Tumor necrosis factor receptor superfamily member 12A (TNFRSF12A), also known as fibroblast growth factor-induced early response protein 14 (FN14), TWEAKR, or CD266, is the TNFSF12/TWEAK receptor. It is a weak inducer of apoptosis in certain cell types. It promotes angiogenesis and endothelial cell proliferation. It may regulate cell adhesion to matrix proteins. The TWEAK/Fn14 signaling axis plays a crucial role in tumor progression by promoting cell survival, angiogenesis, and immune escape.
(Data source: Wang S, et al. Stem Cell Res Ther. 2022)
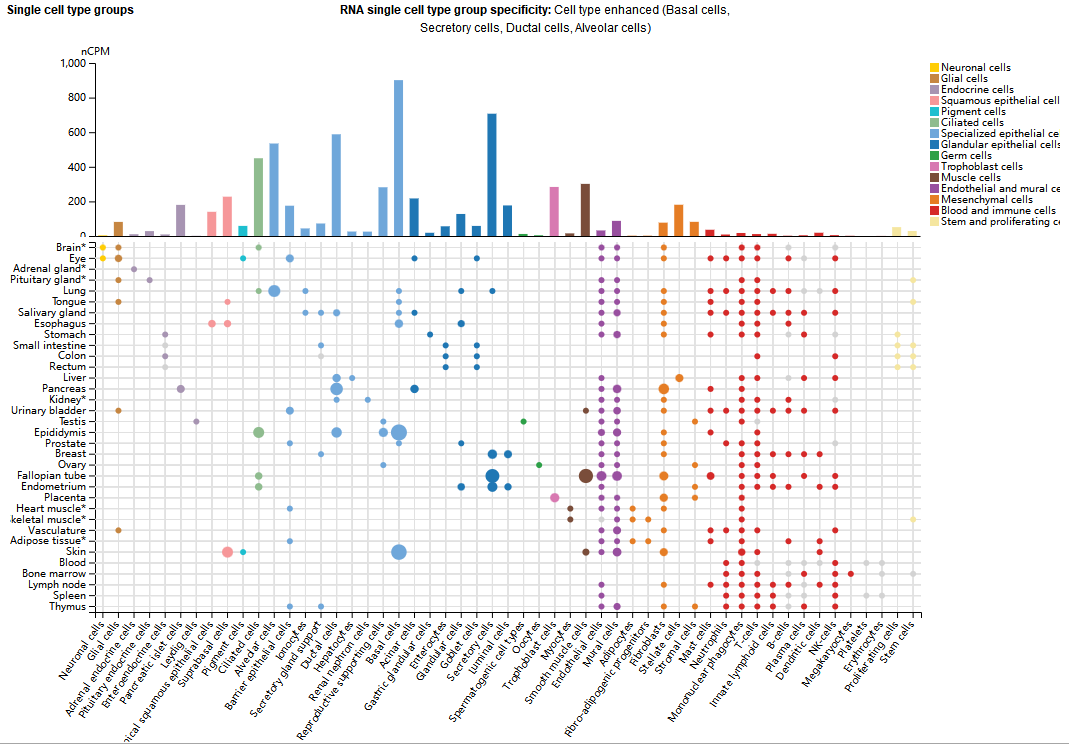
TWEAKR expression distribution
TWEAKR is expressed in endothelial cells, smooth muscle cells, epithelial cells, fibroblasts, and various immune cells. Its expression level is significantly elevated in various solid tumors (such as breast cancer, glioma, and pancreatic cancer), fibrotic diseases (liver, kidney, and lung fibrosis), and chronic inflammatory lesions such as atherosclerosis.
(Data source: uniprot)
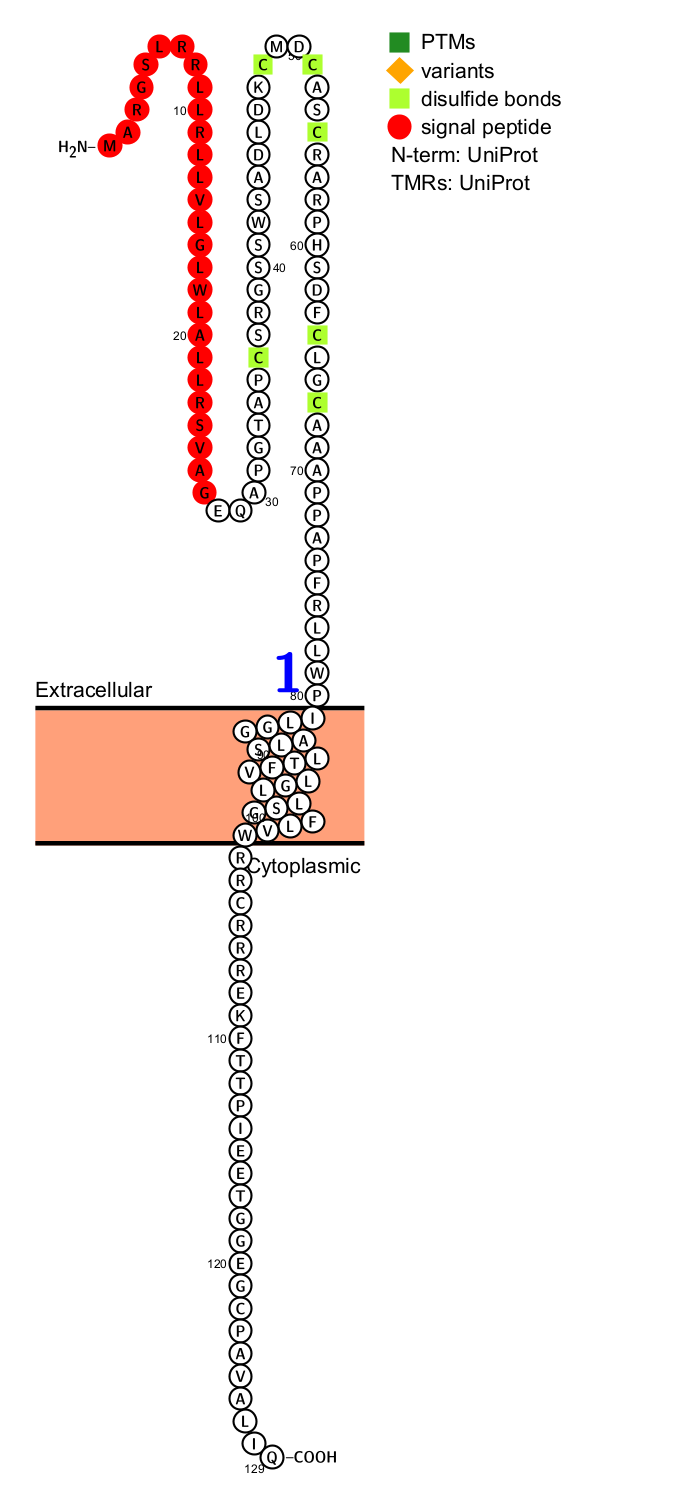
TWEAKR's structure
TWEAKR is a type I transmembrane protein, extremely small in size. Its precursor protein contains only 129 amino acids, and after processing by signal peptidase, the mature form contains only 102 amino acids, making it the smallest known member of the TNFR superfamily. Its extracellular region contains six conserved cysteine residues, forming a typical TNFR-like domain responsible for the specific binding of the ligand TWEAK. The intracellular region is short and lacks intrinsic enzymatic activity, but contains a single TNFR-associated factor (TRAF) consensus binding motif, essential for recruiting downstream adaptor proteins (such as TRAF family proteins) to initiate signal transduction.
(Data source: uniprot)
TWEAKR 's signaling pathways and regulation
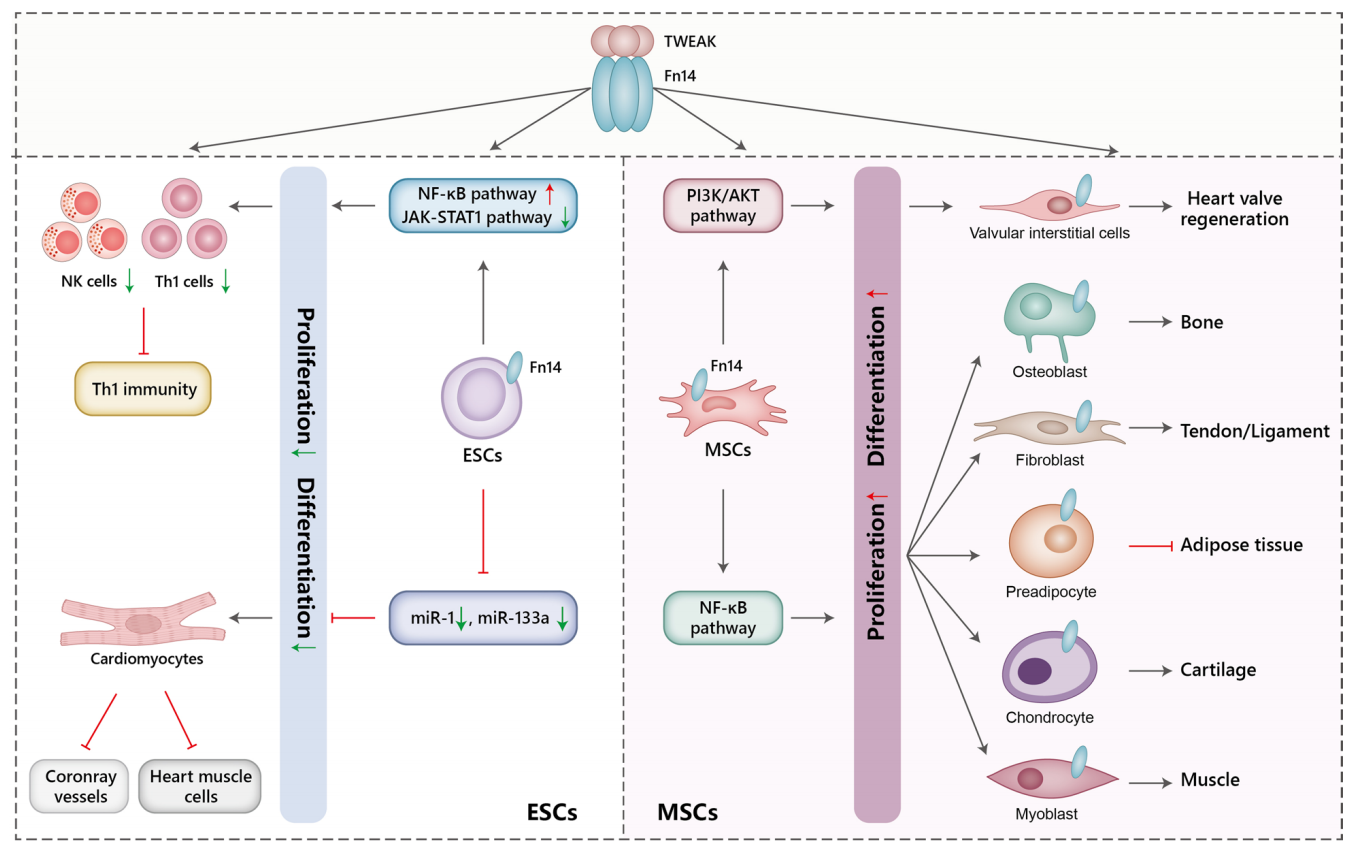
Binding of the ligand TWEAK induces trimerization of the receptor TWEAKR, which in turn recruits adaptor proteins such as TRAF1, TRAF2, TRAF3 and TRAF5 through the intracellular region, activating multiple key signaling pathways.
The classic NF-κB pathway:It is triggered by RIP1 ubiquitination mediated by cIAP1/2 and mainly drives the expression of genes that promote survival, proliferation, and inflammation.
Notch signaling pathway:It can be further induced by NF-κB activated by TWEAK and plays a key role in the fate determination of specific cells (such as muscle satellite cells and pancreatic progenitor cells).
The MAPK signaling pathway:It activates the AP-1 transcription factor via MKKKs, thereby driving gene expression. Activated genes lead to increased satellite cell proliferation and decreased differentiation, as well as decreased erythroid progenitor cell proliferation and differentiation.

(Data source: Wang S, et al. Stem Cell Res Ther. 2022)
The role of TWEAKR in cancer
TWEAK /FN14 is a multifunctional oncogenic signaling hub that drives cancer cell proliferation, invasion, metastasis, and resistance to apoptosis by directly activating key pathways such as NF-κB within cancer cells. This signaling axis can also reshape the tumor microenvironment, creating conditions for tumor growth by inducing fibrosis, angiogenesis, and immunosuppression.
TWEAK released in the tumor microenvironment is primarily produced by macrophages. The signaling pathway involves Fn14, expressed by malignant cholangioblasts and cancer-associated fibroblasts (CAFs) within the tumor niche, and functions via the NF-κB pathway. Upon TWEAK stimulation, CCA cells produce MCP-1, promoting the recruitment of circulating monocytes and generating other factors that polarize monocytes into the tumor-associated macrophage (TAM) phenotype. Conversely, TWEAK-stimulated CAFs begin to proliferate and secrete extracellular matrix (ECM) components. Once the TAM phenotype is formed, macrophages themselves also produce MCP-1, further enhancing monocyte chemotaxis. Furthermore, TAMs may influence various pro-invasive functions within the tumor microenvironment, including tumor cell proliferation and chemotherapy resistance, angiogenesis, and immune escape.

(Data source: Zaitseva O, et al. Front Pharmacol. 2022)
TWEAKR 's targeted therapy
Enavatuzumab (PDL192) is a monoclonal antibody that targets TWEAKR. The antibody directly inhibits tumor growth and survival by binding to Fn14, disrupting this signaling pathway. The binding of Enavatuzumab to the Fn14 receptor triggers multiple cellular responses, including apoptosis (programmed cell death) within cancer cells, thereby preventing their continued proliferation.
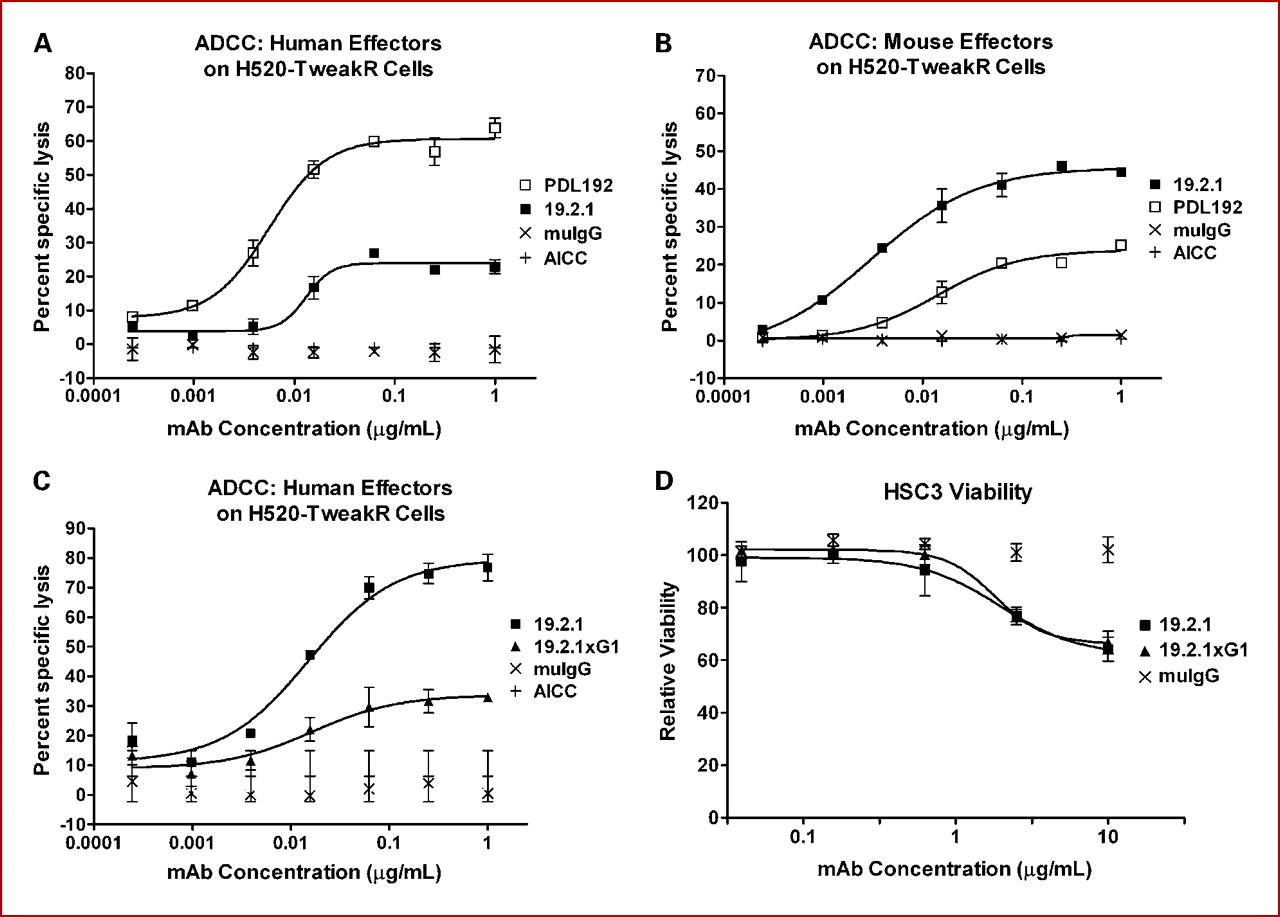
(Data source: Patricia AC, et al. Clin Cancer Res. 2010)
In addition to directly killing tumor cells, Enavatuzumab also enhances the body's immune response through antibody-dependent cell-mediated cytotoxicity (ADCC). During this process, immune effector cells such as natural killer (NK) cells recognize and destroy antibody-bound tumor cells, enhancing the immune system's ability to target and eliminate cancer cells. This dual effect of directly targeting tumor cells and activating the immune system makes Enavatuzumab a powerful tool in precision oncology.
