Inhibins/activins participate in regulating a variety of different functions, such as hypothalamic and pituitary hormone secretion, gonadal hormone secretion, germ cell development and maturation, erythrocyte differentiation, insulin secretion, nerve cell survival, embryonic axial development, or bone growth, depending on their subunit composition. Inhibin βA (INHBA) belongs to the transforming growth factor-β (TGF-β) superfamily, which is divided into two main functional groups: the TGF-β-like group, which includes different TGF-β subtypes (TGFB1, TGFB2, and TGFB3), INHBA, inhibin β B subunit (INHBB), inhibin β C subunit (INHBC), inhibin β E subunit (INHBE), inhibin α subunit (INHA), Nodal, and some growth differentiation factors; and the bone morphogenetic protein-like group, which includes bone morphogenetic proteins, anti-Müllerian hormone (AMH), and most growth differentiation factors. INHBA participates in the formation of activins or inhibins. The homodimer of INHBA, Activin A, has pleiotropic effects in regulating immune responses and tumor progression, including embryonic development, stem cell maintenance and differentiation, hematopoiesis, cell proliferation and tissue fibrosis.
(Data source: Abdel Mouti M, et al. Mol Ther. 2021)
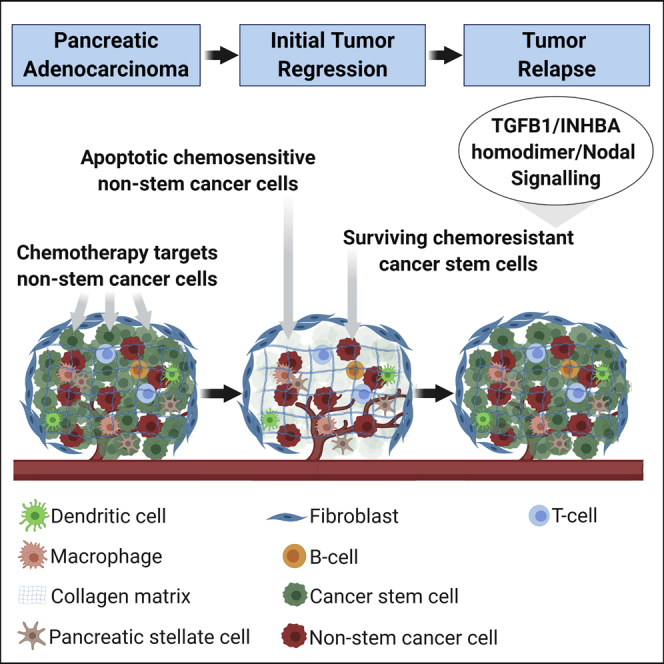
INHBA expression distribution
INHBA is mainly expressed in fibroblasts, trophoblasts, interstitial cells, and neutrophils. INHBA plays a crucial role in the development and function of the testes and regulates the growth of ovarian tissue and oocytes.
(Data source: uniprot)
INHBA expression levels are elevated in multiple cancer types, including breast cancer (BRCA) and colon adenocarcinoma (CRC). Furthermore, survival analyses have confirmed that INHBA expression is negatively correlated with prognosis in many cancer patients.
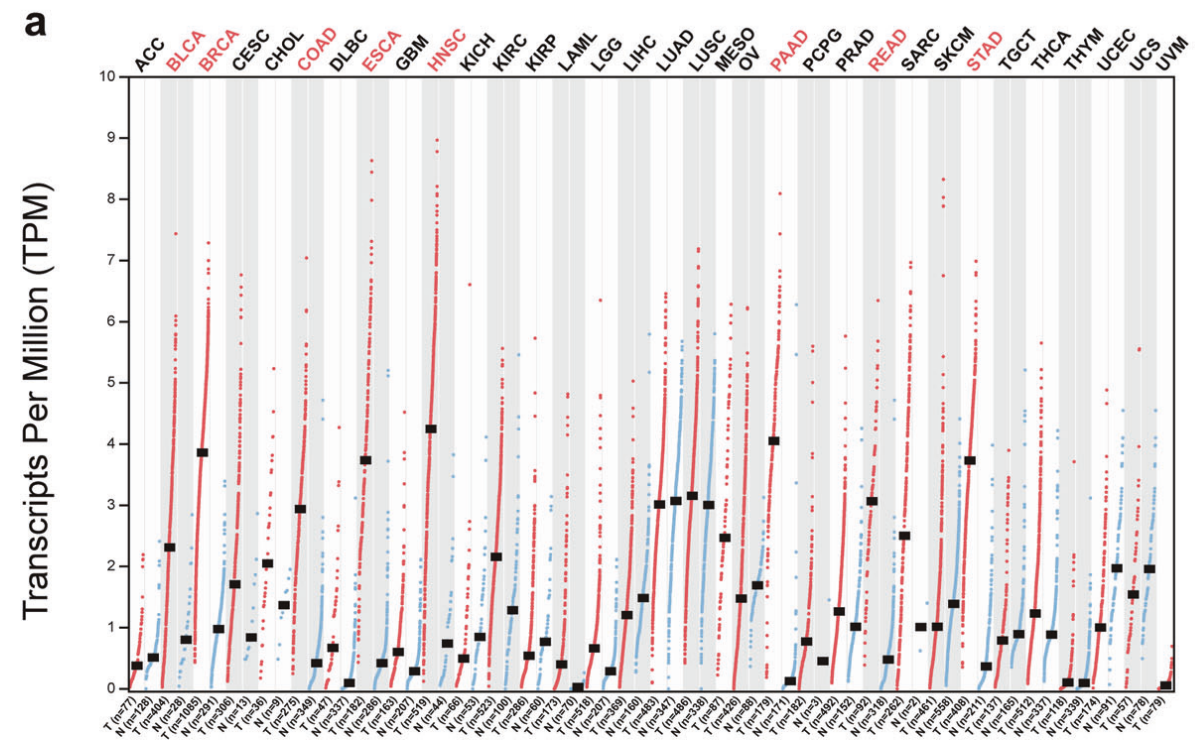
(Data source: Li FL, et al. Acta Pharmacol Sin. 2025)
INHBA Structure
INHBA is a secreted protein composed of 426 amino acids. Mature INHBA contains a highly conserved TGF-β-like domain, a hallmark functional region of the TGF-β superfamily, responsible for mediating the specific binding of ligands to their receptors and subsequent signal transduction. INHBA proteins generally exist as homodimers (i.e., activin A) or heterodimers (such as activin AB).
(Data source: Alphafold)
Signal transduction and role of INHBA in cancer
TGFB1, INHBA homodimer, and Nodal belong to the transforming growth factor superfamily of cytokines. The classical TGFB1/INHBA homodimer/Nodal signaling pathway is SMAD-dependent. TGFβ1, INHBA dimer, and Nodal proteins bind to the extracellular domains of their respective transmembrane type II receptors (TGFBR2 for TGFβ1, ACVR2A/ACVR2B for INHBA dimer/Nodal), thereby recruiting type I transmembrane receptors (TGFBR1 for TGFβ1, ACVR1B for INHBA dimer/Nodal) into the ligand-receptor complex. Receptor activation is achieved through phosphorylation of multiple serine and threonine residues within their GS domains. ZFYVE9 regulates the entry of the SMAD2/3 receptor-activating complex. The interaction between SMAD2/3 and the activating type I receptor is mediated through phosphorylated GS domains, the L45 loop, and the L3 loop of SMAD2/3, leading to phosphorylation of SMAD2/3, which subsequently dissociates from ZFYVE9 and binds to SMAD4. The active SMAD2/3/4 transcription complex then translocates to the nucleus, where it binds to DNA-binding transcription factors and core regulatory factors, as well as specific SBE sequences, thereby controlling the expression of target genes.
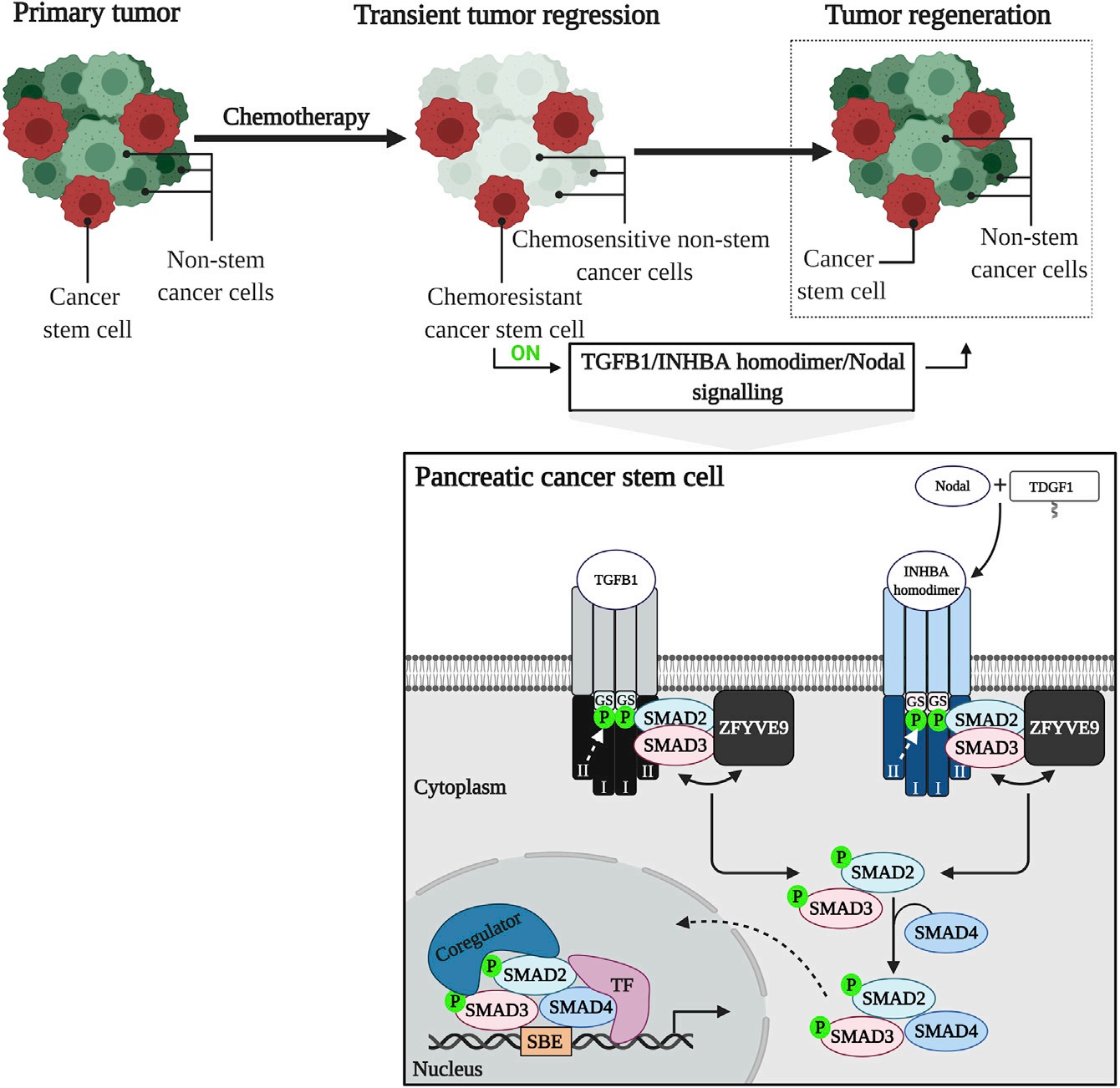
(Data source: Abdel Mouti M, et al. Mol Ther. 2021)
The stromal components in the pancreatic ductal adenocarcinoma microenvironment enhance the stem cell-like characteristics, tumorigenesis, and chemotherapeutic resistance of pancreatic ductal adenocarcinoma (PCSCs) through the TGFB1/INHBA isomer/Nodal pathway. TGFB1 secreted by tumor-associated macrophages (TAMs) and INHBA dimer/Nodal secreted by PSCs (tumor stem cells) maintain the stem cell-like characteristics of PCSCs and enhance their tumorigenesis and chemotherapeutic resistance by activating the TGFB1/INHBA dimer/Nodal signaling pathway, leading to tumor recurrence and treatment failure. The TGFB1-SMAD3 signaling pathway in PSCs also promotes chemotherapeutic resistance by regulating the synthesis and deposition of type I collagen in the extracellular matrix, leading to extensive fibrosis that hinders effective delivery of therapeutic drugs into the tumor. Upon activation by INHBA dimer/Nodal secreted by PCSCs, TAMs secrete the LL-37/hCAP-18 peptide, which drives PCSC self-renewal and promotes their tumorigenesis.
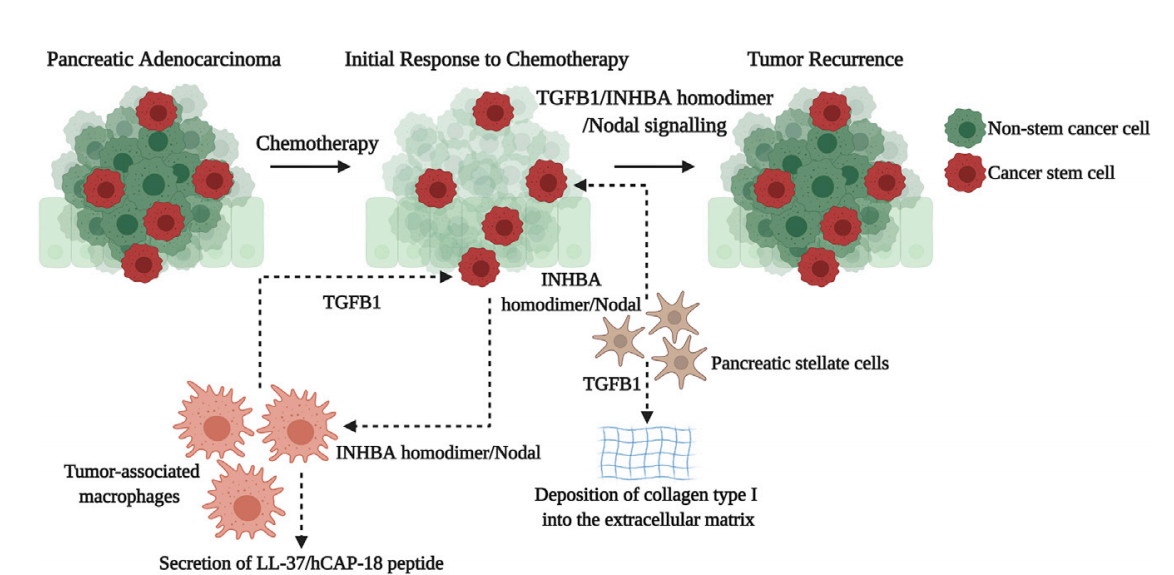
(Data source: Abdel Mouti M, et al. Mol Ther. 2021)
As a key ligand for TGF-β signaling, INHBA can activate the TGF-β pathway, initiate the phosphatidylinositol-3-kinase/protein kinase B (PI3K/Akt) pathway, increase cyclin D1 expression, and accelerate cell cycle progression, thereby driving CRC cell proliferation. Furthermore, silencing the INHBA gene can inhibit the proliferation, migration, and invasion of gastric cancer cells and osteosarcoma cells by suppressing the TGF-β signaling pathway.
The study found that INHBA, SLC25A10, and TRIM21 are interacting proteins. INHBA, acting as a scaffold protein, prevents TRIM21 from degrading SLC25A10 via K48-linked ubiquitination, thereby enhancing its stability. Furthermore, INHBA promotes the succinate/SUCNR1 pathway by enhancing SLC25A10 protein levels, inducing M2 macrophage polarization. It also activates the mtGSH/GPX4 axis, thereby inhibiting mitochondrial-dependent ferroptosis. These alterations ultimately promote the aggressive growth of colorectal cancer.
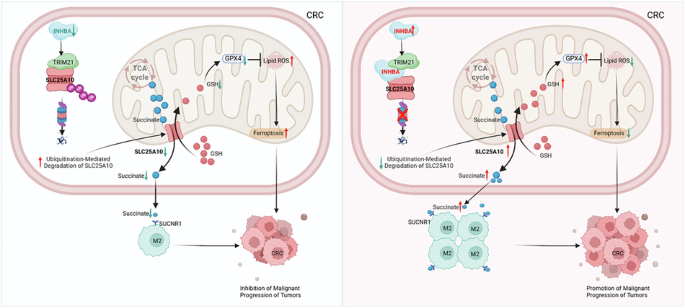
(Data source: Li W, et al. Signal Transduct Target Ther. 2025)
Targeted therapy for INHBA
Due to the upregulation of INHBA in various tumors and its important role in tumor progression, it has become a target for tumor therapy, and many antibodies targeting INHBA are in clinical development.
Garetosmab, developed by Regenereron Pharmaceuticals, is a monoclonal antibody targeting INHBA for the treatment of a rare disease by blocking Activin A. Activin A plays a crucial role in the development of heterotopic ossification (HO) lesions in patients with progressive osteoporosis (FOP). It is the only treatment proven to significantly reduce the number and volume of abnormal bone lesions (heterotopic ossification, or HO lesions) in FOP patients. The primary endpoint of the Phase 3 clinical trial in 2025 was met, with a reduction of 90% or more in new HO lesions at week 56, and Garetosmab also resulted in a reduction of more than 99% in the overall volume of new HO lesions. On February 19, 2026, Regeneron announced that the U.S. Food and Drug Administration (FDA) had accepted its Biologics License Application (BLA) for Garetosmab for the treatment of adults with progressive FOP for priority review.
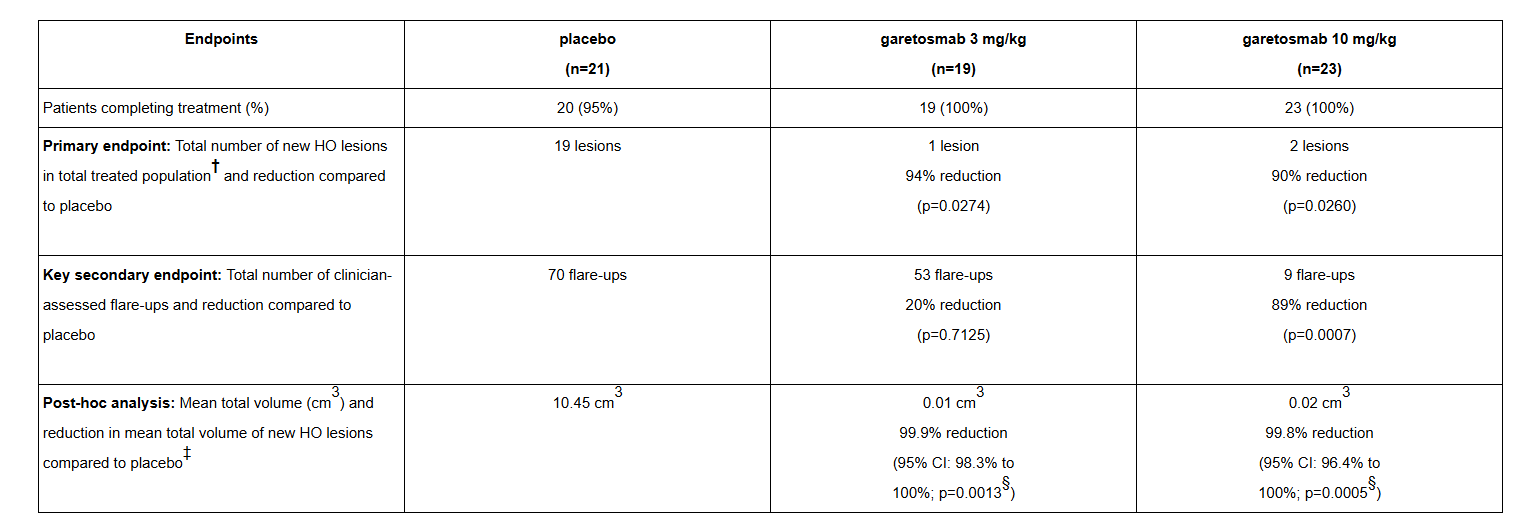
(Data source: Regenereron Pharmaceuticals official website)
Sotatercept, developed by Merck & Co. (Rawley, New Jersey, USA), is an activin signaling inhibitor for the treatment of pulmonary arterial hypertension (PAH) and received its first approval in the United States on March 26, 2024. Sotatercept is a homodimeric recombinant fusion protein consisting of an extracellular domain of human activin receptor type IIA linked to the Fc domain of human immunoglobulin G1. It regulates angiogenesis by binding to activin A ( and other transforming growth factor-β superfamily ligands), inhibiting activin signaling through pro-proliferative and anti-proliferative signaling pathways (mediated by ActRIIA/Smad2/3 and BMPRII/Smad1/5/8, respectively).
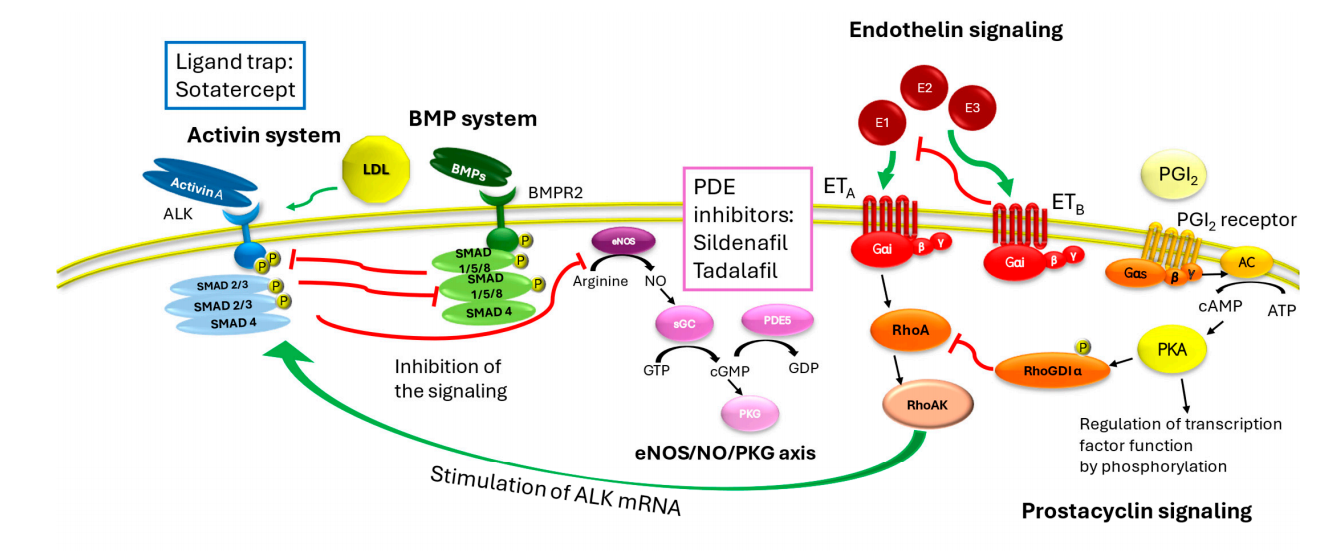
(Data source: Madonna R , et al. Int J Mol Sci. 2025)
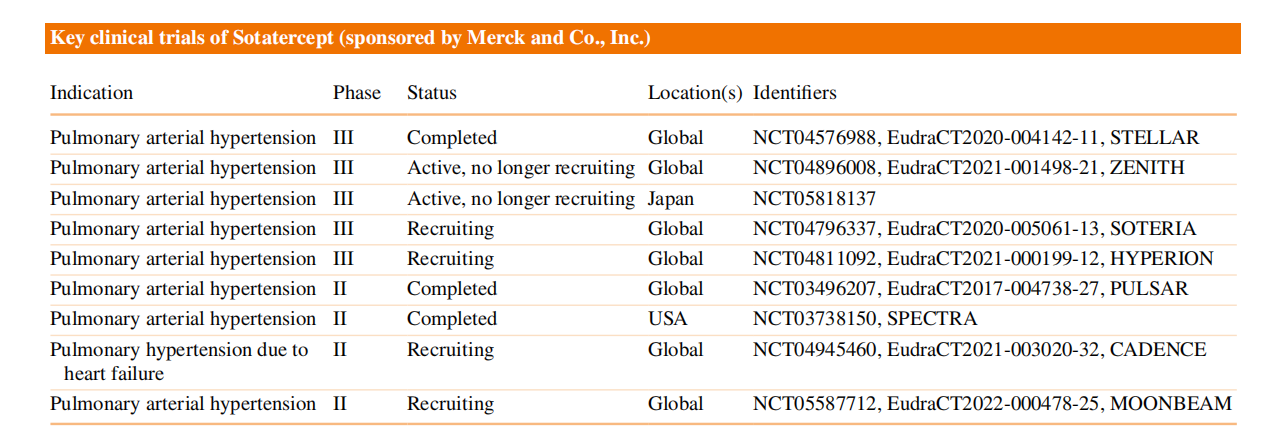
(Data source: Kang C. Drugs. 2024)
