Epidermal growth-like protein 7 (EGFL7) regulates vascular tubular formation in vivo. It inhibits platelet-derived growth factor (PDGF)-BB-induced smooth muscle cell migration and promotes endothelial cell adhesion to the extracellular matrix and angiogenesis. EGFL7 is also associated with various diseases, including preeclampsia, stroke, multiple sclerosis (MS), and cancer.
EGFL7 expression distribution
EGFL7 is mainly expressed in adipocytes, endothelial cells, lymphatic endothelial cells, Hofbauer cells, and spermatogonia. During the embryonic period, EGFL7 is highly expressed in actively proliferating vascular endothelial cells, but its expression is significantly limited in healthy adult tissues, with only a few organs such as the lungs maintaining high levels. However, it can be reactivated under specific pathological conditions (especially cancer and injury repair).

(Data source: uniprot)
The structure of EGFL7
EGFL7, located on human chromosome 9, is a secreted protein with a molecular weight of approximately 30 kDa. It contains multiple domains, including a signal peptide, an Emilin-like domain (EMI) that interacts with extracellular matrix proteins, and two EGF-like domains. The first EGF -like domain contains the RGD motif, which can bind to integrins (such as αvβ3); the second EGF-like domain can bind calcium ions, enhancing protein stability. It also has a C-terminal coiled-coil domain , which may be involved in protein oligomerization.
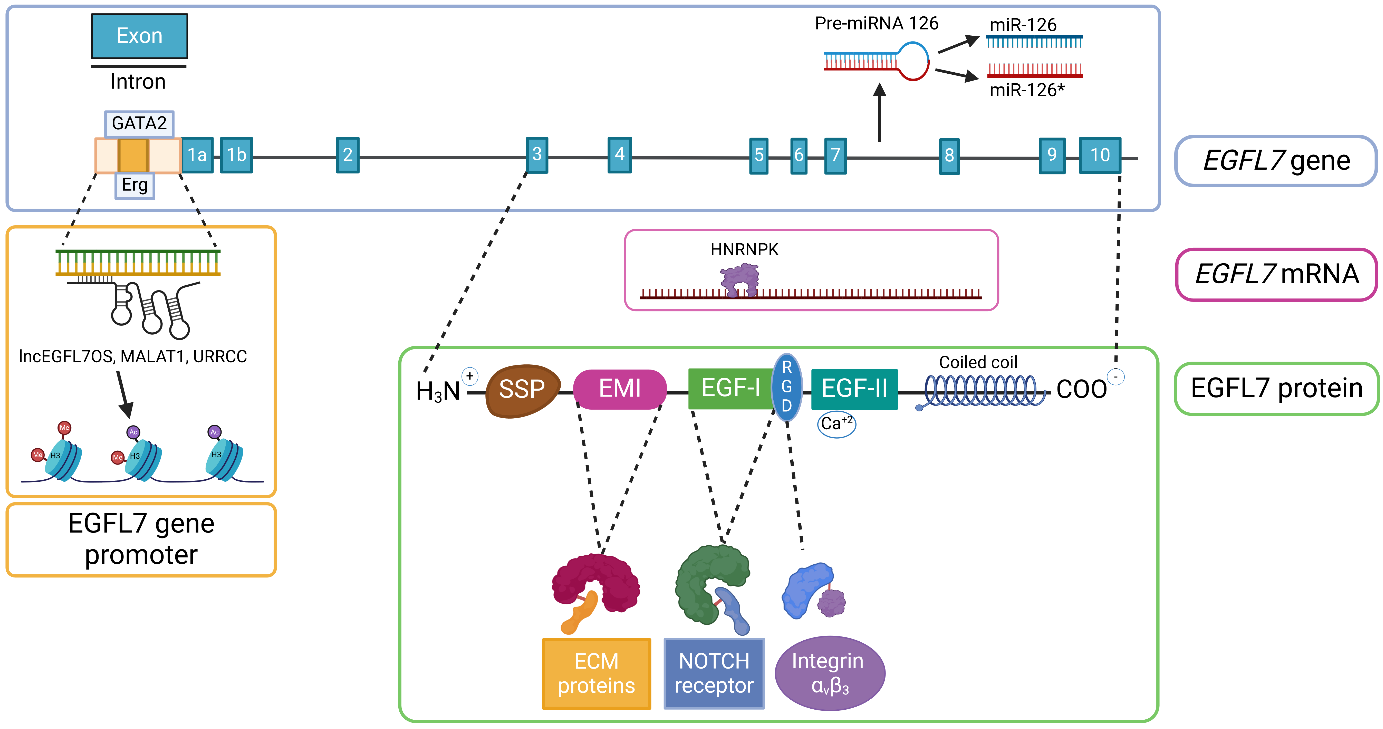
(Data source: Fabian C, et al. Pharmacol Ther. 2025)
Regulation of EGFL7 in the disease
EGFL7 expression is tightly regulated by numerous factors through multiple mechanisms. Disruptions in these regulatory mechanisms can lead to abnormal protein expression, potentially triggering various pathological conditions. Alterations in EGFL7 expression have been identified as a pathogenic factor in a variety of diseases, including preeclampsia, stroke, multiple sclerosis, and various tumor types.
In endothelial cells, EGFL7 inhibits the degradation of the nuclear factor κB light chain gene enhancer in B cell repressor α (IκBα), thereby blocking nuclear factor κB (NF-κB) signaling. This hinders the activation of immune response genes and prevents inflammation. During inflammation, TNFα expression suppresses EGFL7, thereby triggering the activation of the immune response.
EGFL7 is expressed at relatively high levels in healthy brains, which prevents T cells from infiltrating the brain parenchyma. Therefore, T cells preferentially remain in the perivascular space.
In the MS-affected brain, low levels of EGFL7 expression lead to disruption of the brain's white blood cell barrier (BBB). This results in the infiltration of CD4- positive T cells into the brain parenchyma. The T cells interact with and activate microglia, the brain's resident immune cells. Microglia release cytokines, causing neuronal demyelination and axonal degeneration.
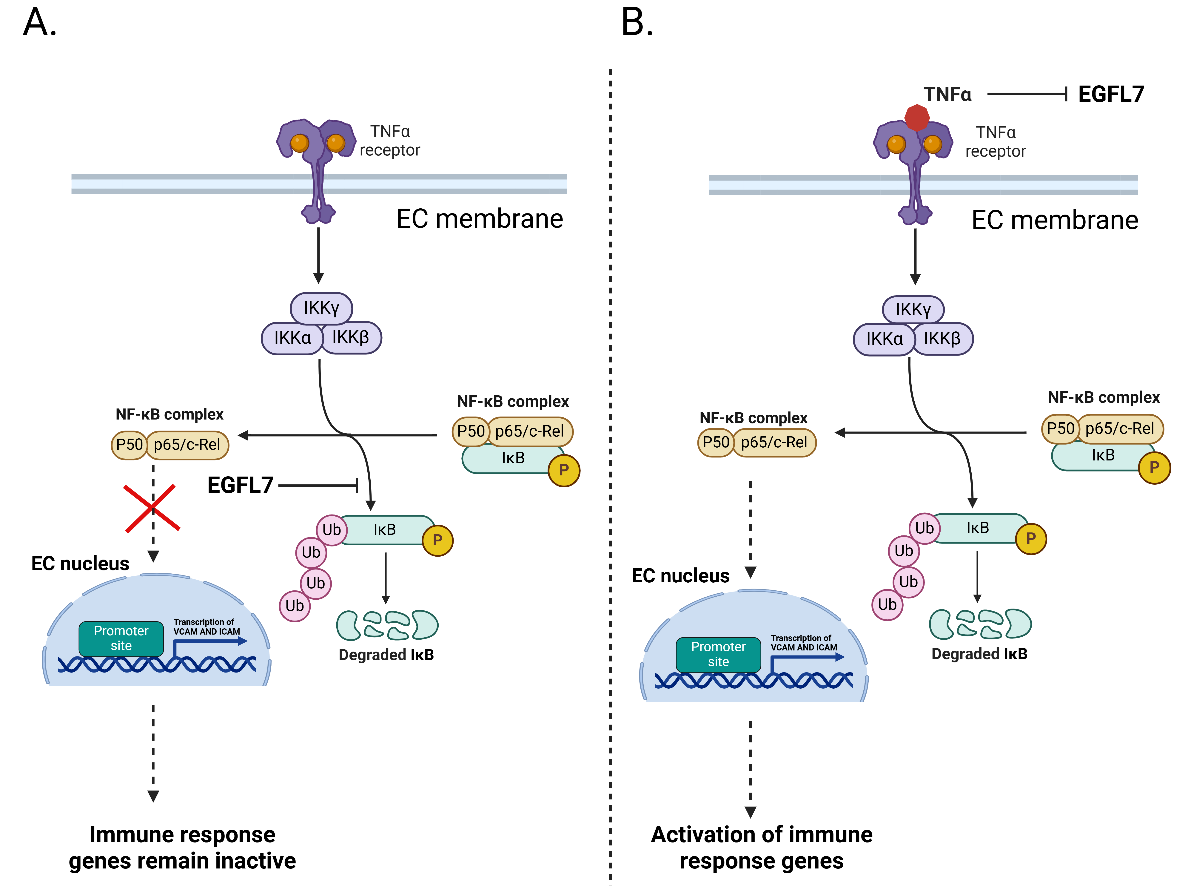
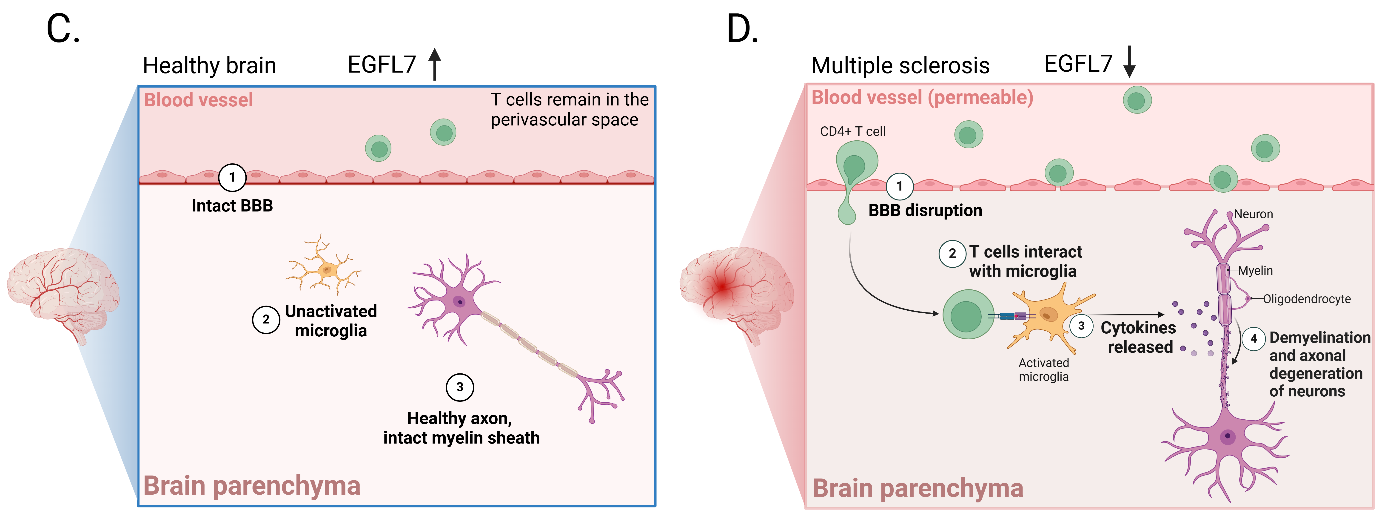
(Data source) Fabian C, et al. Pharmacol Ther. 2025)
The role of EGFL7 in cancer
Upregulation of EGFL7 expression and its involvement in angiogenesis and immune escape may promote accelerated tumor progression.
In gliomas, tumor-secreted EGFL7 promotes angiogenesis by binding to integrin α5β1, thereby stimulating tumor growth. EGFL7 also participates in immune escape in breast and lung cancer by downregulating adhesion molecules ICAM-1 and VCAM-1 on endothelial cells, reducing immune cell adhesion . In multiple myeloma, EGFL7 forms a positive feedback loop with ITGB3 and KLF2, promoting tumor cell survival.
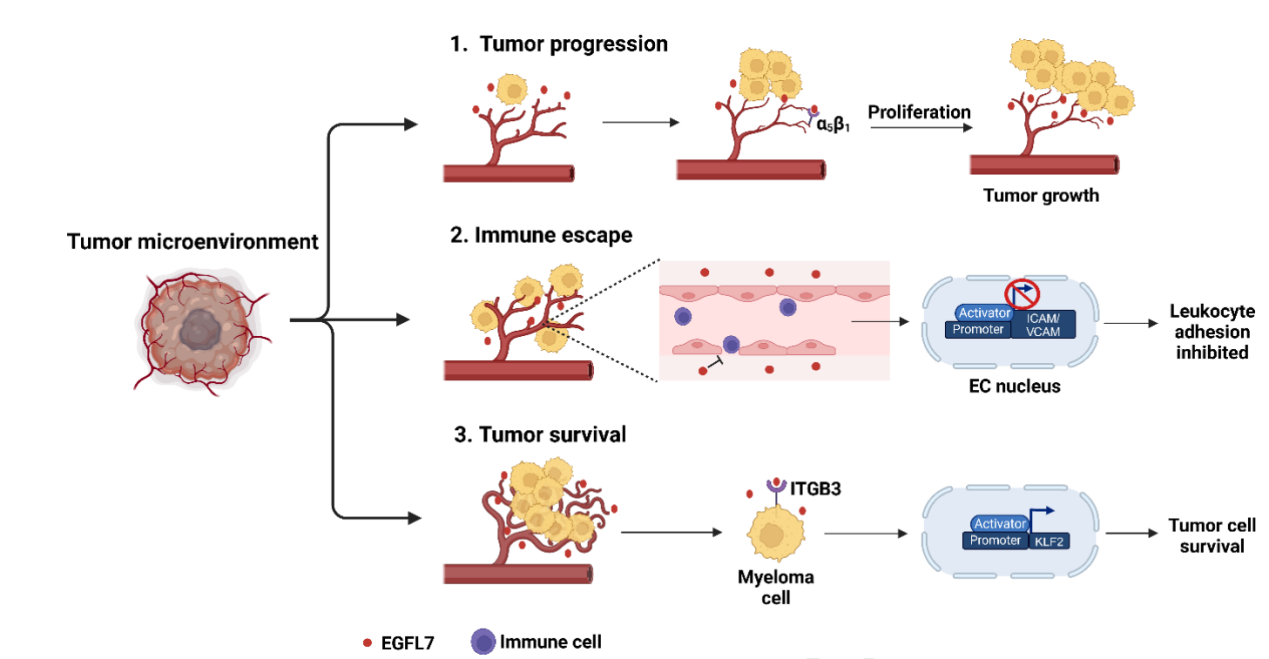
(Data source: Fabian C, et al. Pharmacol Ther. 2025)
Targeted therapy for EGFL7
The therapeutic potential of EGFL7 in disease: Physiologically, EGFL7 regulates various biological processes, such as angiogenesis, neurogenesis, and neurogenesis. In pathological states, the use of EGFL7 antagonists in combination with anti-αvβ3 therapy may inhibit tumor growth and alleviate stroke symptoms. Treatment of preeclampsia with EGFL7 agonists, while simultaneously treating MS with TNFα antagonists, may be beneficial for patients with these diseases.
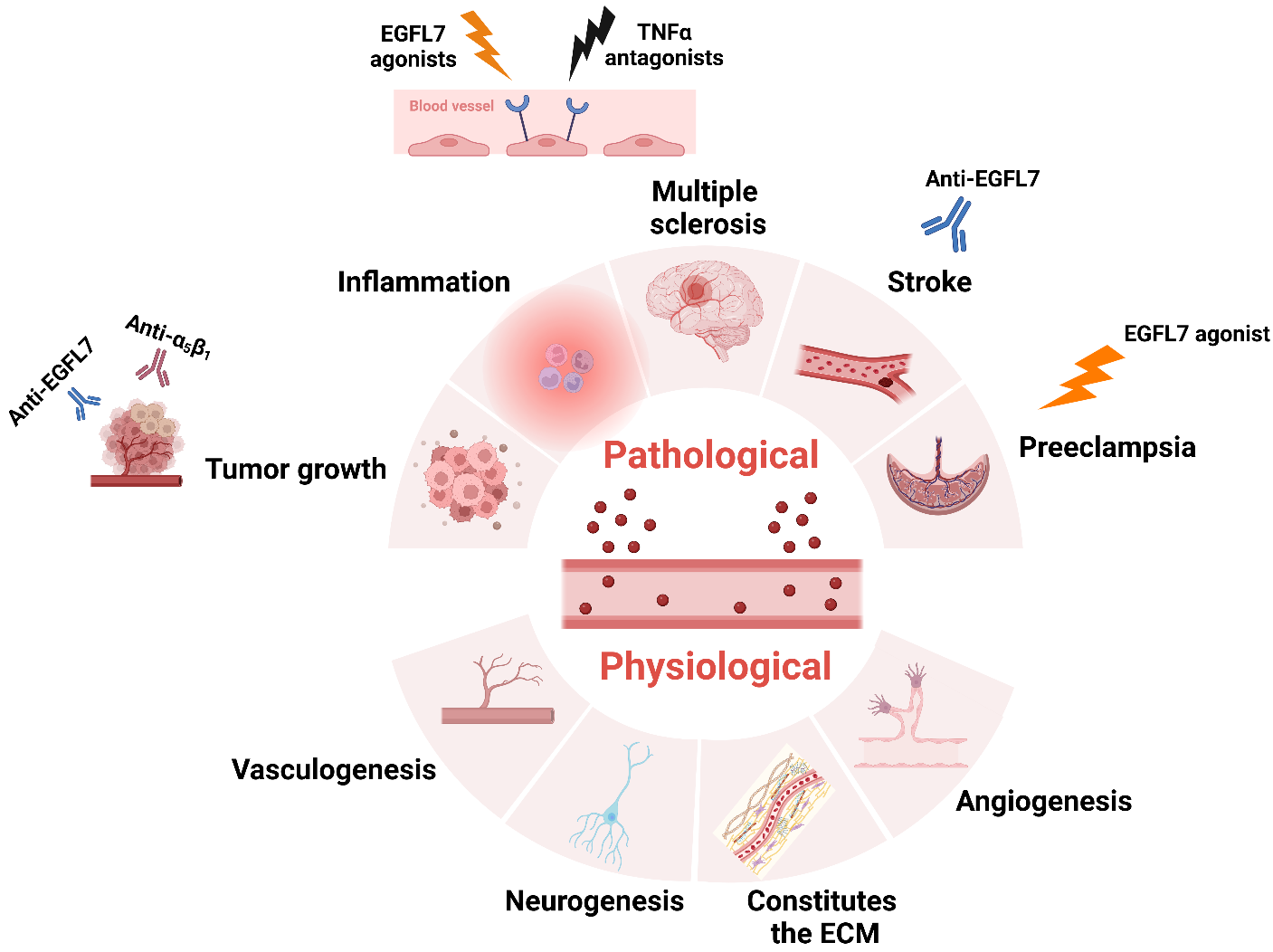
(Data source: Fabian C, et al. Pharmacol Ther. 2025)
Parsatuzumab is an anti-EGFL7 antibody that selectively blocks the interaction between EGFL7 and endothelial cells . A phase II randomized clinical trial showed that in patients with metastatic colorectal cancer, the combination of parsatuzumab and FOLFOX + bevacizumab failed to improve efficacy. Since parsatuzumab did not demonstrate the expected efficacy in the phase II clinical trial , there are currently no further research updates on its efficacy.
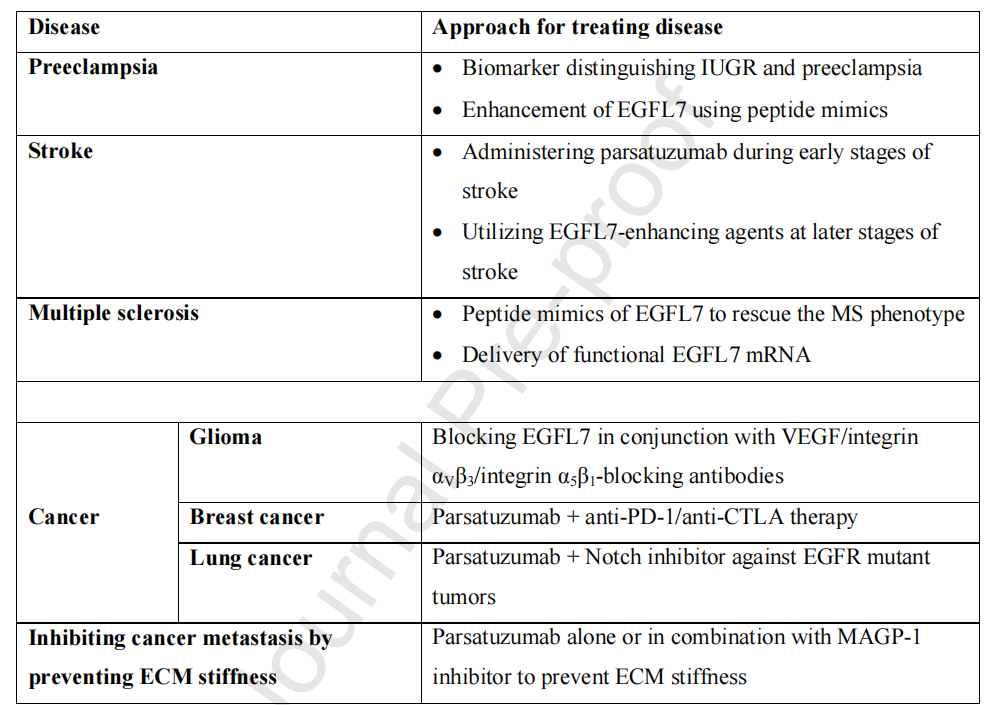
(Data source: Fabian C, et al. Pharmacol Ther. 2025)
