Clusterin (CLU), also known as complement cell lysis inhibitor (CLI), apolipoprotein J (APOJ), complement-associated protein SP-40, or aging-associated gene 4 protein (AAG4), participates in the occurrence and development of neurological, fibrotic, and metabolic diseases by regulating processes such as endocytosis and apoptosis. CLU is an important cancer regulator, controlling various cancer-related cellular events, including cancer cell proliferation, stem cell characteristics, survival, metastasis, epithelial-mesenchymal transition, treatment resistance, and inhibition of programmed cell death, to support cancer growth and recurrence.

(Data source: Praharaj PP, et al. Biochim Biophys Acta Rev Cancer. 2021)
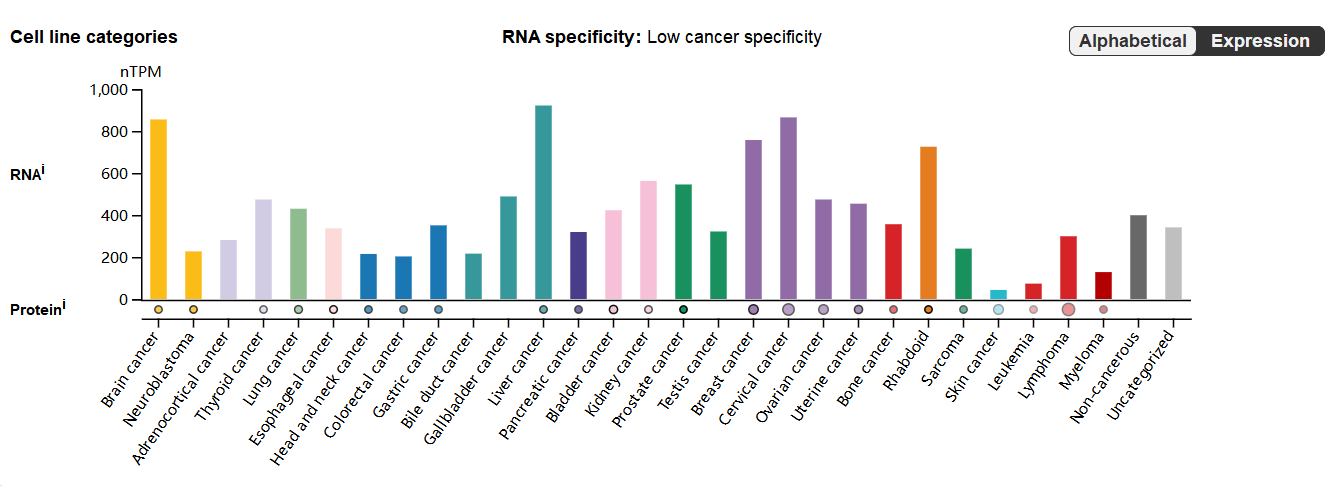
CLU expression distribution
CLU is mainly expressed in barrier epithelial cells, glial cells, ductal cells, secretory cells, and germline cells. CLU is highly expressed in tumor cells, including those with liver cancer, cervical cancer, brain cancer, breast cancer, prostate cancer, gallbladder cancer, ovarian cancer, thyroid cancer, uterine cancer, and lung cancer.

(Data source: uniprot)
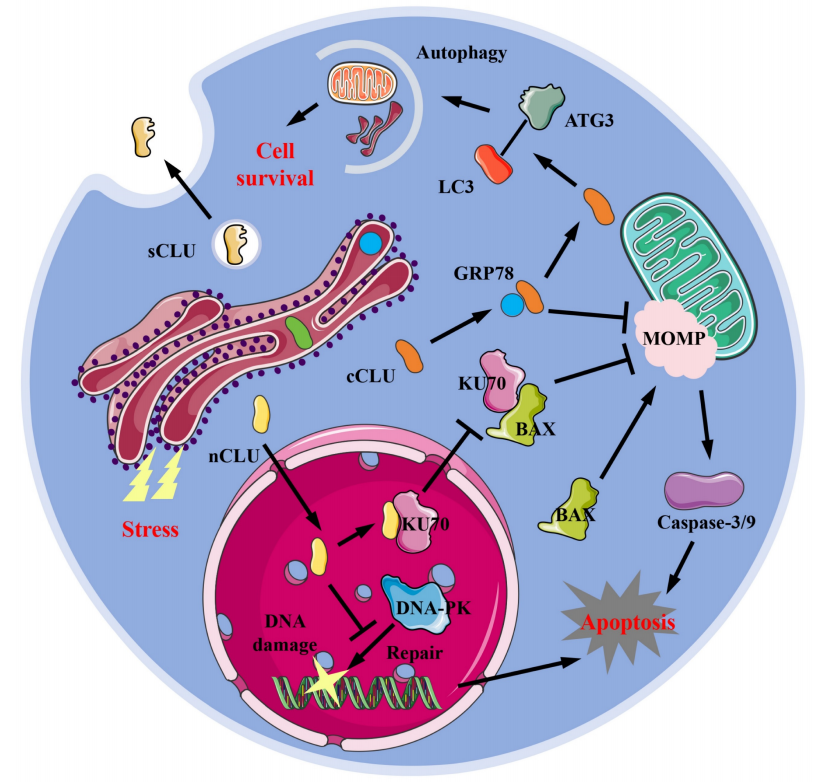
Structure and composition of CLU
The CLU gene is located on chromosome 8p21-p12. This gene produces at least 17 splice variants, including coding and non-coding variants. CLU is a glycoprotein that exists in cells in multiple forms, including karyotype (nCLU), cytoplasmic type (cCLU), and secretory type (sCLU).
Secretory CLU (sCLU): In the Golgi apparatus, CLU is cleaved into two segments by proteases: an α-chain (approximately 35-40 kDa) and a β-chain (approximately 35-40 kDa). The cleaved α- and β-chains do not separate but are tightly cross-linked by five intramolecular disulfide bonds, forming a stable, highly folded heterodimer, the mature secretory clusterin (sCLU), with a total molecular weight of approximately 75-80 kDa. sCLU typically exists stably in body fluids as a dimer or tetramer.
Cytoplasmic CLU (cCLU): A type of CLU consisting of two chains with a lower degree of glycosylation than sCLU, which remain in the cytoplasm to perform its function.
Nucleotype CLU (nCLU): The peptide chain formed does not undergo cleavage or N-glycosylation modification and usually functions within the cell nucleus.

(Data source: Yuste-Checa P, et al. Nat Struct Mol Biol. 2025)
(Data source: Du X, et al. Int J Med Sci. 2025)
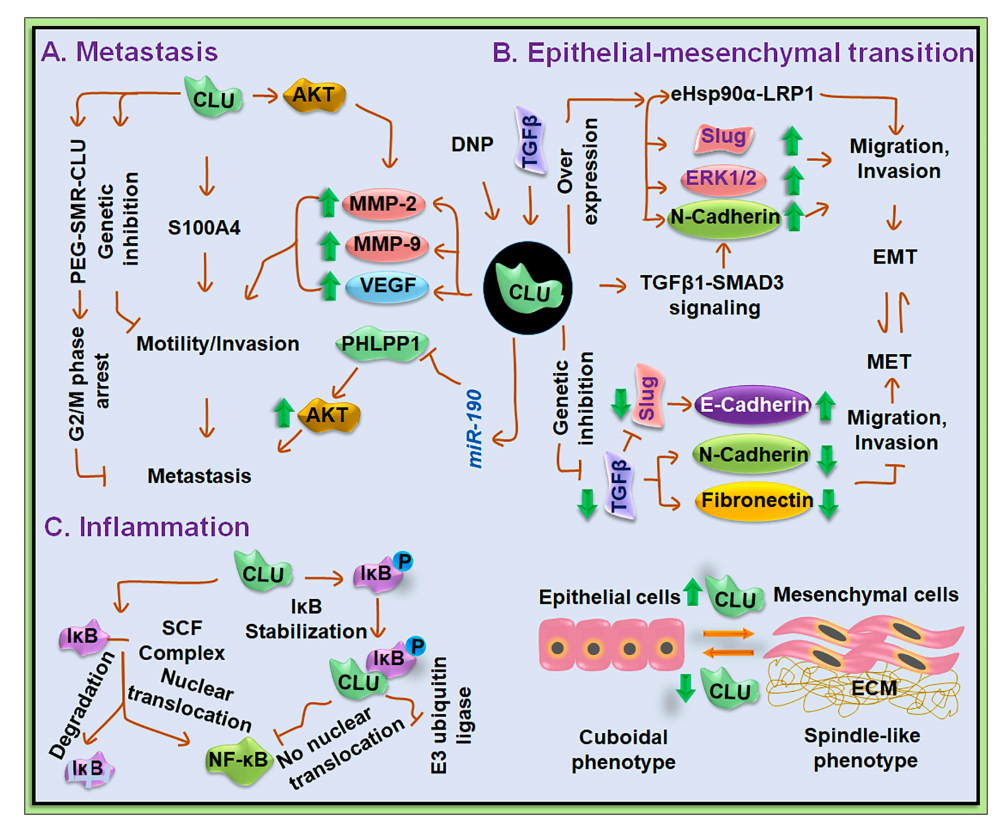
The role of CLU in cancer
sCLU promotes tumorigenesis and stabilizes the Ku70-BAX complex, thereby inhibiting mitochondrial translocation of BAX and preventing apoptosis. CLU gene repression weakens the interaction between the Ku70-BAX complex, leading to increased BAX translocation to mitochondria, subsequent release of cytochrome C, and sequential activation of caspase 9, initiating apoptosis. CLU also interacts with conformationally altered BAX on the mitochondria, hindering BAX dimerization and thus inhibiting BAX-dependent release of cytochrome C into the cytoplasm to suppress apoptosis. CLU gene repression also triggers Cdc25C activation, eliminating negative feedback inhibition and promoting Wee1-CDK1 activity, resulting in delayed mitotic termination. CLU also improves cancer cell survival by activating autophagy, whereby CLU interacts with LC3 through its LIR region and promotes LC3 lipidation via a stable LC3-Atg3 heterocomplex, triggering autophagosome generation. It also interacts with ULK1, promoting starvation-induced autophagy by modulating the AMPK/mTOR/ULK1 axis. CLU also plays a crucial role in the survival of cancer stem cells, promoting the growth of liver cancer stem cells by activating the AKT/GSK3β/β-catenin signaling pathway.
nCLU binds directly to the Ku70 protein, inhibiting the formation of the complex (Ku70-BAX), leading to the translocation of more free cytoplasmic BAX into the mitochondria, thereby triggering mitochondrial apoptosis. nCLU also promotes cell death by interacting with an anti-apoptotic protein (Bcl-xL), which is present on the outer mitochondrial membrane and regulates the permeability of the outer mitochondrial membrane (MOMP), thereby inducing apoptosis.

(Data source: Praharaj PP, et al. Biochim Biophys Acta Rev Cancer. 2021)
Clusterin is a key molecule driving cancer metastasis. It directly promotes the growth, migration, and invasion of tumor cells by upregulating invasion and metastasis markers such as VEGF and MMP-9 and activating signaling pathways such as AKT2.
In regulating epithelial-mesenchymal transition (EMT), CLU induces cells to acquire a spindle-shaped mesenchymal phenotype and enhances mobility by activating multiple signaling pathways such as TGF-β1-SMAD3 and eHsp90α-LRP1; while inhibiting CLU can reverse this process.
CLU also regulates the inflammatory response in the tumor microenvironment by modulating MMP-9 expression and affecting pathways such as ERK/PI3K/NF-κB. Its effects are context-dependent; it can both inhibit NF-κB by stabilizing IκBα and promote its nuclear translocation under certain circumstances.
Targeted therapy for CLU
Sotevtamab (AB-16B5) is a humanized IgG2 antibody targeting EMT-induced secretory agglutinin (sCLU). AB-16B5 effectively blocks EMT, and preclinical data indicate that treatment with AB-16B5 alone reduces tumor growth, while animals treated with AB-16B5 showed increased survival. These findings, along with several other in vivo results, suggest that inhibition of tumor-associated sCLU with AB-16B5 increases response to chemotherapy and reduces tumor invasion and growth.
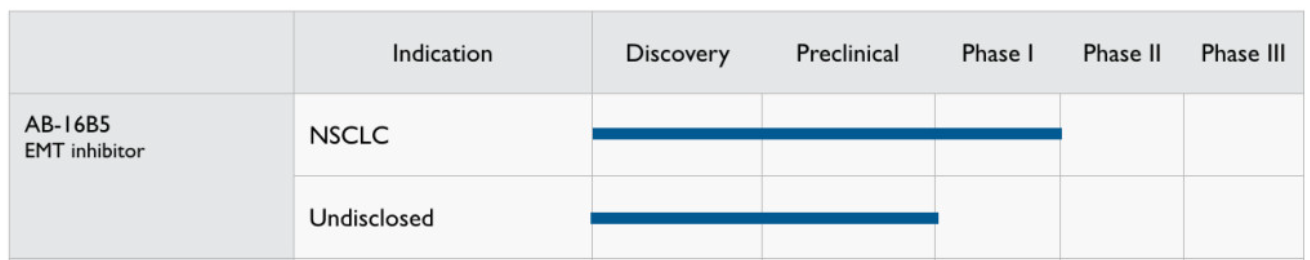
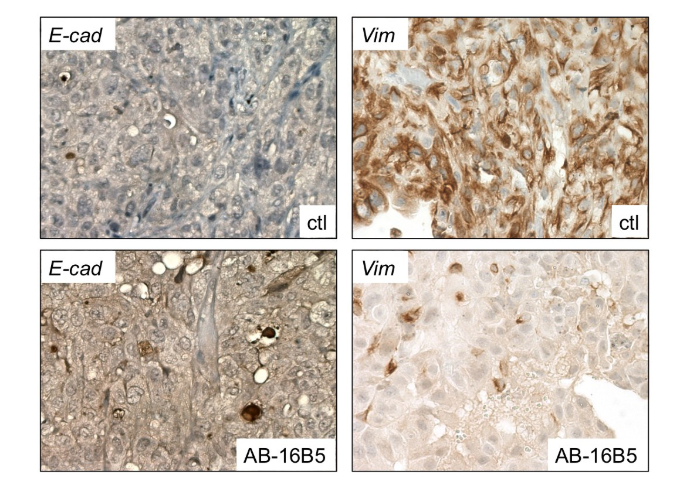
(Data source: Alethia Biotherapeutics official website)
