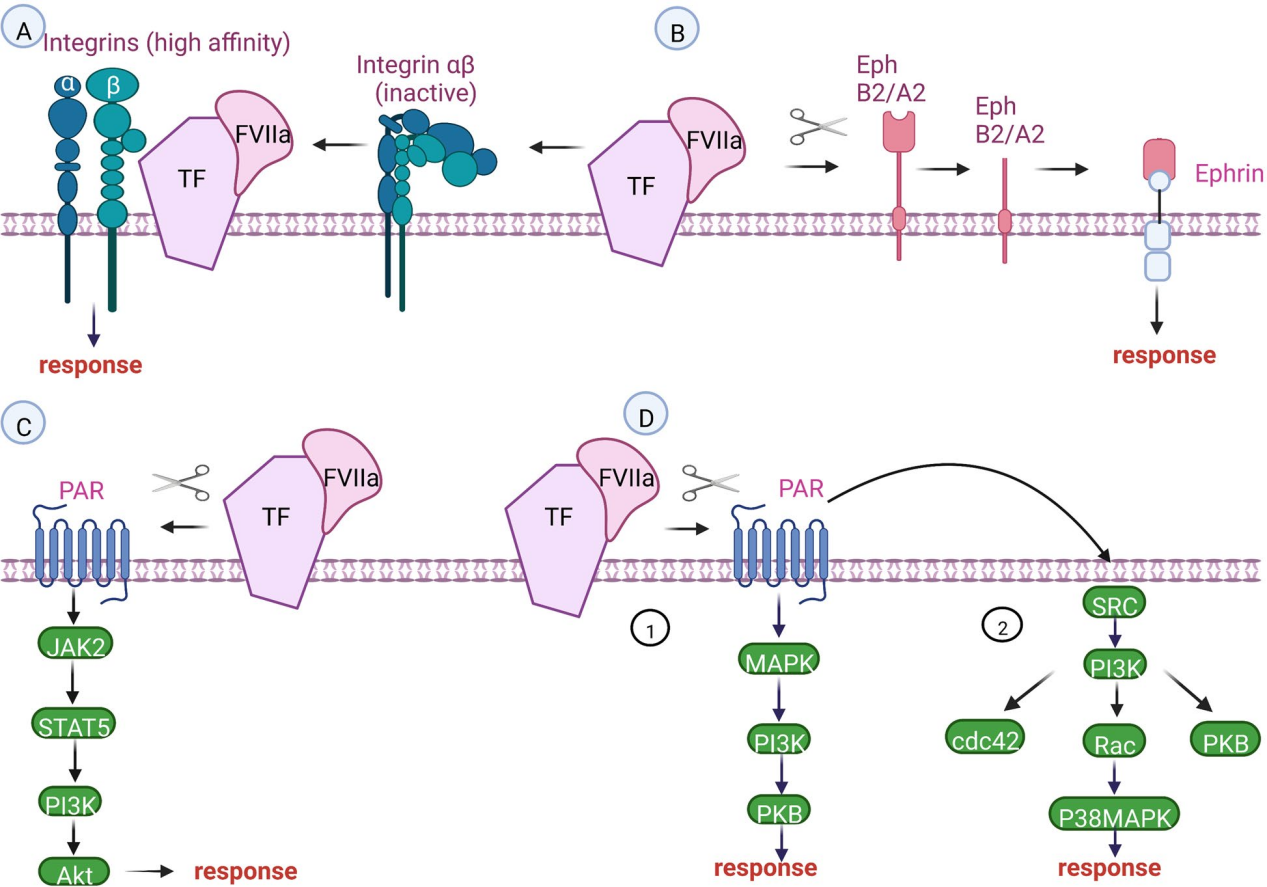
The role of CD142 in the signal path
The CD142 /FVIIa complex and the CD142 /FVIIa/FXa complex mediate CD142 signaling. Signaling can be dependent on or independent of PAR2 and PAR1 by activating integrins and transactivating or proteastomically cleaving certain members of the receptor tyrosine kinase (RTK) family.
CD142 in the MAPK, JAK, Src, PI3K and Rac signaling pathways: CD142 binds to coagulation factor VIIa (FVIIa) to form the CD142-FVIIa complex. This complex activates downstream signaling pathways through multiple mechanisms, including protease-activated receptors (PARs): the PI3K/AKT pathway, the MAPK pathway, the JAK/STAT pathway, and the SRC/P13K/Rac pathway.
PARs-dependent signaling pathways: The CD142-FVIIa complex activates protease-activated receptors (PARs), especially PAR1 and PAR2.
PAR1 is primarily activated by thrombin, promoting tumor growth, angiogenesis, and metastasis. PAR2 is activated by the CD142-FVIIa-Xa complex, promoting the expression of angiogenic factors (such as VEGF and IL-8) and enhancing tumor invasiveness.
Integrin signaling pathway: Integrins promote CD142-dependent PAR2 activation and signal transduction. Independent of PAR-2, direct binding of CD142 to integrins also affects processes such as cell migration and signal transduction. CD142-dependent integrin signaling is involved in angiogenesis, and CD142-PAR2 signaling leads to the production of VEGF and pro-angiogenic molecules, including IL-8 and CXCL-1.
(Data source: Ahmadi SE, et al. Biomark Res. 2023)
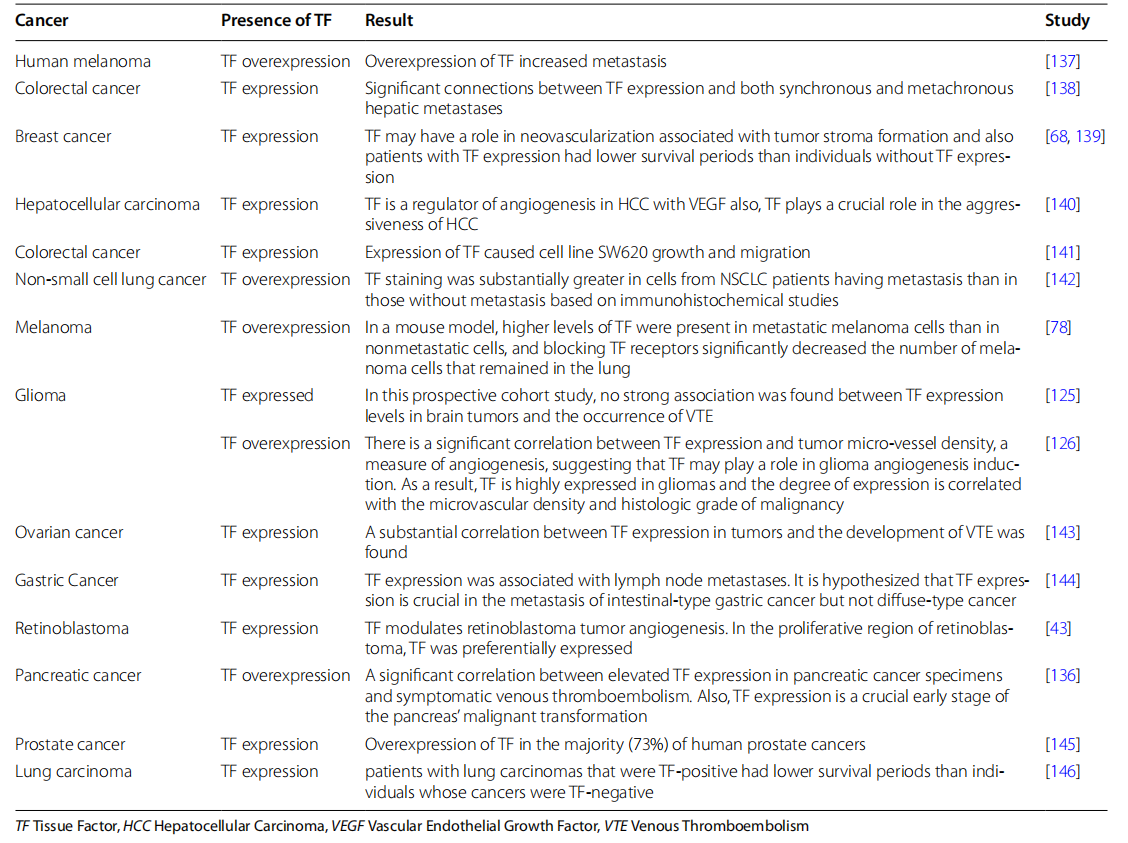
The role of CD142 in cancer
CD142 plays a role in tumor progression, invasion, metastasis, and angiogenesis in various cancers . Expression of CD142 on the surface of tumor cells leads to increased VEGF expression, thereby increasing angiogenesis. The cytoplasmic domain of CD142 plays a crucial role in angiogenesis. CD142 promotes uncontrolled cancer cell growth and inhibits apoptosis by activating the JAK2-STAT5 pathway, and can also promote tumor cell growth by activating RTKs. CD142 increases tumor cell angiogenesis and metastasis by increasing VEGF and inhibiting TSP. CD142 activates rac1, which in turn activates PKA, regulating MAPK and leading to cancer cell growth and metastasis. CD142 activates PAR2, thereby increasing cancer cell angiogenesis, invasion, and metastasis through multiple mechanisms. It leads to increased VEGF, bEGF, IL8, and βTGF, thus promoting tumor cell angiogenesis, invasion, and metastasis. Furthermore, it can stabilize β-catenin, inducing tumor cell invasion. PAR2 also activates the MAPK and ERK1/2 pathways, increases β-arrestin, phosphorylates cofilin, and leads to the polymerization of actin filaments at the cell periphery, thereby increasing tumor cell invasion and metastasis. CD142 activates the PI3K/AKT, MAPK, and FAK signaling pathways by binding to integrin α6β1 and β3vα.
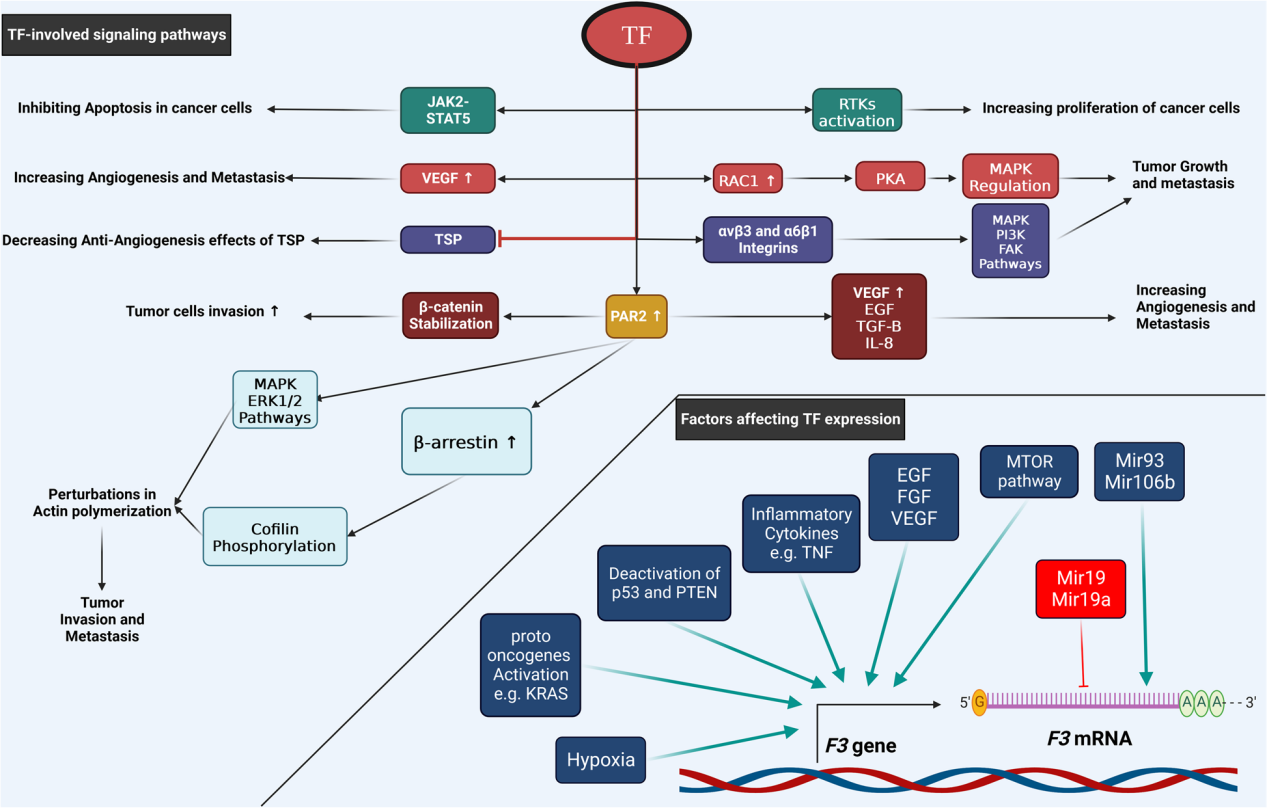
(Data source: Ahmadi SE, et al. Biomark Res. 2023)
CD142 targeted therapy
CD142 is highly expressed in various types of cancer. It activates the extrinsic coagulation pathway, promoting tumor progression and metastasis. Numerous studies have revealed the relationship between blood clotting and cancer progression. Therefore, CD142-targeted therapy can effectively reduce tumor growth, angiogenesis, and metastasis in many cancers. Currently, many drugs targeting CD142 are in clinical development.
Tisotumab vedotin is an antibody-drug conjugate targeting CD142, co-developed by Seagen and Genmab, and has been approved by the FDA for the treatment of solid tumors. It consists of Genmab's human monoclonal antibody that binds to CD142, Seagen's ADC technology (using a cleavable linker), and the cytotoxic drug monomethylauristatin E (MMAE).
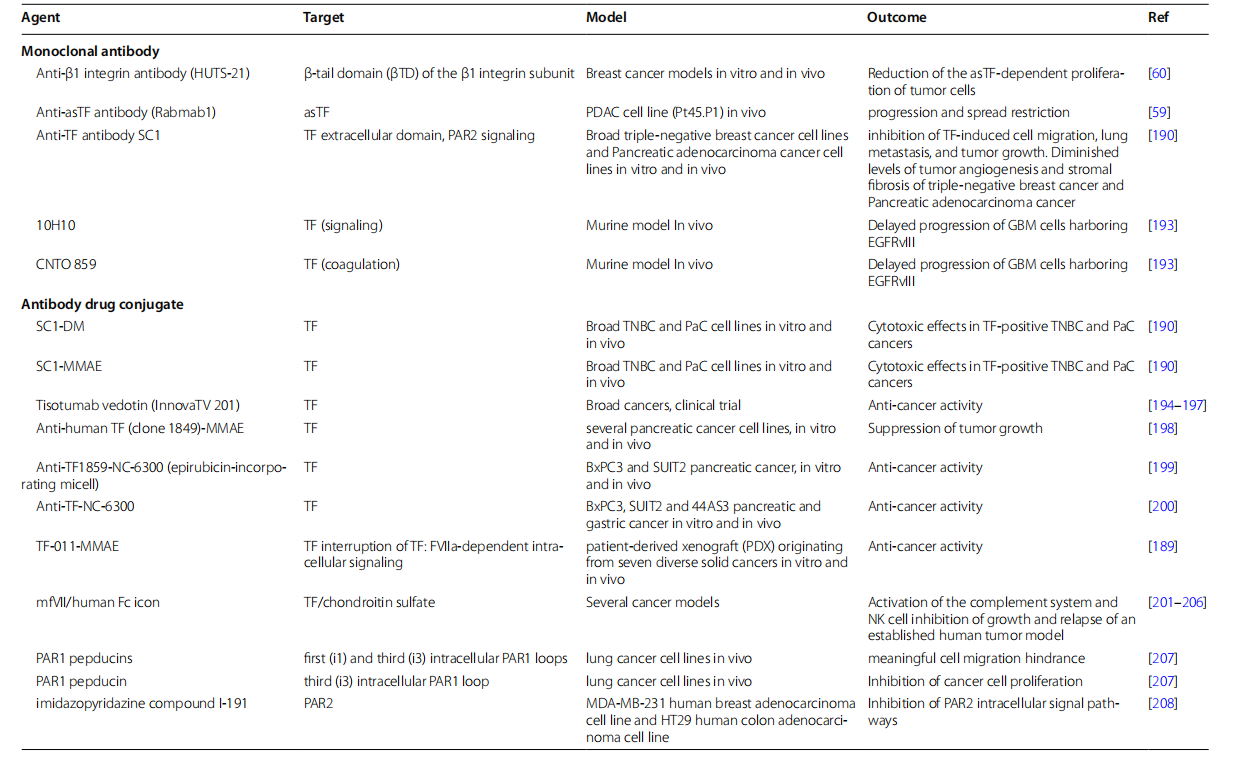
STRO-004 is an ADC targeting tumor-associated antigen (TF) developed by Supero Biopharma. TF is a clinically validated tumor-associated antigen expressed in multiple solid tumors. In preclinical studies, STRO-004 demonstrated robust antitumor activity, good tolerability, and higher exposure compared to approved treatments. Developed using Supero Biopharma's proprietary cell-free platform, STRO-004 features an Fc-silenced, high-affinity antibody with a site-specific β-glucuronidase-cleavable linker and a drug-to-antibody ratio of 8 (DAR8) heteroglycopolyphenyl loading. This design aims to improve stability, reduce off-target toxicity, and maximize efficacy. A phase 1 open-label, multicenter trial is evaluating the safety, pharmacokinetics, and preliminary antitumor activity of STRO-004 in patients with advanced TF-expressing solid tumors, including non-small cell lung cancer, head and neck squamous cell carcinoma, cervical cancer, colorectal cancer, pancreatic ductal adenocarcinoma, and bladder cancer. The dose-escalation phase includes multiple cohorts with escalating dose levels, supporting strong tolerability in non-human primates up to 50 mg/kg. Sutro is designed to allow for high introductory doses, with the goal of rapidly determining recommended phase 2 doses and early clinical indications.
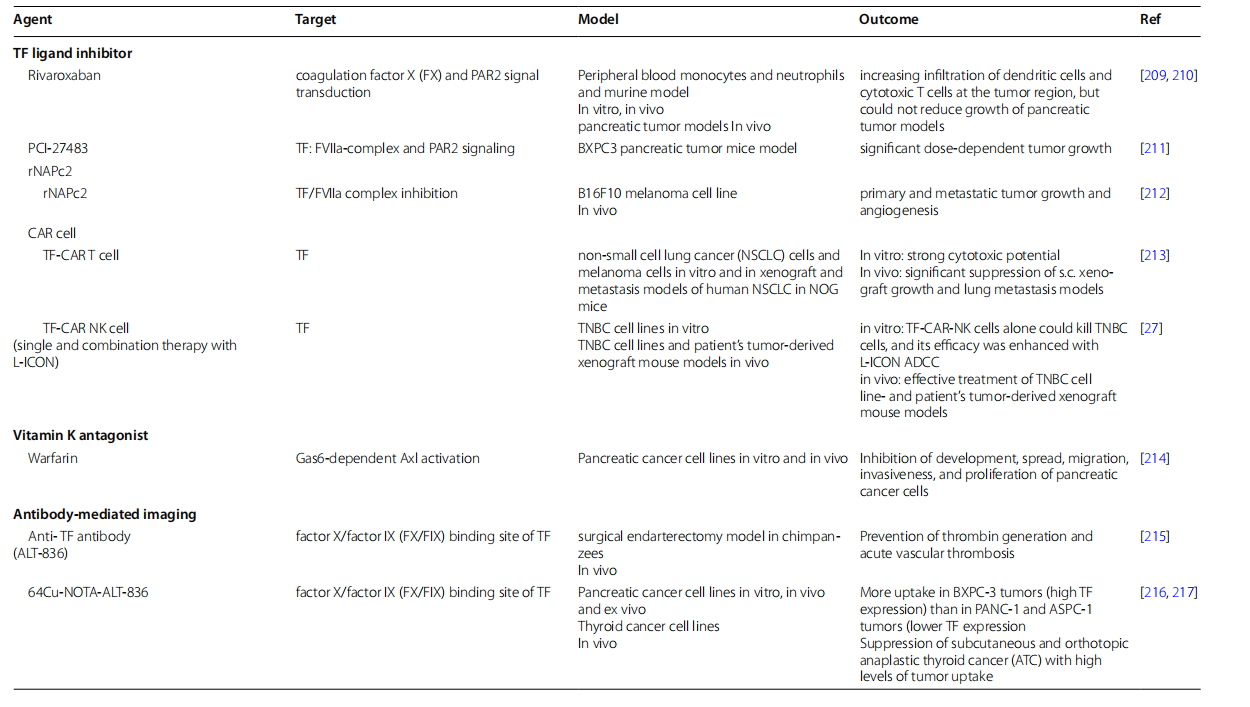
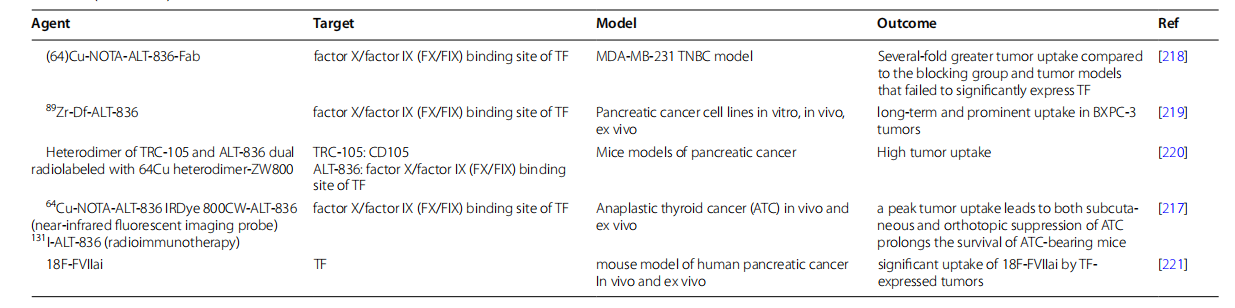
(Data source: Ahmadi SE, et al. Biomark Res. 2023)
