Myelin-associated glycoprotein (MAG) belongs to the immunoglobulin superfamily and is a type I transmembrane glycoprotein specifically expressed by oligodendrocytes and Schwann cells in the nervous system. It primarily exerts a key signaling regulatory role through its extracellular immunoglobulin (Ig)-like domain. It participates in the pathological processes of various neurological diseases, serving as a core diagnostic marker and therapeutic target for anti-MAG antibody-associated peripheral neuropathy, and is also an important pathogenic factor in diseases such as multiple sclerosis, spinal cord injury, and hereditary spastic paraplegia (SPG75). Currently, MAG-based immunotherapy and antigen-specific treatment strategies are moving from basic research to clinical application.
Basic Structure of MAG
The MAG gene is located on chromosome 19 (19q13.12) and encodes 626 amino acids (aa). The initial hydrophobic region consisting of the first 19 amino acid residues is called the signal peptide (SSP). The region from 20 aa to 626 aa is the main chain of the gene, containing 8 N-glycosylation sites linked to asparagine and multiple disulfide bond sites. The extracellular region of the MAG protein is from 20 aa to 516 aa, containing 5 immunoglobulin (Ig)-like domains, which are the most prominent structural feature of MAG. Each domain is stabilized by intrachain disulfide bonds. The transmembrane region from 517 aa to 536 aa anchors the MAG protein to the myelin sheath on the cell membrane. The intracellular region from 537 aa to 626 aa contains two isoforms generated by alternative splicing: long (L-MAG) and short (S-MAG), which can initiate different intracellular signal transduction pathways for fine-tuning.

(Data source: Quarles RH, et al. Journal of neurochemistry. 2007)
MAG distribution and location
At the tissue level, MAGs are mainly distributed in the myelin fiber bundles of the white matter of the brain and spinal cord, with the corpus callosum being a highly enriched area. Its subcellular localization is very precise, mainly located in the paraaxonal membrane of the innermost layer of myelin that is in direct contact with the axon, and partly in the mesaxon and paranodal loops. That is, it is mainly present in the non-compacted myelin region, while there is no MAG distribution in the compact myelin.
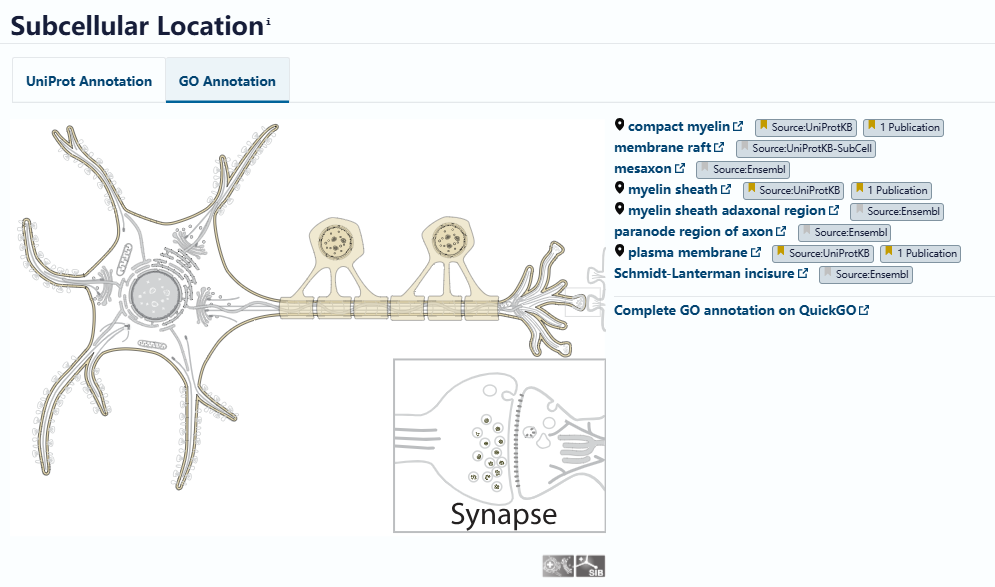
(Data source: uniprot)
MAG in neurological diseases
1. Core targets of autoimmune neurosis
Studies have found a causal relationship between IgM protein and polyneuropathy, with IgM targeting myelin-associated glycoprotein (MAG) in about half of the cases. Rituximab is a commonly used therapy, but its efficacy has not reached the ideal treatment goals in randomized controlled trials, which has driven the development of novel targeted therapies (such as BTK inhibitors).
2. Inhibitory factors of axonal regeneration after central nervous system injury
MAG is the first myelin-derived axonal growth inhibitor protein to be identified. It works in conjunction with other inhibitory factors such as Nogo and OMgp to inhibit axonal regeneration by promoting growth cone collapse. This is one of the important reasons why central nervous system injuries such as spinal cord injuries are difficult to repair.
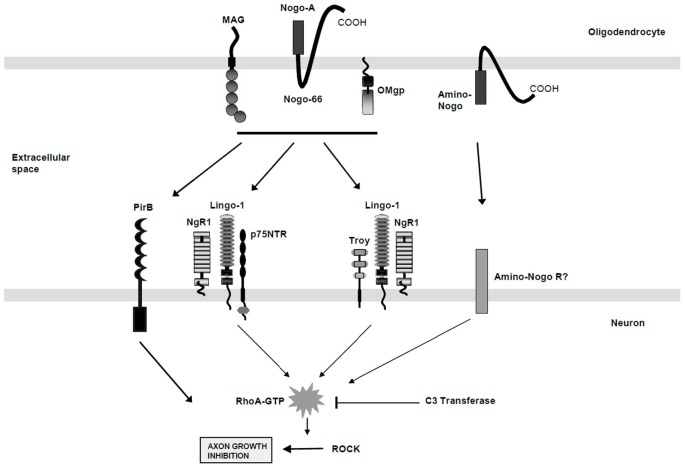
(Data source: McKerracher L, et al. Front Mol Neurosci. 2015)
3. Pathological markers of multiple sclerosis (MS)
In the pathological classification of multiple sclerosis (MS), type III lesions are characterized by the preferential loss of myelin-associated glycoprotein (MAG) compared to other myelin proteins, which is considered a marker of metabolically stressed oligodendrocyte damage.
4. The pathogenic gene for hereditary spastic paraplegia (SPG75)
Mutations in the gene encoding myelin-associated glycoprotein (MAG) can lead to a rare, complex, autosomal recessive form of spastic paraplegia. Patients often present with spastic paraplegia, peripheral sensorimotor neuropathy, intellectual disability, and may also have sensory dysfunction.
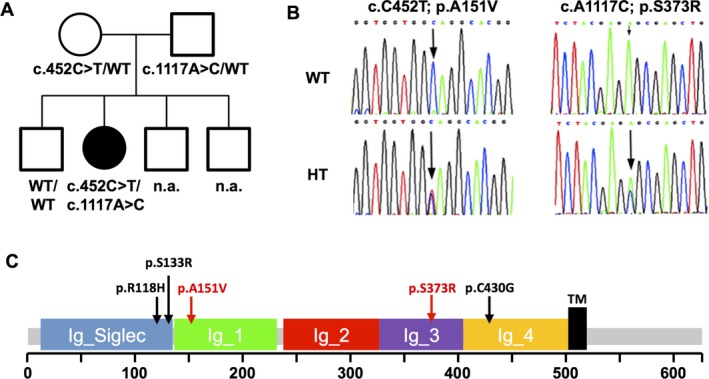
(Data source: Roubertie A, et al. Ann Clin Transl Neuro. 2019)
Application Prospects of MAG
Targeted BTK inhibitors: Zanubrutinib is a second-generation BTK inhibitor representing a new direction in treatment. Clinical cases have shown that zanubrutinib can significantly reduce antibody titers and stabilize symptoms in patients with anti-MAG neuropathy. Furthermore, a phase II clinical trial called MAZINGA is currently underway.
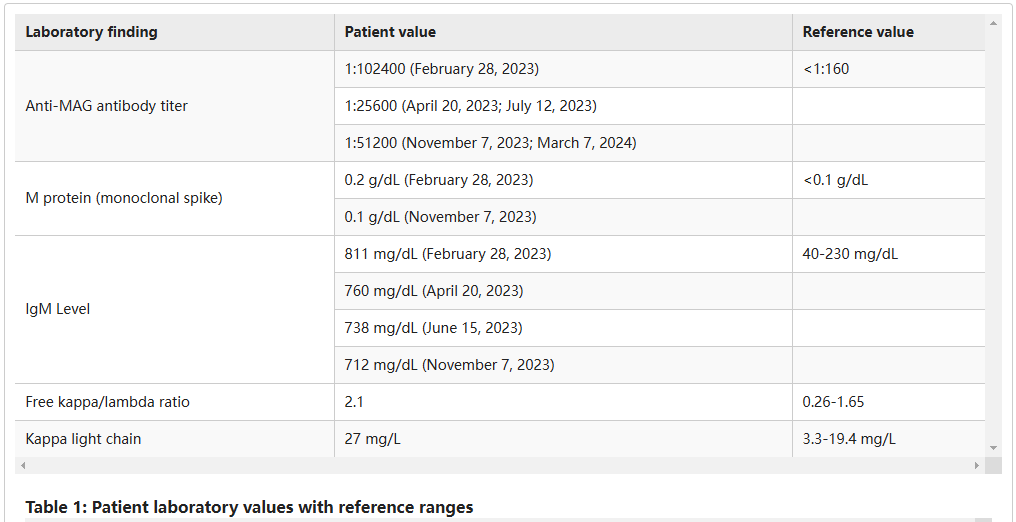
(Data source: Pein R, et al. Cureus, 2025)
