Mucins are structurally classified into four classes: membrane-restricted mucins containing tandem repeat (TR) sequences, cysteine-rich secretory mucins (containing TR sequences), cysteine-deficient secretory mucins (containing TR sequences), and mucins without TR sequences. MUC5A is a cysteine-rich secretory mucin, a gel-like glycoprotein of the gastric and respiratory epithelium. This mucin imparts the characteristic viscoelasticity to mucus, a property crucial for ciliated cells to clear exogenous substances. MUC5A signal transduction is regulated by upstream factors such as inflammatory cytokines and growth factors, influencing downstream effector molecules such as cell proliferation, apoptosis, migration, and invasion. In terms of disease, it is closely related to various diseases, including asthma, chronic obstructive pulmonary disease, gastric cancer, and enteritis. Targeted therapies against MUC5A, including small molecule inhibitors and gene therapy strategies, show potential application prospects.

(Data source: Li J, et al. Molecules. 2020)
(Data source: Arai J, et al. Gastroenterology. 2024)
MUC5A expression distribution
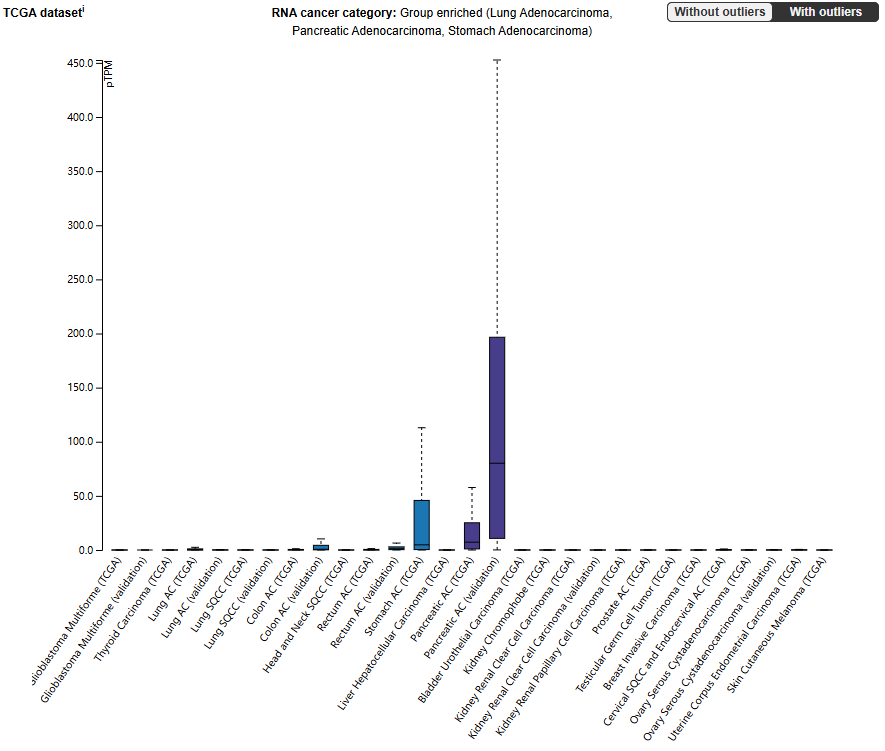
MUC5A expression is tissue-specific, with high expression in respiratory tract surface mucosal cells and gastric epithelial cells. It is overexpressed in many carcinomas, such as lung adenocarcinoma, pancreatic adenocarcinoma, and gastric adenocarcinoma. In airway epithelial cells, expression is significantly increased (at the protein level) during cell differentiation.
MUC5AC may be an important biomarker because this protein is expressed in fetal and precancerous colonic mucosa, but at a lower level in normal colon. Overexpression of the MUC5AC antigen is associated with pancreatic and colorectal cancer. MUC5AC is expressed in colon cancer in an aberrantly glycosylated form.

(Data source: uniprot)
MUC5A Structure
MUC5A, composed of 5654 amino acids, belongs to the gel-forming type of secretory mucins. The N-terminal VWD1 and VWD2 domains of MUC5A interact with the C8 domain of adjacent molecules to assemble into "bead-like" units, which then form linear fibers, forming the basis of the mucogel network. The VWD3 domain can drive the formation of a network polymer by forming covalent dimers and further assembling into tetramers.
The central region is a proline-, threonine-, and serine-rich PTS domain, which is highly O-glycosylated to form a rigid "rod-like" structure. The PTS region of MUC5A is separated by multiple cysteine-rich CysD domains (nine in total, CysD1 to CysD9). These domains participate in isomorphic interactions, finely regulating the mucus structure.
The high polymorphism of the MUC5AC gene leads to individual differences in the copy number of its domains, affecting protein length, which may result in differences in mucus barrier properties between individuals.

(Data source: Haberman M, et al. Proc Natl Acad Sci US A. 2025)
The role of MUC5A in disease
The role of MUC5A in chronic obstructive pulmonary disease
Chronic obstructive pulmonary disease (COPD) has a high morbidity and mortality rate worldwide. COPD is also considered a mucoobulsive disease, accompanied by increased mucin content, particularly MUC5AC, which may uniquely alter the properties of mucus. MUC5AC can lead to severe mucoobulsive lung disease and exacerbate the pathogenesis of COPD. Abnormal production and secretion of MUC5AC involve multiple pathways.
In chronic obstructive pulmonary disease (COPD), extracellular signaling factors, such as tumor necrosis factor-α (TNF-α), bind to cell surface receptors, triggering a series of downstream reactions. Receptor stimulation triggers the activation of IκB kinase (IKK). IKK phosphorylates serine residues at the regulatory site of the IκB subunit of intracellular NF-κB, causing ubiquitination and degradation of the IκB subunit. This releases the NF-κB complex, and the free NF-κB then translocates to the nucleus, where it drives the expression of target genes, including MUC5AC.
The IL-13-STAT6-SPDEF-MUC5AC signaling pathway is a major signaling pathway driving the differentiation of epithelial cells into goblet cells. One study found that knocking down SPDEF reduced IL-13-induced MUC5AC expression in human airway epithelial cells.

(Data source: Li J, et al. Molecules. 2020)
The role of MUC5A in cancer
MUC5A plays a crucial role in the carcinogenesis and drug sensitivity of pancreatic ductal adenocarcinoma (PDAC). In normal pancreatic cells, MUC5AC production is suppressed but restarted upon appropriate stimulation. The composition of MUC5AC, and consequently its distribution and production levels, can vary depending on the nature of the stimulus. If the stimulus has a potentially malignant characteristic, higher levels of mature MUC5AC are produced, accompanied by aberrant arrangement. Invasive tumors with poor treatment response and worse prognosis tend to have fewer mature MUC5ACs in their apical and intercellular regions compared to benign tumors. If the stimulus leads to a precancerous or benign state, more immature MUC5ACs are produced, and their arrangement is controlled. During malignant transformation, the composition and distribution of MUC5AC change.

(Data source: Manne A, et al. Int J Mol Sci. 2023)
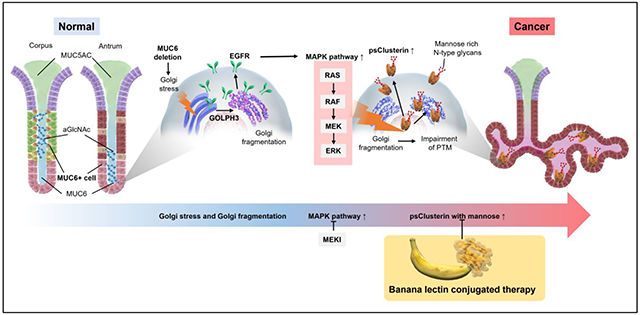
Gastric cancer (GC) is a leading cause of cancer-related death worldwide and exhibits multiple phenotypic variations, classifying it into gastric and intestinal types based on mucin expression. Immunohistochemical markers such as MUC5AC and MUC6 (representing the gastric type), MUC2 and CD10 (representing the intestinal type) help classify gastric cancer cases into four subtypes: gastric, intestinal, mixed gastrointestinal, and a type that does not conform to either the gastric or intestinal type. Early gastric cancer typically presents with a gastric mucin phenotype. As the tumor progresses, the clear transition from the gastric mucin phenotype to the intestinal mucin phenotype becomes more pronounced. Alterations in mucin and mucin-associated sugars can potentially influence carcinogenesis in various cancer types beyond GC. Loss of MUC6 triggers a Golgi stress response and upregulates GOLPH3 expression, leading to EGFP/MAPK activation. Loss of αGlcNAc results in phosphorylation of the C-terminus of MUC1, thereby promoting the binding of Src and β-catenin to this C-terminus induced by galactoglucan-3. Deficiency of O-glycosyl groups in the gastric epithelium leads to activation of caspases 1 and 11-dependent inflammasomes, resulting in the progression of gastritis and gastric cancer. This also enhances cancer-related function of CD44 through RON pathway activation or the Wnt pathway, while simultaneously activating EGFR, ErbB2, SRPX2, and RUNX1. Loss of MUC5AC results in increased expression of Tff1, Tff2, and Muc1, while decreasing expression of Tnf and Il17a. Loss of MUC2 leads to defects in intercellular connections, mitochondrial damage, and ATP depletion in the intestinal epithelium.

(Data source: Arai J, et al. Cancer Sci. 2024)
MUC5A -targeted therapy
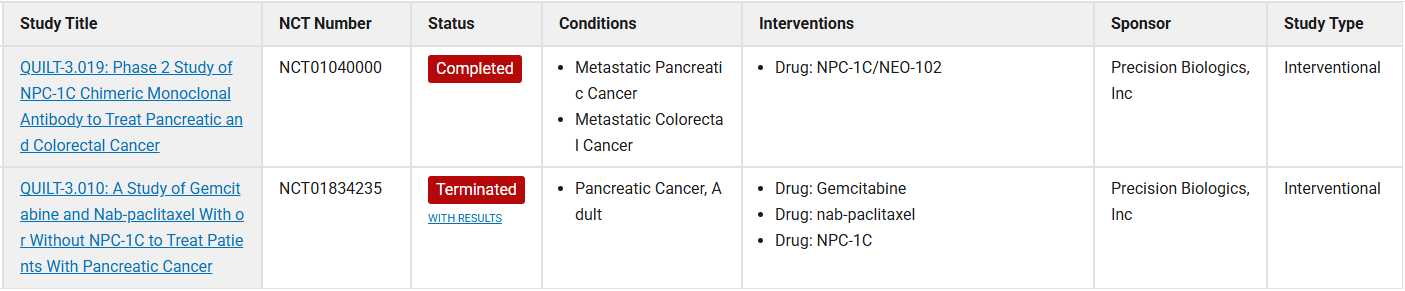
Ensituximab (NEO-101) is a novel chimeric antibody targeting MUC5A for the treatment of colorectal cancer. Ensituximab can distinguish between aberrantly glycosylated forms and native MUC5AC. Preclinical data indicate that Ensituximab targets only MUC5AC-positive tumors and does not damage the MUC5AC antigen in non-malignant tissues. A single-arm, phase II trial evaluated the efficacy and safety of Ensituximab in patients with advanced, refractory cancers expressing the MUC5AC antigen. Ensituximab was administered intravenously at the recommended phase II dose (RP2D) of 3 mg/kg every 2 weeks. Ensituximab was well tolerated in patients with colorectal cancer and showed some antitumor activity.
(Data source: clinicaltrials)

(Data source: Kim RD, et al. Clin Cancer Res. 2020)
