Background
Antibodies with high affinity and specificity, obtained through high-throughput screening and rapid production, are crucial to the contemporary biomedical industry. Traditional hybridoma strategies for antibody production face challenges such as low efficiency, long production cycles, large batch variability, and high labor intensity. Advances in molecular biology and gene editing have revolutionized antibody production. New high-throughput technologies such as antibody library display, single B cell antibody technology, and single-cell sequencing have significantly reduced antibody R&D costs and improved efficiency. These innovations have accelerated the commercial application of antibodies and met the evolving needs of the biopharmaceutical industry.
On May 8, 2025, an article titled "High-throughput strategies for monoclonal antibody screening: advances and challenges" was published in J Biol Eng. This article reviewed the research progress of high-throughput antibody development in recent years, emphasized its potential advantages over traditional methods, and looked forward to its development prospects.

Antibody library display technology
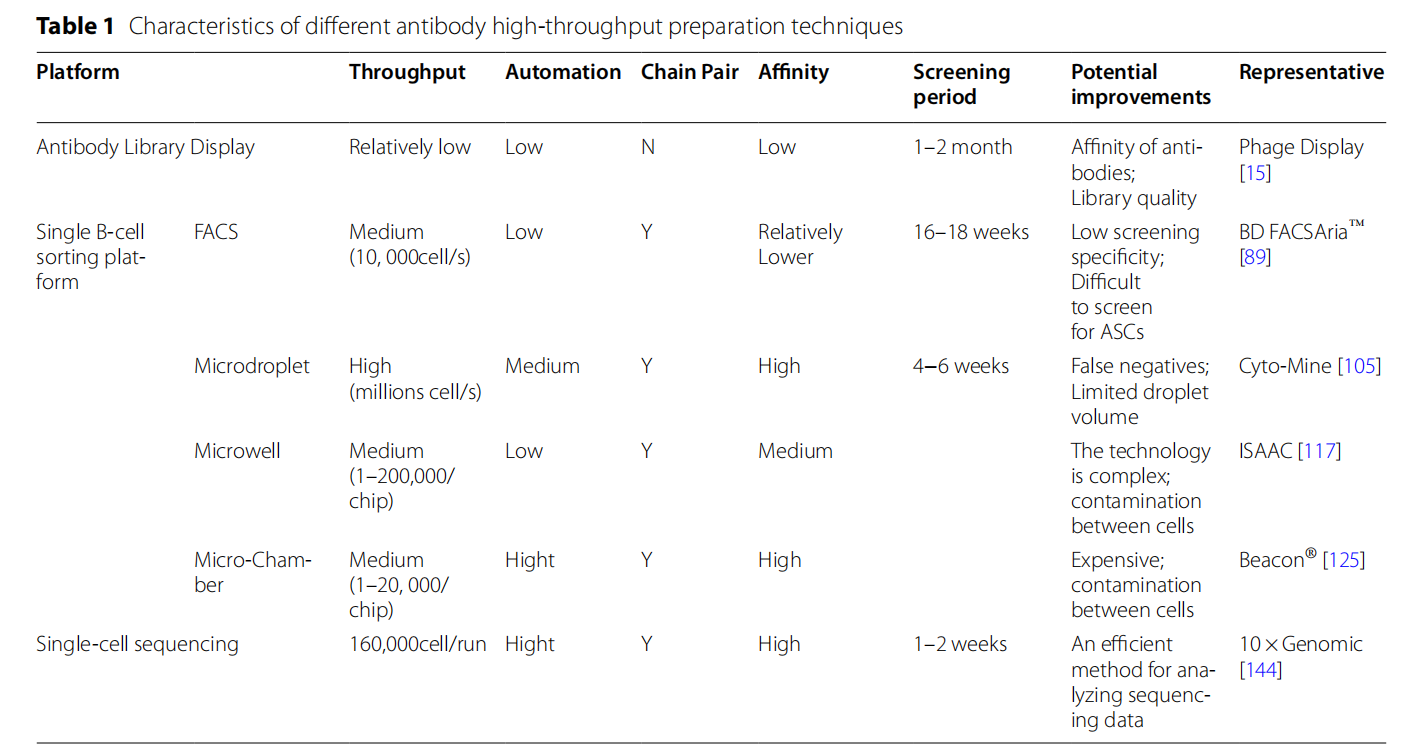
Through somatic DNA recombination, mammalian B cell antibody repertoires can generate 10^ 12 to 10^ 18 unique antibodies to recognize and combat a wide range of microbial pathogens . Antibody repertoires from immunized animals and humans are displayed using a variety of vectors (e.g., phage, yeast, ribosomes, etc.) to construct diverse antibody libraries. High-affinity binding vectors are then isolated through multiple rounds of iterative screening. The variable region gene sequences of the antibodies are determined by ELISA and sequencing analysis. These variable region gene sequences are then integrated into designated expression vectors for antibody production, followed by downstream characterization and research.
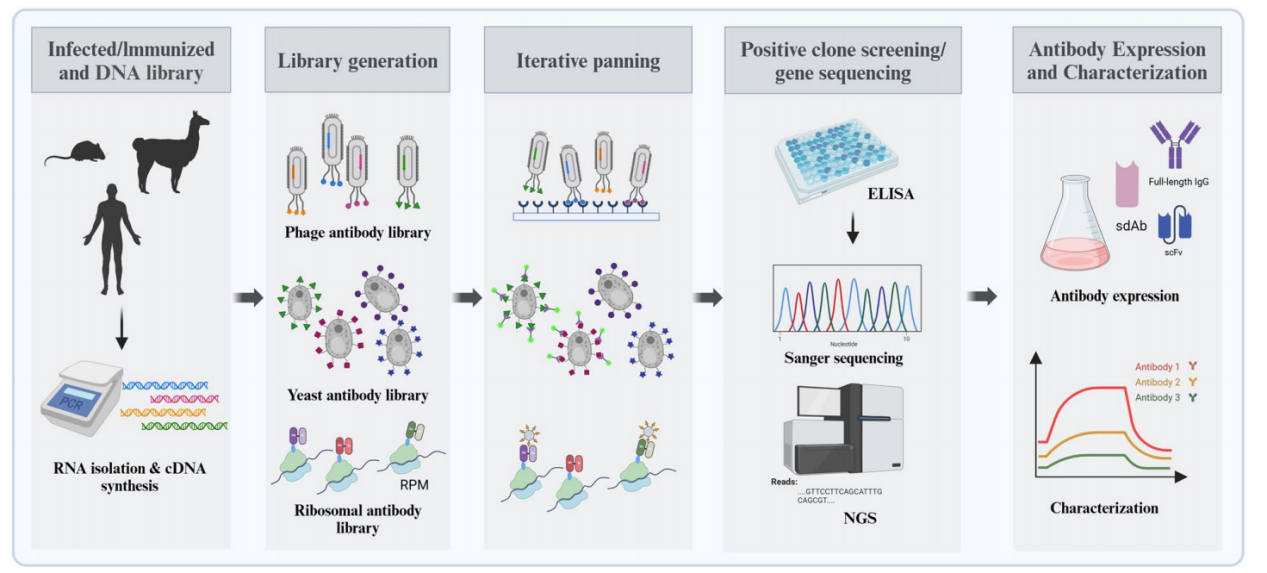
The main antibody library display technologies are phage display technology and cell display antibody library technology. Cell display platforms include yeast display, bacterial cell display and mammalian cell display, each of which has its unique advantages in expression efficiency and screening capabilities.
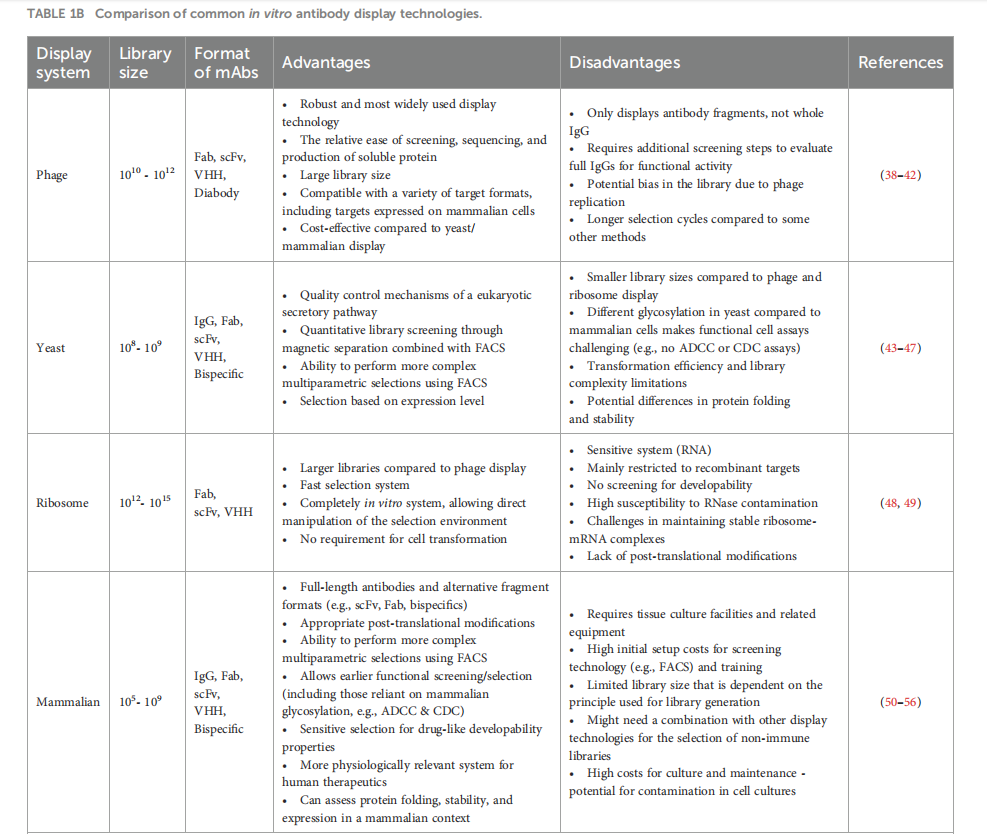
(Data source: Slavny P, et al. Front Immunol. 2024)
Single B cell antibody technology
Mammalian B lymphocytes are highly diverse, capable of producing up to 1012 antibody clonotypes . Single B cell antibody technology allows the direct isolation of single antigen-specific B cells from animal tissues or peripheral blood. The specific process of this technology includes antigen-specific B cell isolation, antibody gene amplification, and recombinant antibody expression and purification. Compared with traditional antibody production technologies, single B cell antibody production technology offers the advantage of rapidly obtaining naturally paired light and heavy chain variable regions through high-throughput screening of antigen-specific B cells from animals or humans.
Single B cell antibody technology mainly includes single B cell antibody preparation based on fluorescence-activated cell sorting, droplet microfluidics, and microplate-based microfluidic sorting.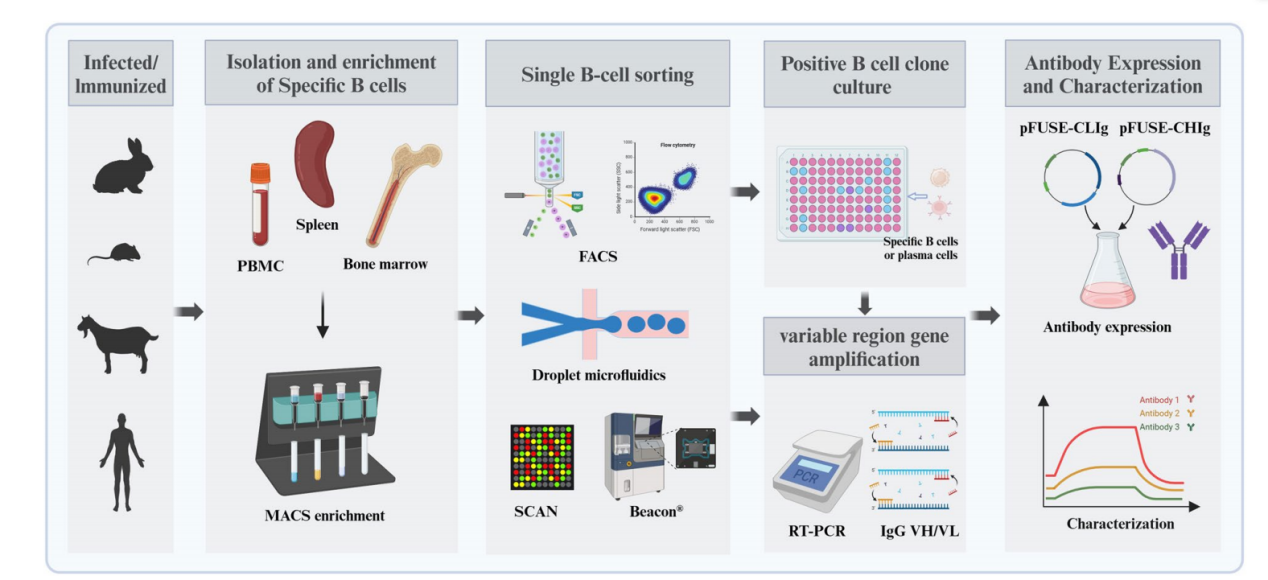
Single B cell antibody production based on fluorescence-activated cell sorting
B cell sorting using flow cytometry (FACS) to prepare single B cell antibodies is done by immunolabeling antigen-specific B cells in a cell suspension using fluorescently labeled antigens and antibodies against various cell surface markers. Single cells are then separated by flow cytometry, RNA is extracted to obtain cDNA, and the variable region genes of the antibody are amplified.
Advantages: Single antigen-specific B cells can be quickly and accurately identified through multi-color fluorescence, which promotes the development of antibodies.
Disadvantages:
1) Low sensitivity and high detection noise leading to false positive and false negative results;
2)FACS sorting relies on marker expression, and low marker expression can affect FACS separation and identification;
3)The application of FACS screening may be limited by unclear or absent cell surface markers, which may lead to the loss of ideal antibody clones ;
4)FACS usually requires a large number of starting cells to screen antigen-specific B cells, and operations during pretreatment may affect cell viability, resulting in difficulties in the identification of downstream antibody affinity and antibody activity.
Antibody preparation from single B cells using microfluidic sorting
Based on their working principles, microfluidics technologies are categorized into microdroplet and microwell systems. These microfluidic platforms are capable of detecting and isolating specific B cells at the single-cell level and supporting the cultivation of positive clones in a controlled nutrient environment.
Antibody production by single-cell sequencing
The complex process of V(D)J gene rearrangement, somatic hypermutation (SHM), and class switching generates a large number of variable region sequences in antibody heavy and light chains, contributing to the huge capacity of the B cell immune repertoire.
Single B cells are isolated and enriched from peripheral blood mononuclear cells (PBMCs), and RNA is extracted and reverse-transcribed into cDNA for sequencing library construction. B cells are sequenced using single-cell next-generation sequencing (NGS) technology to obtain large-scale, single-cell resolution data. Bioinformatics tools are used to analyze the sequencing data from individual B cells to obtain paired IgG heavy and light chain variable region genes. High-throughput single-cell sequencing provides a faster and more efficient solution by bypassing intermediate steps and directly obtaining high-quality antibody data.
This method, which selectively enriches target cells before sequencing, significantly increases the likelihood of identifying high-performance antibodies, thereby promoting the development of potent therapeutic antibodies. Artificial intelligence models such as machine learning and deep learning enable computational predictions of antibody structures, antigen interfaces, and interactions. These advanced methods improve data processing efficiency and support high-throughput antibody production and screening. These technologies can rapidly identify and optimize therapeutically valuable antibodies, accelerating the antibody development process and driving the discovery of new antibody drugs. The application of these technologies marks the arrival of a new era in high-throughput antibody screening, making the transition from discovery to product development more efficient and precise.

Summarize
Current high-throughput antibody screening technologies include screening strategies based on antibody library display, single-cell technology, and single-cell sequencing. Among these technologies, high-throughput single-cell sequencing stands out as the most effective approach for antibody production. It can provide gene expression profiles and immune profiles of large numbers of single cells in a single run. Combining it with machine learning and deep learning techniques can provide more effective strategies for data discovery. However, it also has many limitations, and future improvements and enhancements to algorithms, scoring functions, databases, and benchmarking tools are needed to better address these challenges.