EPHA3 belongs to the Eph receptor tyrosine kinase (RTK) family and is a type I transmembrane glycoprotein with a highly conserved structure. EPHA3 plays a crucial role in intercellular signaling, particularly in various physiological and pathological processes such as nervous system development and cancer. Currently, the application prospects of EPHA3 are beginning to translate into clinical applications, with substantial research progress in areas ranging from neurological diseases and cardiovascular diseases to cancer and tissue regeneration, potentially leading to novel treatment strategies for many intractable diseases in the future.
Eph receptor tyrosine kinase family
The Eph receptor tyrosine kinase family is mainly divided into two subclasses, EphA and EphB, based on sequence homology and ligand binding preference. It includes nine members of the EphA receptor (EphA1-8, EphA10) and five members of the EphB receptor (EphB1-4, EphB6). The ligands (Ephrins) are further divided into two subtypes, Ephrin-A and Ephrin-B, based on their attachment method to the cell membrane. The Ephrin-A subtype, anchored to the cell membrane via a glycosylphosphatidylinositol chain, comprises five types, while the Ephrin-B subtype comprises three types. These are single-transmembrane proteins composed of intracellular structures, transmembrane portions, and extracellular structures.
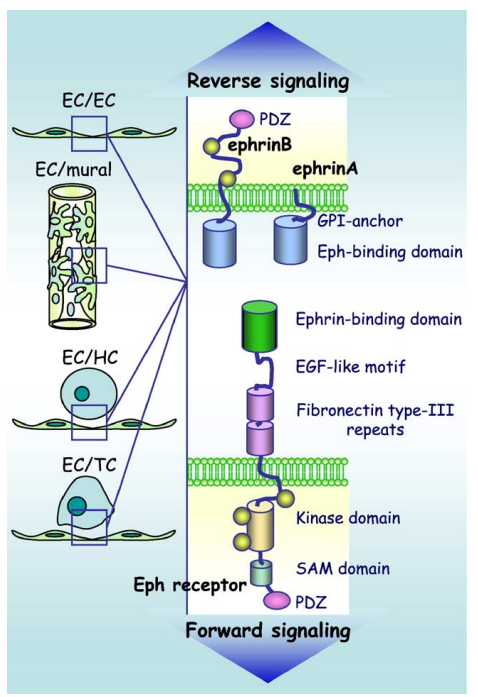
(Data source: Héroult M, et al. Héroult M. 2006)
Basic structure of EPHA3
EPHA3 gene is located on chromosome 3 (3p11.2) and encodes 983 amino acids (aa). It is a secreted protein. The initial hydrophobic region, consisting of the first 20 amino acid residues, is the signal peptide (SSP). The region from 21 aa to 983 aa is the main chain of the gene, containing numerous ubiquitination, phosphorylation, and glycosylation modification sites. The extracellular region of the EPHA3 protein is from 21 aa to 541 aa, including an N-terminal globular domain (GIb), a cysteine-rich linker region composed of a Sushi domain and an EGF-like domain, and two fibronectin type III repeat regions. The GIb domain is the key domain for ligand binding, determining the receptor-ligand binding characteristics. The transmembrane region is from 542 aa to 565 aa; the intracellular region is from 566 aa to 983 aa, including the tyrosine kinase activity domain, the SAM domain, and the PDZ domain. The domain with tyrosine kinase activity includes a juxtamembrane region and an adjacent kinase region. The SAM domain is highly conserved in the Eph protein family, and the tyrosine residues in the SAM structure are essential sites for the aggregation of receptor signaling molecules.
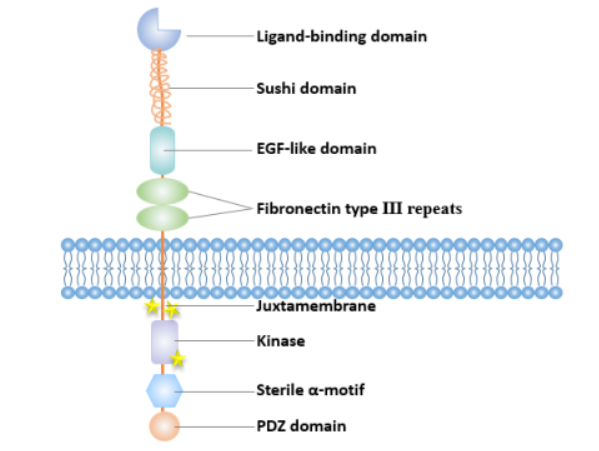
(Data source: Pasquale EB, et al. Nat Rev Mol Cell Biol. 2005)
Distribution and expression of EPHA3
EPHA3 is expressed weakly in most normal tissues, but its expression is more widespread during embryonic development, especially in the nervous and cardiac systems. However, it is significantly overexpressed in various cancers, including bladder cancer, colorectal cancer, esophageal cancer, gastric cancer, and prostate cancer, and is associated with tumor invasion and migration.
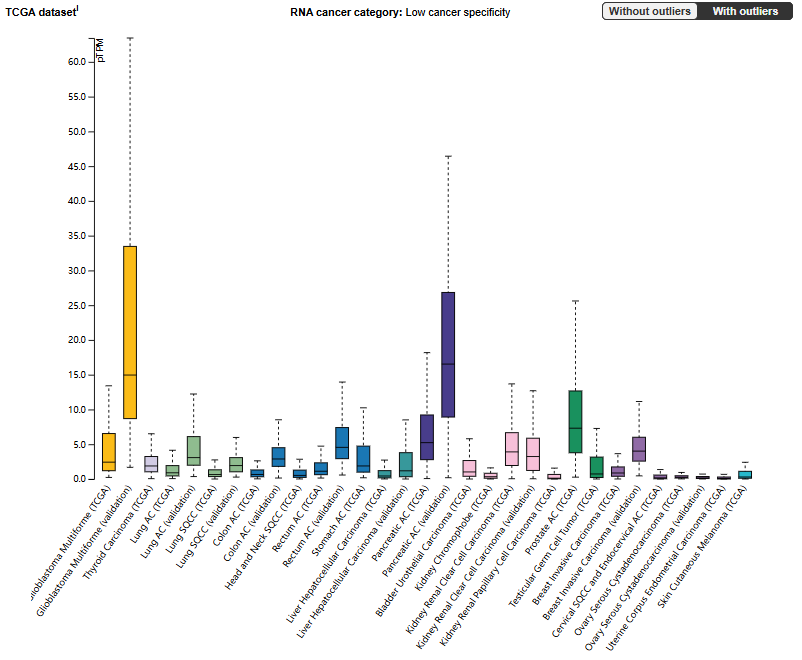
(Data source: proteinatlas.org)
The role of EPHA3 in neural development
Eph-Ephrin binding influences cell development by modulating cell-cell adhesion and deadhesion, controlling cell localization during normal and oncogenic development. This is crucial for embryological processes, particularly the development of the nervous system.
1. Axon guidance: This is one of its most fundamental functions in the nervous system. Studies have shown that presenilin-1 (PS1)/γ-secretase can cleave EPHA3 in a ligand-independent manner to produce an intracellular domain (ICD) fragment. This fragment binds to nonmyosin IIA and increases its phosphorylation (Ser1943), thereby promoting NMIIA filament depolymerization and cytoskeleton rearrangement, mediating axon elongation.

(Data source: Javier-Torrent M, et al. Elife. 2019)
2. Synaptic plasticity: EPHA3 also plays a role in synaptic membrane adhesion, which may affect cognitive functions such as learning and memory.
3. Heart Development: During heart formation, EPHA3 participates in regulating the migration and differentiation of cardiomyocytes, as well as the formation of the atrioventricular canal and ventricular septum. Studies have reported that EPHA3 knockout mice exhibit severe heart development defects, with approximately 75% of homozygous mutants dying from heart failure within 48 hours of birth.


(Data source: Stephen LJ, et al. Dev Biol. 2007)
Clinical application of EPHA3
1. CAR-T cell therapy targeting EPHA3: Preclinical studies have shown that this therapy can not only effectively eliminate tumors, but also establish a protective memory T cell response and achieve sustained complete remission for up to 6 months in a mouse model carrying tumors.
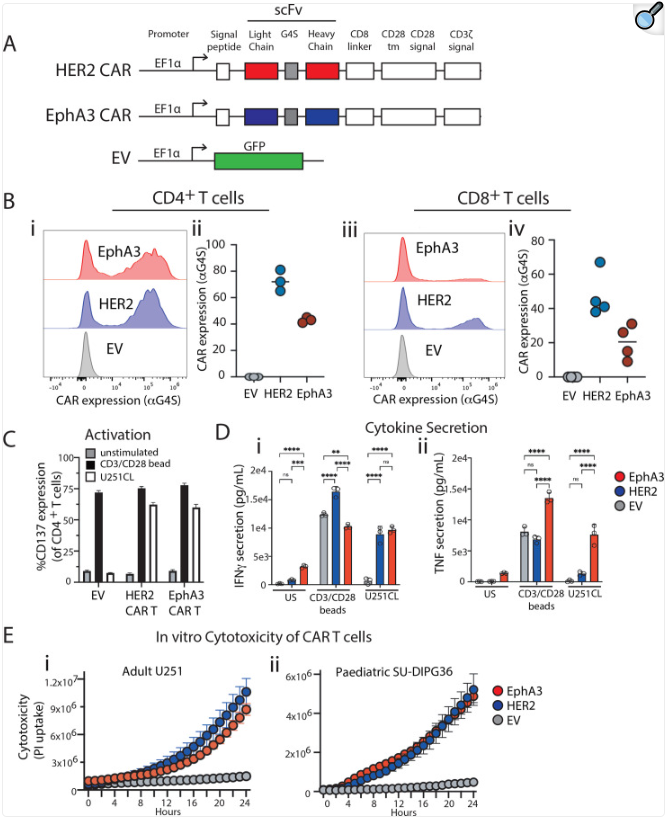
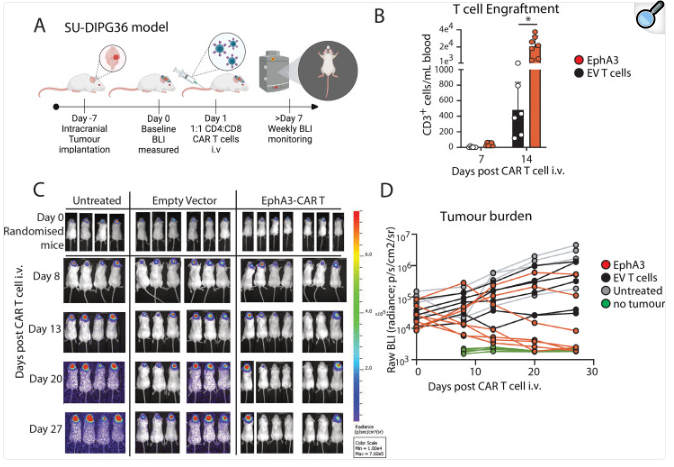
(Data source: Lertsumitkul L, et al. J Immunother Cancer. 2024)
2. Monoclonal antibody drug: Ifabotuzumab (KB004), a monoclonal antibody targeting EPHA3, has had its safety preliminarily validated in clinical studies of hematologic malignancies. Based on this, a Phase I clinical trial of KB004 for the treatment of recurrent glioblastoma has been completed, aiming to explore its safety, biodistribution, and preliminary efficacy.
