The GDNF family has four members: GFRα1, GFRα2, GFRα3, and GFRα4. They are anchored to the cell membrane via extracellular vesicle anchoring structures containing extracellular vesicle-binding domains (CRDs). GDNF family ligands (GFLs) have four characteristic ligands: GDNF, NRTN, ARTN, and PSPN. Two GFL monomers form an intertwined homodimer that binds to the corresponding GFRα receptor binder. GFRA3 is the GDNF family receptor α-3, which primarily binds to the retinoblastoma protein receptor (ARTN), a growth factor that supports the survival of sensory and sympathetic peripheral neurons. ARTN binding leads to autophosphorylation and activation of the RET receptor. GFRA3 has become a highly promising therapeutic target due to its mediating neuropathic pain and potential role in tumor progression. Currently, research targeting this target mainly focuses on developing antibody drugs to block pain signals or exploring its application in targeted cancer therapy.
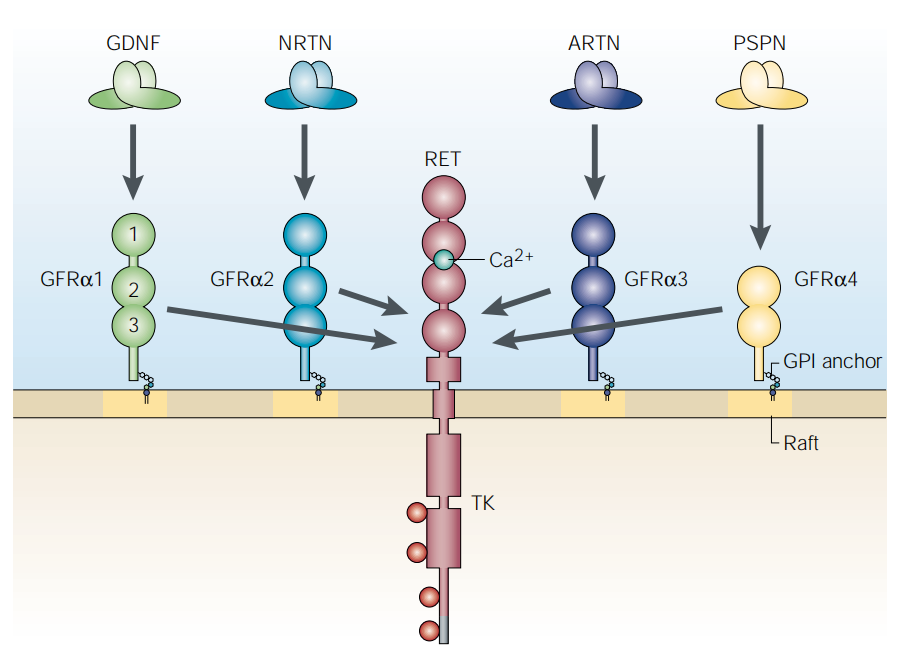
(Data source: Airaksinen MS, et al. Nat Rev Neurosci. 2002)
GFRA3 expression distribution
GFRA3 is primarily expressed in glial cells, enteroendocrine cells, oocytes, and spermatogenic cell types. It is widely expressed in adults and fetuses, exhibiting a similar pattern. It is essentially not expressed in the central nervous system, but is highly expressed in several sensory and sympathetic ganglia of the peripheral nervous system. It shows moderate expression in many non-nervous tissues, particularly the digestive and genitourinary systems, but high expression is found in the stomach and appendix. Low expression is observed in several types of glandular tissue. Extremely low or no expression was detected in the hematopoietic system.
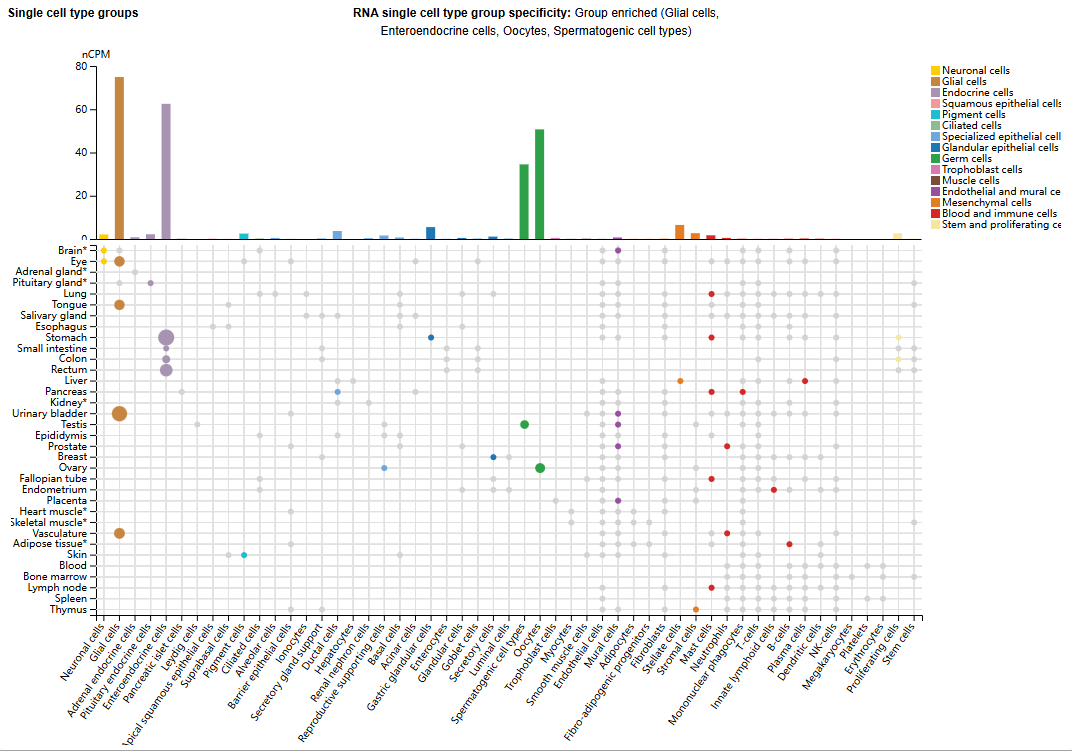
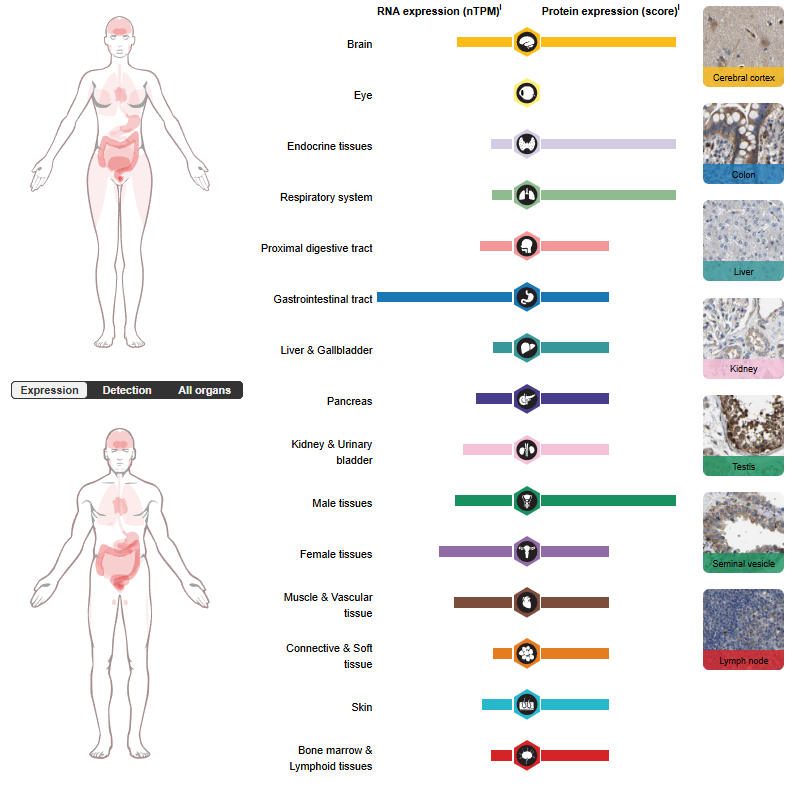
(Data source: uniprot)
Structure and signal transduction regulation of GFRA3
GFRA3 is a glycosylphosphatidylinositol (GPI)-anchored cell surface protein composed of 400 amino acids. It mainly consists of a signal peptide, an extracellular domain, and a GPI-anchored structure. The extracellular domain contains two CUB domains and one GFRα-specific domain, responsible for ligand recognition. The GPI-anchored structure is a non-transmembrane domain that is anchored to the outer side of the cell membrane via GPI, restricting its mobility across the membrane.
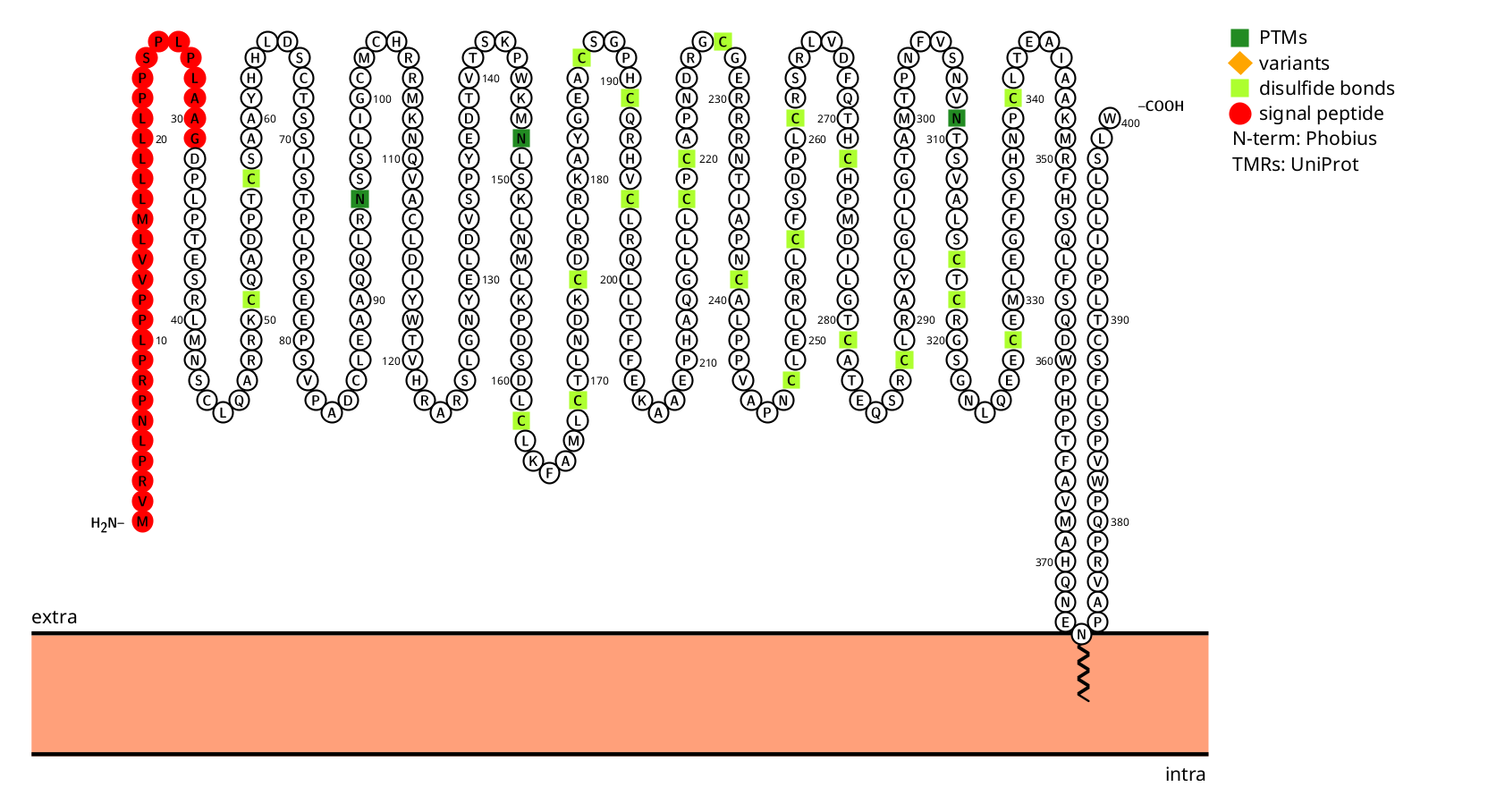
(Data source: protest)
Following the binding of GFL and GFRα, the complex binds to RET (a transmembrane tyrosine kinase receptor), forming the GFL-GFRα-RET ternary complex. GFRA3 signaling relies on a ligand-receptor-co-receptor ternary complex model. RET-dependent GFRα signaling is activated by phosphorylation of GFRα on multiple intracellular tyrosine residues. Two signal transduction pathways contribute to GFL-induced RET activation: the Ras/MAPK and PI3K/Akt signaling pathways: membrane-bound GFRα (cis signaling) and soluble GFRα molecules released from nearby cells (trans signaling). The presence of GDNF promotes the binding of CFRα to NCAM, thereby activating the NCAM-mediated Fyn-FAK-MAPK signaling pathway.
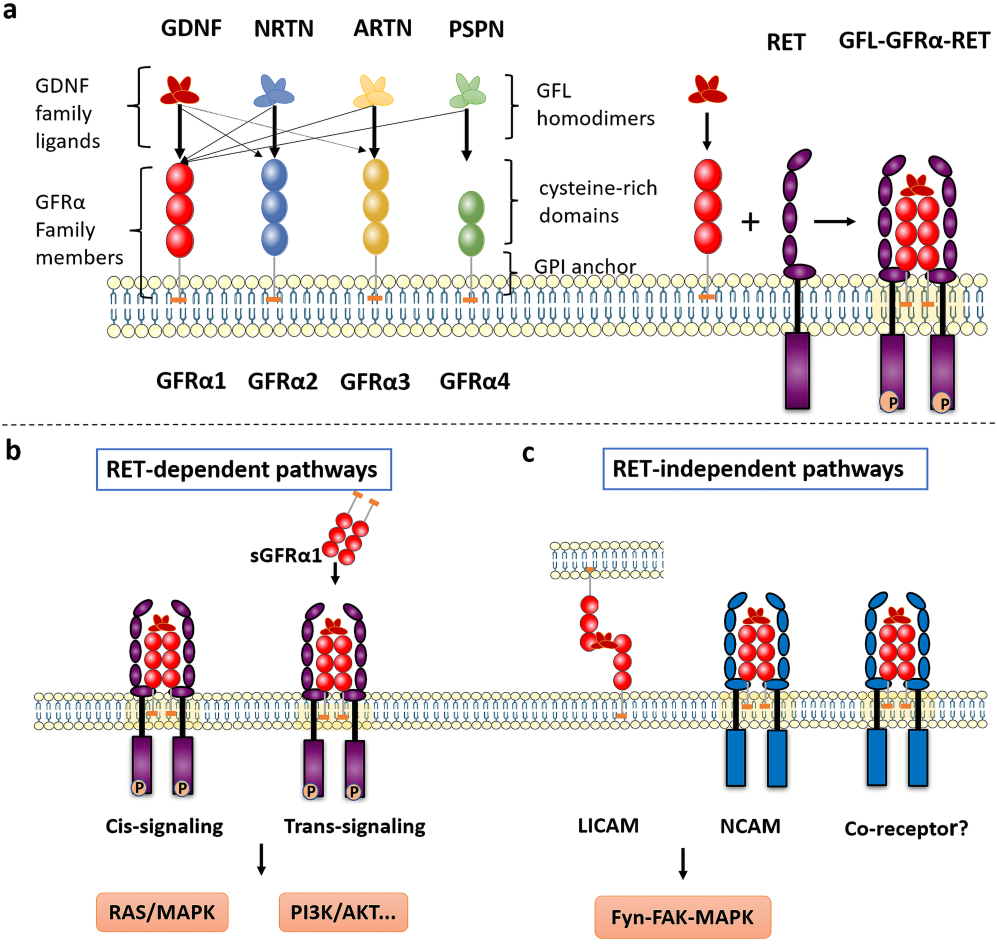
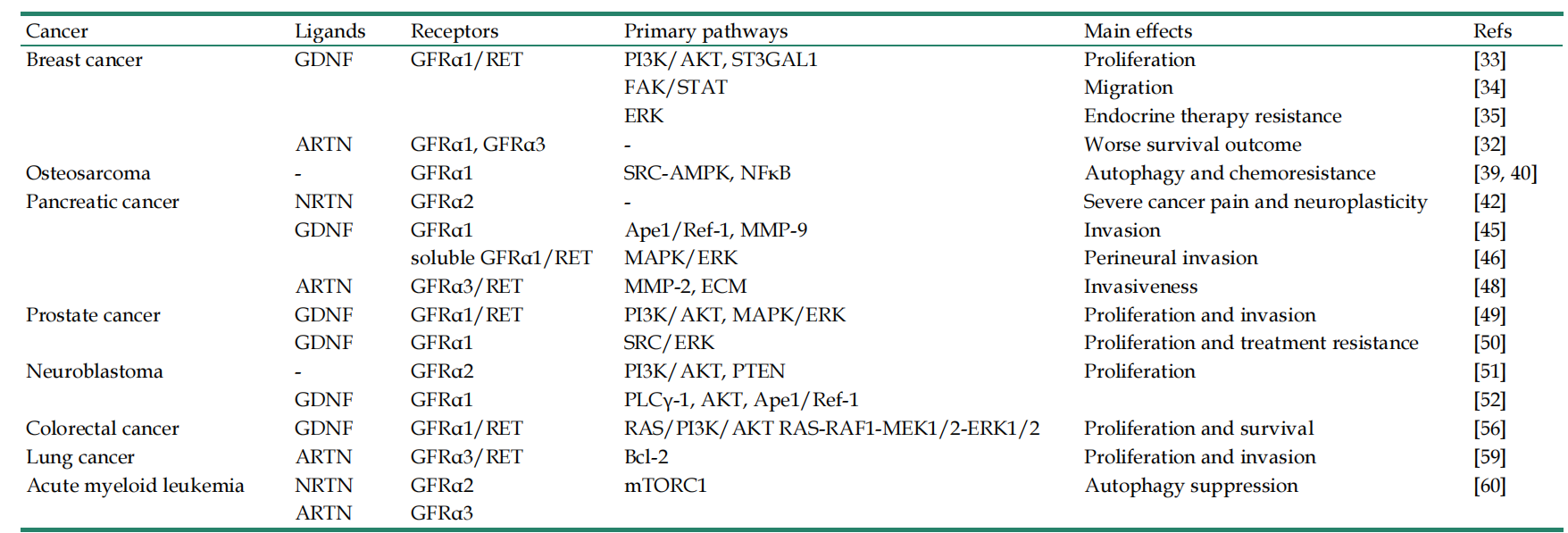
(Data source: Li Q, et al. Int J Med Sci. 2022)
The role of GFRA3 in pain
GFRα3 plays a crucial role in persistent pain, including musculoskeletal pain, and is therefore a potential therapeutic target for developing novel, clinically effective musculoskeletal pain analgesics. GFRα3 is a key receptor for Artemin (ARTN)-mediated chronic musculoskeletal pain, particularly osteoarthritis (OA) and inflammatory bone pain. Artemin/GFRα3 signaling enhances the expression and activity of TRPV1/TRPA1 ion channels by activating RET receptors and downstream pathways such as MAPK and PI3K/AKT, mediating thermal hyperalgesia, cold anomalous pain, and mechanosensitive responses. In animal models of osteoarthritis and inflammatory bone pain, blocking Artemin or GFRα3 reduces the sensitivity of Aδ and C fibers to mechanical stress, reverses abnormal weight-bearing behavior and thermal/cold hyperalgesia, suggesting that this signaling axis plays a key driving role in chronic musculoskeletal pain. Artemin can enhance neuronal excitability and promote pain persistence by upregulating the expression of TRP channels (such as TRPV1 and TRPA1). GFRα3-mediated signaling exhibits a dual effect in neuropathic pain: on the one hand, it promotes nerve regeneration and repair (e.g., in clinical studies of neublastin), and on the other hand, it participates in the maintenance of cold abnormal pain (e.g., oxaliplatin-induced neuropathy). Targeting this receptor requires balancing analgesia and neuroprotection.
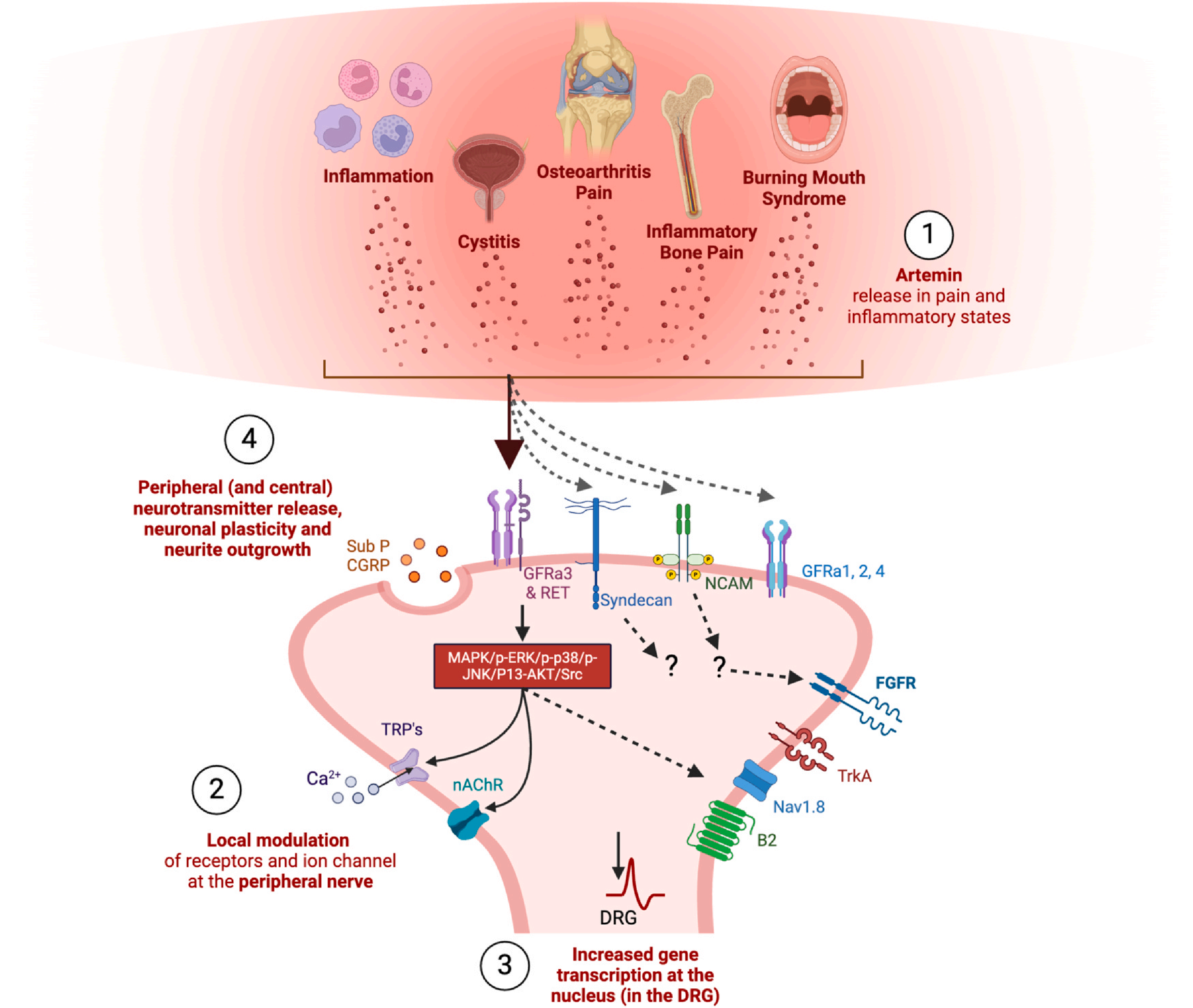
(Data source: Gupta A, et al. Osteoarthritis Cartilage. 2025)
Targeted therapy for GFRA3
Nadecnemab (REGN5069) is a monoclonal antibody targeting GFRA3, developed by Regeneron for the treatment of pain. Its highest development stage was Phase 2 clinical trials, but these have since been terminated.
