Integrin alpha-2 (ITGA2), also known as CD49b, is a key member of the integrin family. It serves as a receptor for laminin, collagen, collagen C-peptide, fibronectin, and E-cadherin. It recognizes the proline hydroxylation sequence GFPGER in collagen. It is responsible for the adhesion of platelets and other cells to collagen, the regulation of collagen and collagenase gene expression, force generation, and the organization of newly synthesized extracellular matrix. ITGA2 primarily mediates cell adhesion to the extracellular matrix (ECM), playing a central role in collagen recognition. Its aberrant expression is closely associated with tumor progression, stem cell maintenance, immune evasion, and vascular disease, making it a promising target for cross-cancer therapeutics.
Expression distribution of ITGA2
ITGA2 is expressed in multiple cell types, widely expressed in epithelial cells, endothelial cells and mesenchymal stromal cells (MSCs), and is involved in maintaining tissue homeostasis.
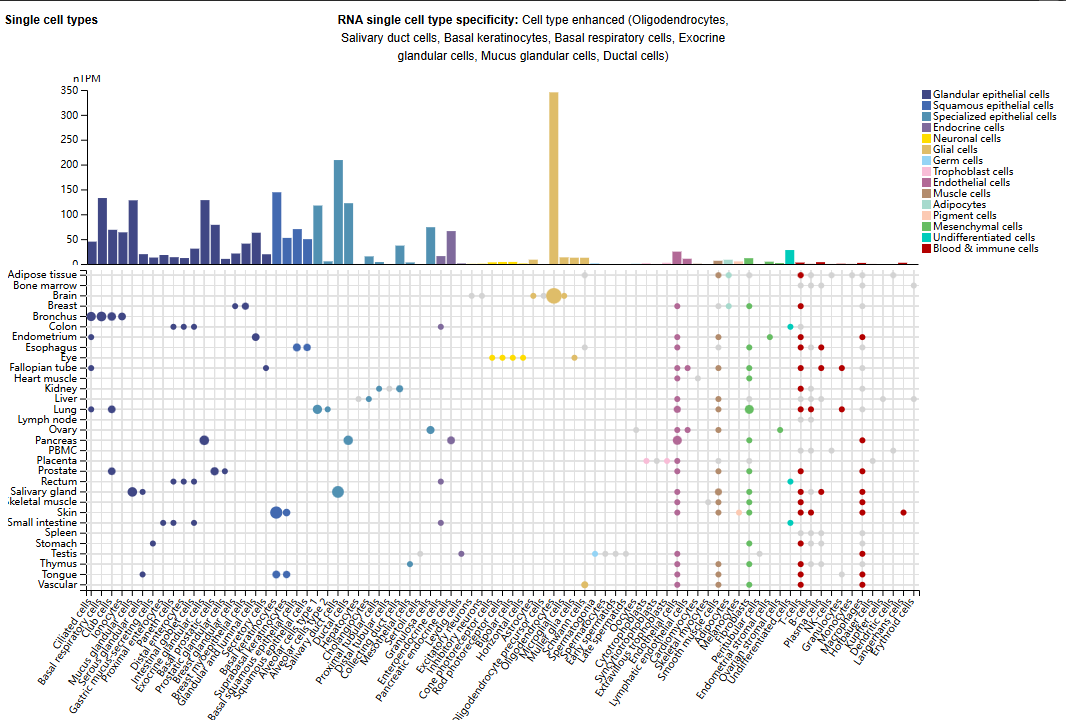
(Data source: Uniprot)
The structure of ITGA2 and its receptor
ITGA2 is a type I transmembrane protein composed of 1,181 amino acids, with a long N-terminal extracellular domain, a transmembrane region, and a short C-terminal cytoplasmic region. The extracellular domain contains a conserved MIDAS domain (metal ion-dependent adhesion site), which directly binds to specific sequences in type I and type IV collagen (e.g., GFOGER) and undergoes Mg²⁺ -dependent conformational changes.

(Data source: Uniprot)
ITGA2 normally associates with integrin β1 ( ITGB1 ) to form a heterodimer, acting as a specific receptor for type I and type IV collagen.
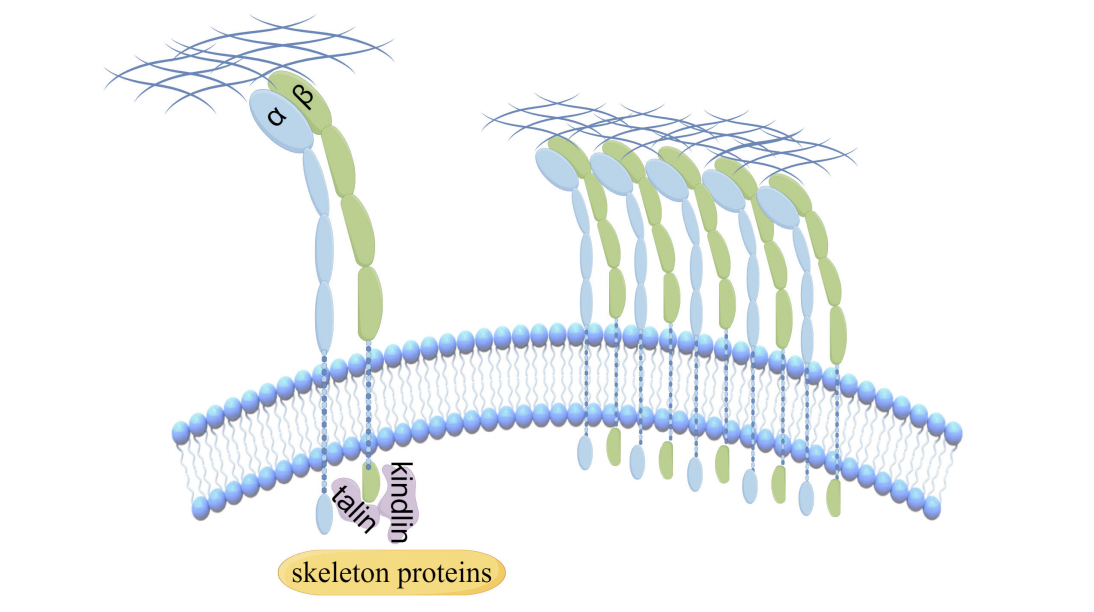
(Data source: Liu T, et al. Front Oncol. 2024)
Signaling pathway and regulation of ITGA2
ITGA2 (integrin α2) binds to collagen or laminin in the extracellular matrix, initiating signaling. Upon binding, integrins undergo conformational changes, recruiting intracellular kinases (such as Src and FAK) and activating downstream pathways. Wnt signaling within integrins triggers phosphatidylinositol 3-kinase (PI3K) activation, which in turn regulates glycogen synthase kinase 3β (GSK3β), ubiquitin (Ub)-induced β-catenin degradation, nuclear localization, and expression of Wnt-targeted genes, leading to tumorigenesis and malignant progression.

(Data source: Liu T, et al. Front Oncol. 2024)
Regulation of ITGA2 in the tumor microenvironment
ITGA2 regulates immune cell function, CAFs activation, and immune checkpoint regulation in the tumor microenvironment through multiple mechanisms, thereby affecting tumor progression and immune escape.
Regulation of Immune Cell Function: ITGA2 serves as a maturation marker in regulatory T cells (Tregs) and type 1 regulatory T cells (Tr1). Although its function in these cells may not be directly involved in immunosuppression, ITGA2 expression is closely associated with the activity of immunosuppressive cells. ITGA2 is expressed in tumor-infiltrating CD8+ T cells and may affect T cell migration and function through interaction with collagen, thereby affecting immune surveillance.
Activation of tumor-associated fibroblasts (CAFs): ITGA2 serves as a surface marker in CAFs. CAFs secrete TSP-4, which binds to ITGA2 and activates heat shock factor 1 (HSF1), promoting the expression of TGF-β1 and paracrine signaling, thereby enhancing the positive feedback loop between CAFs and tumor cells and promoting tumor malignant progression. In hepatocellular carcinoma (HCC), CAFs interact with tumor cells through the COL1A1-ITGA2 signaling pathway, activating the YAP signaling pathway, regulating the transcriptional heterogeneity and mesenchymalization of tumor cells, and thus promoting tumor progression.
Immune checkpoint regulation: ITGA2 promotes tumor immune escape by enhancing STAT3 phosphorylation and upregulating programmed death ligand 1 (PD-L1) expression. This suggests that ITGA2 may inhibit anti-tumor immune responses by regulating immune checkpoints in the tumor microenvironment.
ITGA2 is a tumor marker for colorectal cancer (CRC) . Upregulation of ITGA2 promotes cancer cell migration and proliferation by enhancing CRC cell adhesion to collagen and laminin. Furthermore, TGF-α secreted by CRC cells enhances ITGA2 expression, leading to increased cell adhesion to type IV collagen, thereby promoting cancer cell metastasis and spread .

(Data source: Liu T, et al. Front Oncol. 2024)
Targeted therapy for ITGA2
Vatelizumab is a humanized monoclonal antibody that binds to integrin α2. Originally developed by Chromos Molecular Systems, it was subsequently developed by Glenmark Pharmaceuticals SA and Sanofi. Vatelizumab has been investigated for the treatment of a variety of conditions, including chronic obstructive pulmonary disease, multiple sclerosis, retinal diseases, rheumatoid arthritis, and ulcerative colitis. However, due to unsatisfactory clinical trial results, Sanofi abandoned its joint development with Indian pharmaceutical company Glenmark Pharmaceuticals in 2016. Currently, development of this antibody drug is terminated.

(Data source: clinicaltrials)
