BTN1A1 has been shown to inhibit T cell activation both in vitro and in vivo. Strong BTN1A1 expression was detected in a range of solid tumor tissue samples, and its expression is mutually exclusive with PD-L1 expression because it inhibits PD-L1 upregulation induced by the Janus activating kinase/signal transduction and transcription activator pathway. Antibody-mediated BTN1A1 blockade inhibited tumor growth and enhanced immune cell infiltration in syngeneic tumor-carrying mice.

(Data source: Kim YS, et al. J Immunother Cancer. 2024)
BTN1A1 Basic Information
BTN1A1 (Butyrophilin Subfamily 1 Member A1) is an important member of the butyl whey protein (BTN) family. TN1A1 was first discovered in lactating mammary epithelial cells. It is a type I transmembrane glycoprotein with an extracellular IgV/IgC-like domain and an intracellular tail (related to signal transduction and intracellular protein interactions).
(Data source: Protter)
BTN1A1 inhibits CD8+ T cell activation
When T cells were stimulated with microbeads conjugated with anti-CD3, anti-CD28, and either BTN1A1, PD-L1, or control IgG, the ECD of BTN1A1 and PD-L1 inhibited T cell proliferation. In co-culture with activated T cells, PC3 tumor cells expressing the empty vector (EV) showed significant apoptosis induction, while PC3 cells overexpressing BTN1A1 exhibited reduced apoptosis and decreased IFN-γ production. BTN1A1-Fc exposure inhibited anti-CD3/IL-2-induced T cell IFN-γ secretion and aggregation in a dose- and time-dependent manner, confirming the immunosuppressive effect of this protein.

(Data source: Kim YS, et al. J Immunother Cancer. 2024)
Tumor cells overexpress BTN1A1
Given that BTN1A1 can suppress T cell responses both in vitro and in vivo when expressed in tumor cells, IHC staining with a rabbit monoclonal antibody against BTN1A1 (STC43H11-1) revealed significant and intense expression of BTN1A1 in head and neck, bladder, squamous non-small cell lung cancer (NSCLC), ovary, cervix, prostate, and other tumor tissues, while PD-L1 expression was relatively low in both frequency and intensity. There was little overlap in BTN1A1 and PD-L1 expression across any tumor analyzed, and BTN1A1 and PD-L1 messenger RNA (mRNA) levels were negatively correlated in cell lines.

(Data source: Kim YS, et al. J Immunother Cancer. 2024)
BTN1A1 Regulation of PD-L1
By transfecting PC3 cells with BTN1A1 overexpression or knockout with a luciferase reporter gene construct driven by the BTN1A1 or PD-L1 promoter, an empty pGL4 vector was used as a control. Exogenous BTN1A1 expression did not alter endogenous BTN1A1 promoter activity but did reduce PD-L1 promoter activity, while BTN1A1 knockout restored this reduced activity. PD-L1 protein levels decreased and increased after exogenous BTN1A1 expression and knockout, respectively. Co-immunoprecipitation showed that when amino acid (aa) 380-526 of BTN1A1 was deleted but not aa 331-526, BTN1A1 could bind to JAK1, thereby inhibiting the upregulation of downstream STAT/IRF9-mediated target genes, including PD-L1.

(Data source: Kim YS, et al. J Immunother Cancer. 2024)
BTN1A1 blocking characterization
When mice were intraperitoneally injected with different syngeneic tumor models (MC38, CT26, 4T1, LLC), anti-mBTN1A1 significantly inhibited tumor growth. When evaluating the therapeutic effect of anti-mBTN1A1/anti- PD-L1 combination therapy in these tumor models, better inhibition of MC38 and CT26 tumor growth was observed in mice receiving the combination therapy compared to anti-mBTN1A1 alone.
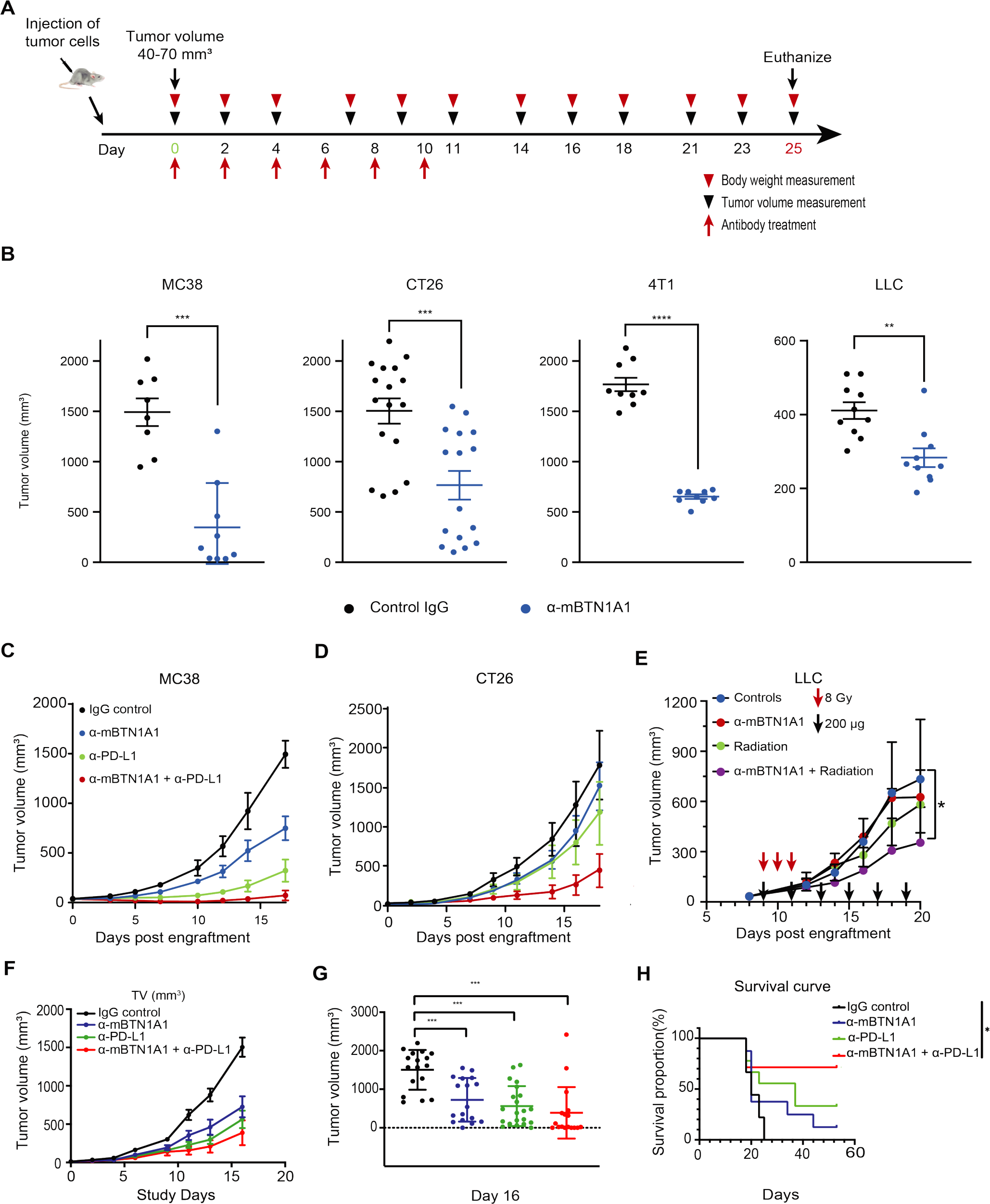
(Data source: Kim YS, et al. J Immunother Cancer. 2024)
BTN1A1 Therapeutic Potential
To evaluate the therapeutic potential of BTN1A1 inhibition, the anti-human BTN1A1 antibody STC810 increased STAT1/2 phosphorylation, IRF9 expression, and PD-L1 expression in treated tumor cells, and further enhanced the upregulation of CXCL9 and CXCL10 under IFN-γ stimulation. STC810 also enhanced the ability of anti-CD3/CD28 activated T cells to induce apoptosis in target PC3 cells. Targeting the immune checkpoint protein BTN1A1 is a feasible immunotherapeutic intervention strategy.

(Data source: Kim YS, et al. J Immunother Cancer. 2024)
