C-type lectin domain family 12 member A (CLEC12A), also known as C-type lectin-like molecule-1 (CLL-1), myeloid inhibitory C-type lectin-like receptor (MICL) , killer cell lectin-like receptor 1 (KLRL1), dendritic cell-associated lectin 2 (DCAL2) , and CD371, is a myeloid inhibitory C-type lectin receptor that acts as a negative regulator of myeloid cell activation , playing multiple roles, including phagocytosis, pathogen recognition, complement activation, and cell adhesion . It is highly expressed on AML cells and exhibits stable expression throughout disease progression, making it a potential therapeutic target for the treatment of acute myeloid leukemia (AML).

(Data source: Soleimani Samarkhazan H, et al. Biomark Res. 2025)
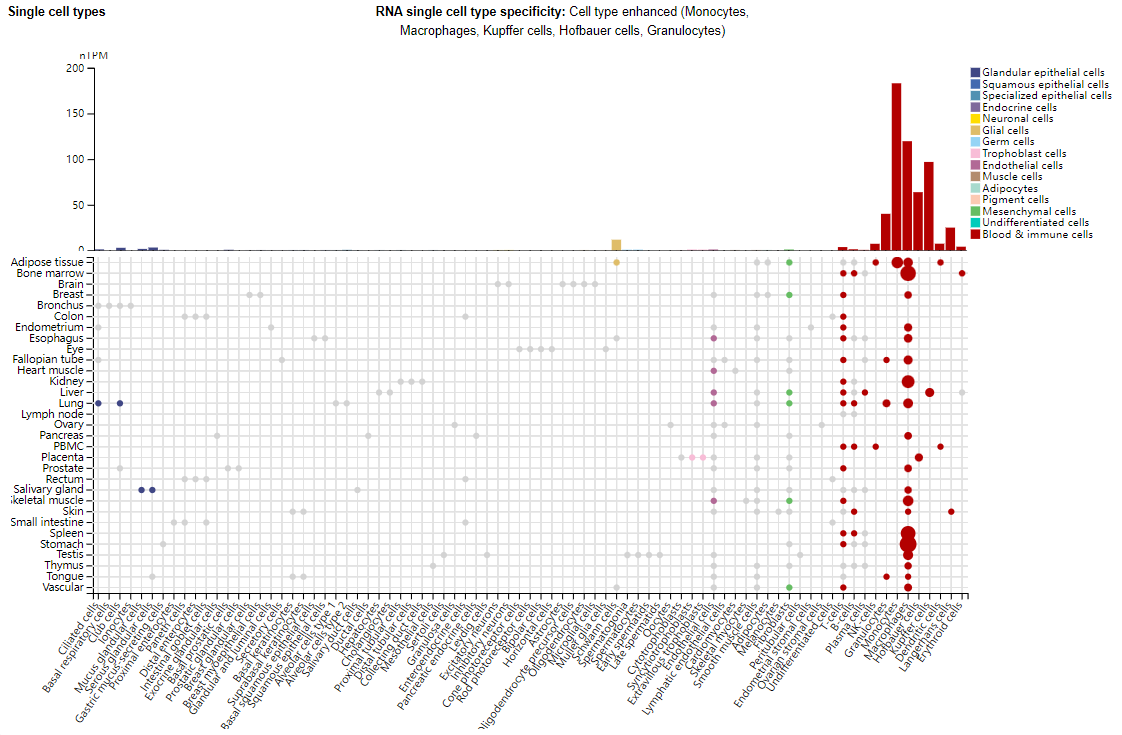
Expression distribution of CLEC12A
CLEC12A is primarily expressed in monocytes, macrophages, Kupffer cells, Hofbart cells, and granulocytes. CLL-1 is not expressed in erythrocytes, platelets, natural killer cells, B cells, T cells, or plasmacytoid dendritic cells. Although CLL-1 is expressed in immature monocytes, its expression decreases as the cells differentiate into macrophages. CLL-1 is highly expressed in ALL cells.
(Data source: Uniprot)
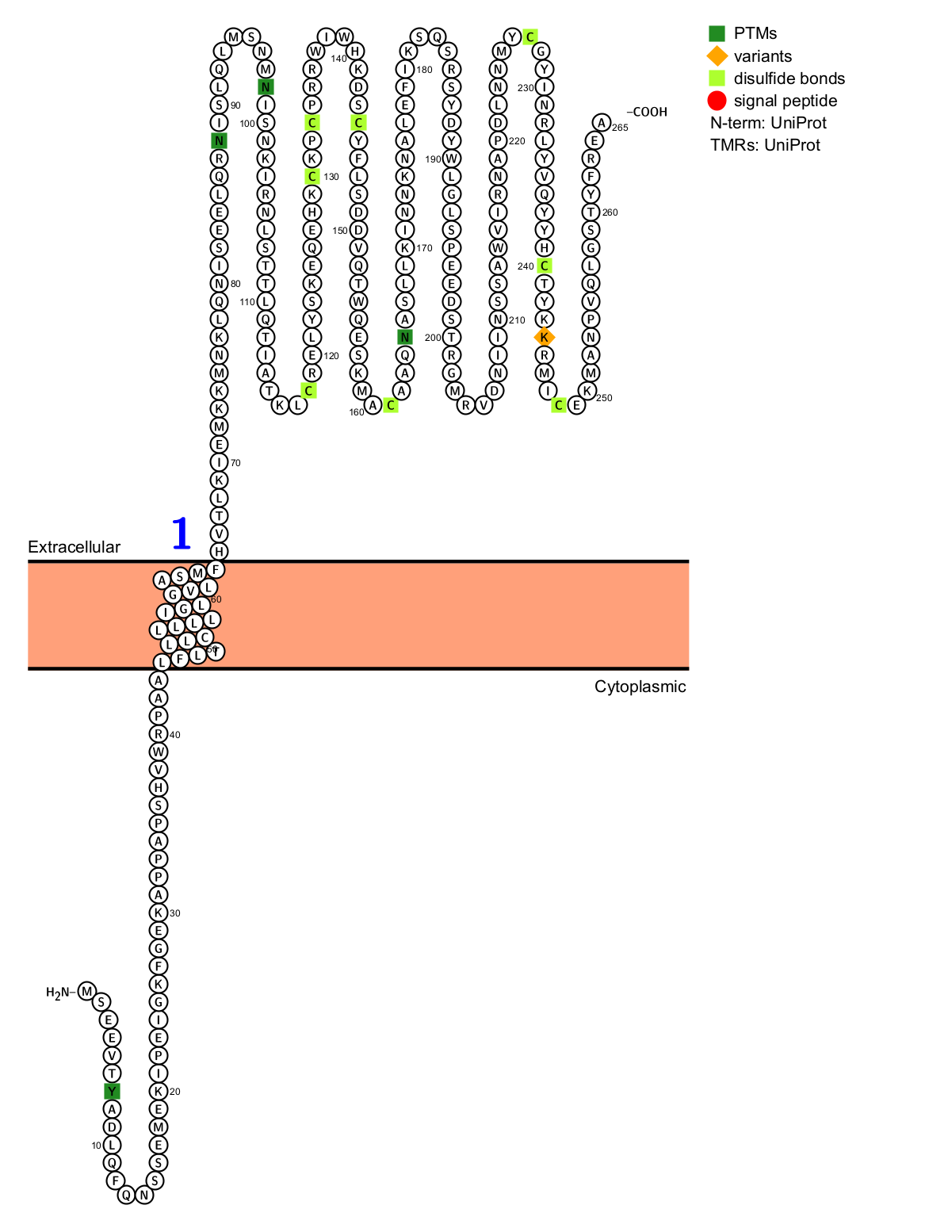
Structure of CLEC12A
The CLEC12A gene, located on chromosome 12p13, consists of 265 amino acids and is characterized by an extracellular segment that constructs a unique C-type lectin-like domain containing six cysteines commonly found in C-type lectins. Furthermore, this domain contains six potential N-glycosylation sites and one predicted O-glycosylation site. The remainder of the protein is composed of a prominent, hydrophobic, single-span membrane segment and a cytoplasmic tail, completing its structural organization. Within this cytoplasmic domain lies an I/VXYXXL immunotyrosine-based inhibitory motif (ITIM) and a YXXM motif, contributing to the functional complexity of the CLL-1 glycoprotein.
(Data source: Uniprot)
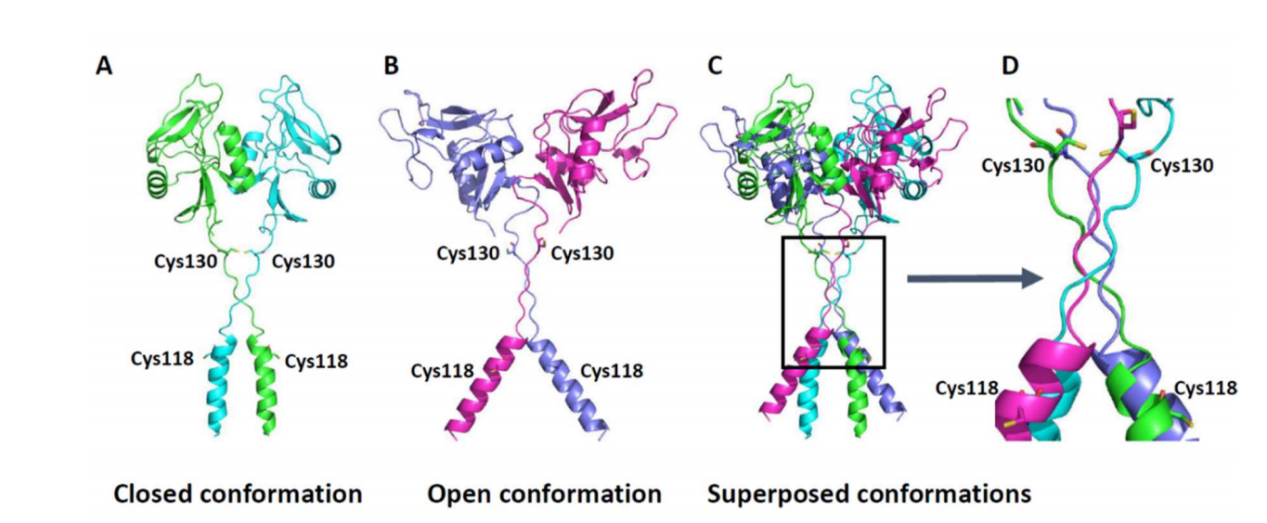
(Data source: Vitry J, et al. Int J Mol Sci. 2021)
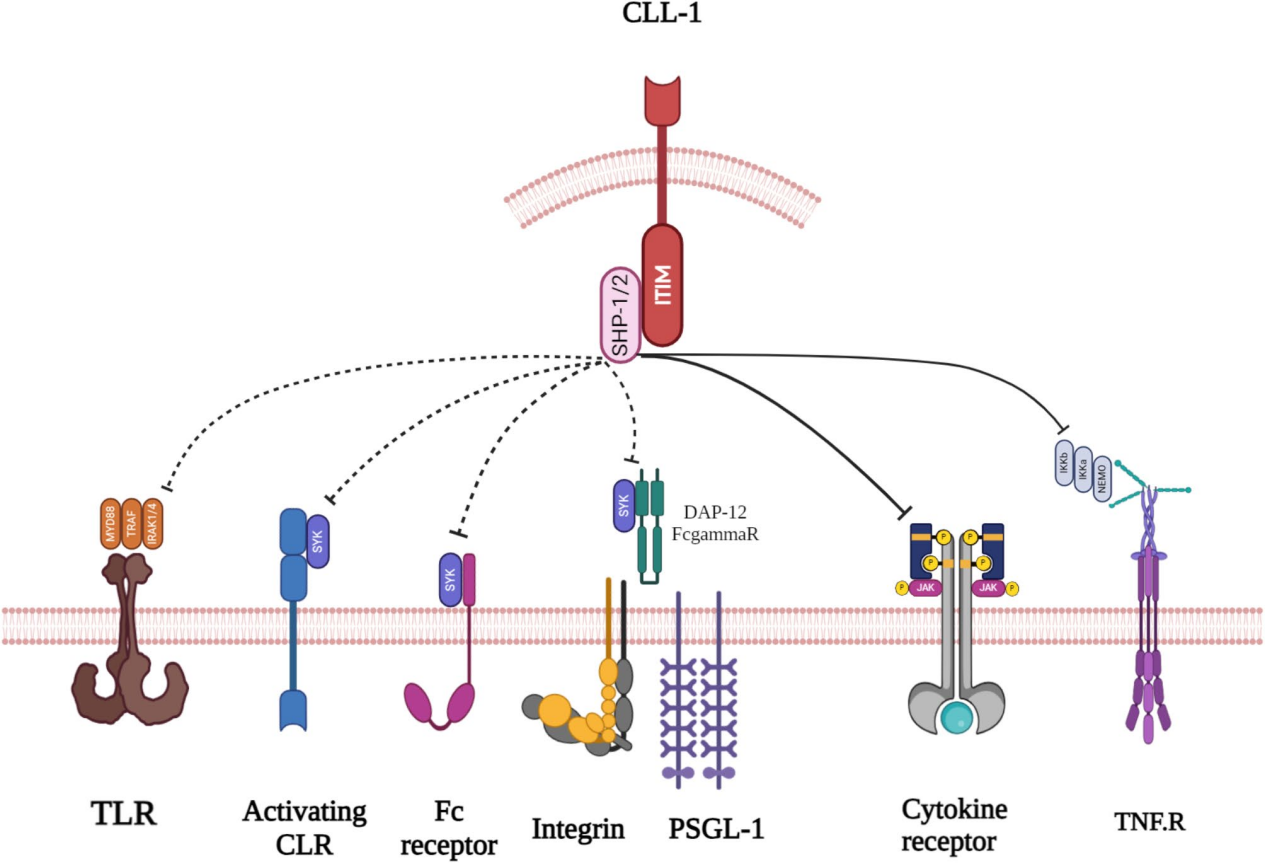
Signaling pathway and regulation of CLEC12A
CLRs, integrins, selectin receptors (such as PSGL-1), and Fc receptors triggers Syk activation through ITAMs on the receptors themselves or through adaptor proteins (DAP12/Fcrγ). Furthermore, SHP-1/2 play a role in dephosphorylating and inactivating Syk. CLL-1 can inhibit the signaling functions of these four receptors. The only receptor that CLL-1 inhibits neutrophil activation is the TNF receptor (TNFR). In other myeloid cells, SHP-1/2 dephosphorylates and inhibits IKK and TRAF1/2, proteins that may be potential targets of neutrophils. CLL-1 inhibits Toll-like receptor (TLR) activity on dendritic cells. In other myeloid cells, SHP-1/2 dephosphorylates and inhibits JAK/STAT proteins associated with cytokine receptors, suggesting a potential mechanism by which CLL-1 inhibits cytokine signaling.
(Data source: Soleimani Samarkhazan H, et al. Biomark Res. 2025)
Targeted therapy for CLEC12A
Currently, the drugs being developed specifically for CLL-1 are mainly chimeric antigen receptor (CAR)-modified T cell immunotherapy, and monoclonal antibodies, bispecific antibodies, and antibody-drug conjugates are also in preclinical development.

(Data source: Soleimani Samarkhazan H, et al. Biomark Res. 2025)
MCLA-117, a CLL-1-CD3 bispecific antibody with a native human IgG format, stimulates T cell activation and proliferation, releases pro-inflammatory cytokines, and redirects T cells to lyse CLL-1-positive cells. Currently, there is an ongoing clinical trial ( NCT03038230 ) investigating the use of MCLA-117 in patients with primary or secondary acute myeloid leukemia (AML).
DCLL9718S is a humanized monoclonal IgG1 antibody targeting CLL-1. It consists of two PBD dimers connected by a disulfide-based, self-destructive linker that acts as a DNA alkylating and cross-linking agent. Once DCLL9718S binds to CLL-1 on target cells, it is internalized and degraded in lysosomes. This releases the active form of the PBD dimer, which forms a covalent bond with DNA in the minor groove. This interaction damages DNA, leading to cell death. The study revealed that CLL-1 and CD33 have similar frequency and trafficking properties. These findings suggest that CLL-1 may be a target for ADCs in AML . DCLL9718S successfully eradicated tumor cells in an AML xenograft model and exhibited no target-independent toxicity at doses that resulted in depletion of target myeloid cells in cynomolgus monkeys.
CAR-T cell therapy is a novel and highly effective technology for treating hematologic malignancies. CD33 and CD123 are currently the most commonly targeted antigens for CAR-T cell therapy in AML. Although AML cells or LSCs may have higher levels of CD33 and CD123 than normal cells, targeting these cells still carries the risk of off-target effects, leading to long-term or permanent bone marrow suppression. CLL-1-targeted CAR-T is a novel approach for treating AML.
Laborda E et al. created a new CLL-1 CAR T cell that exhibited potent and specific cytotoxicity against the AML cell lines HL60, MOLM13, and MOLM14. In vivo studies demonstrated complete tumor regression, with no signs of relapse within 80 days of tumor regression, and persistent CAR-T cell survival in a xenograft HL-60 mouse model.
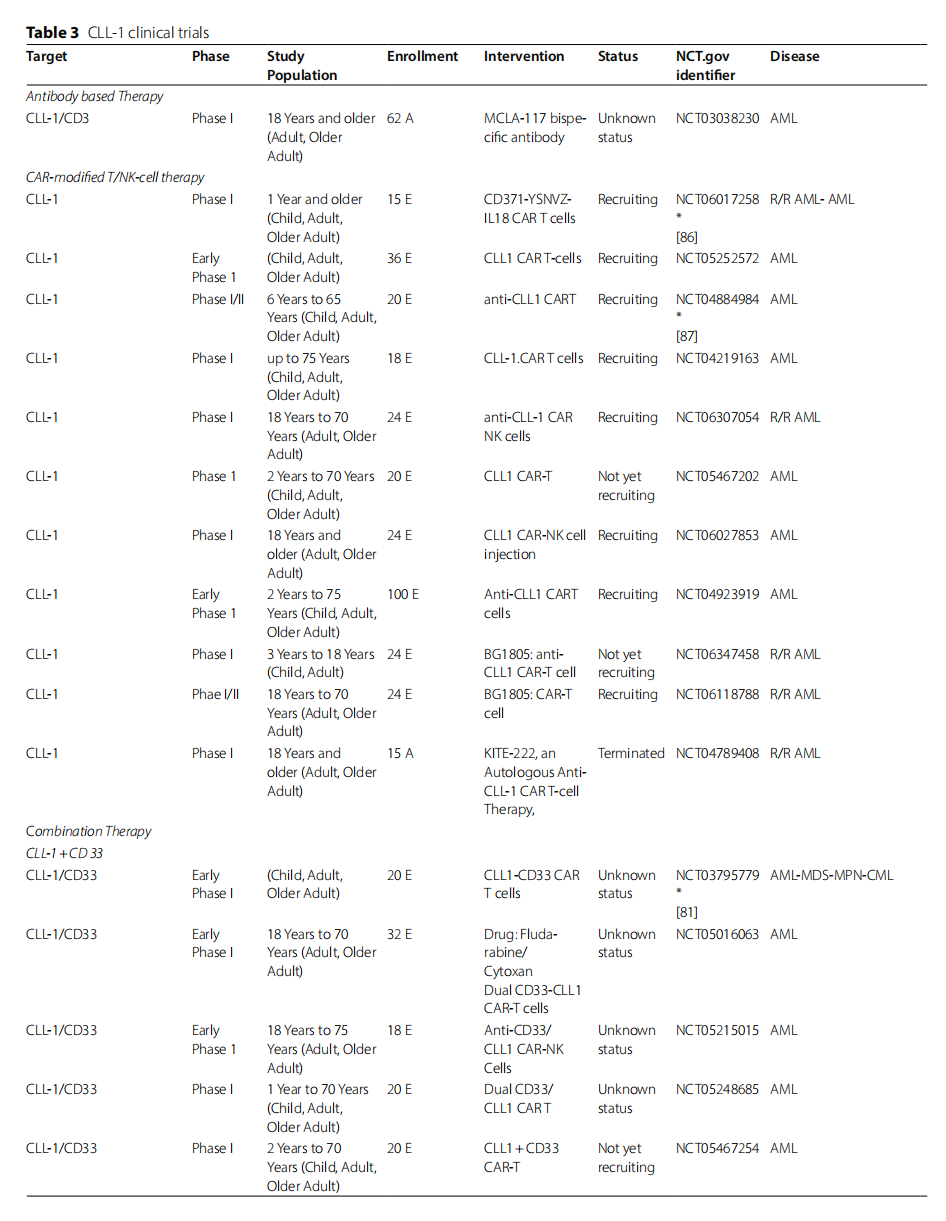
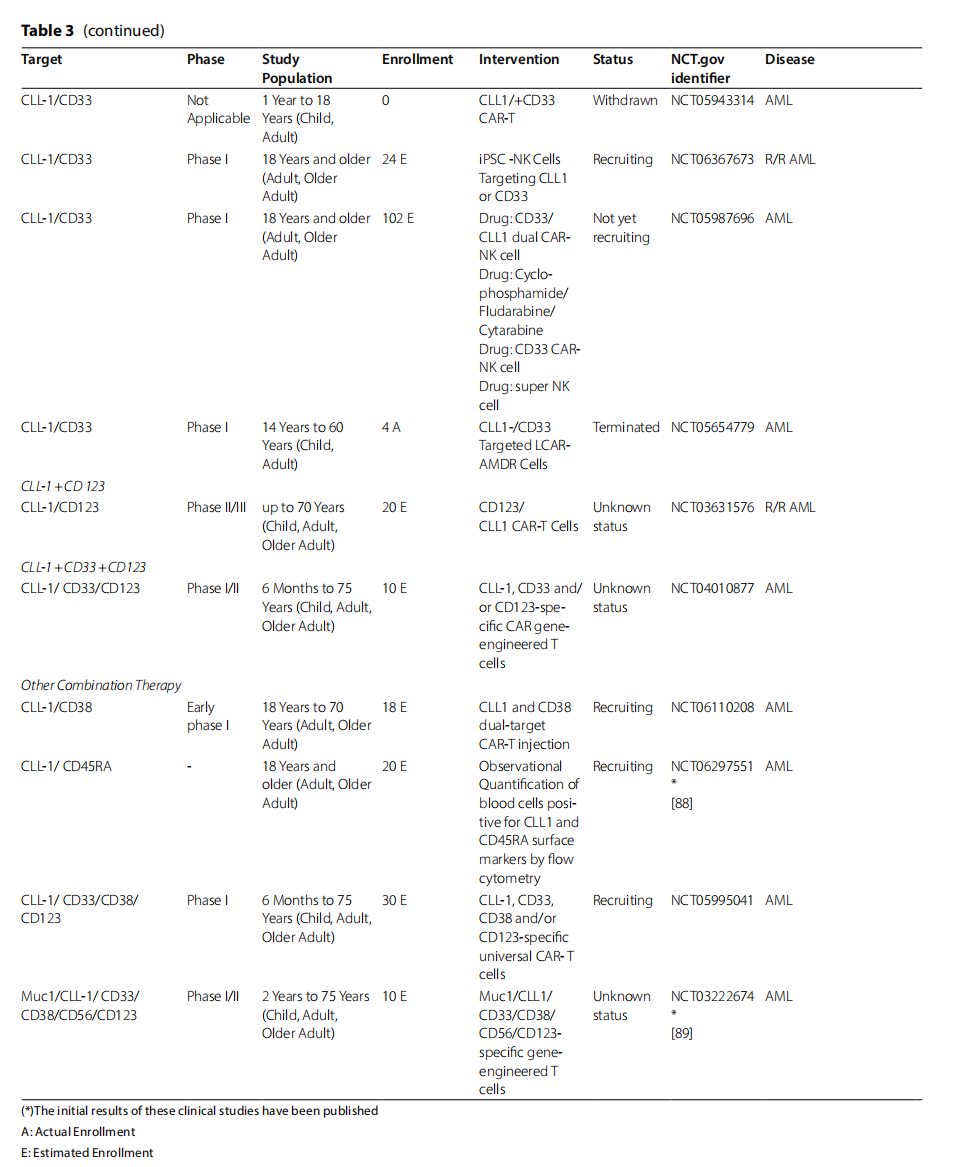
(Data source: Soleimani Samarkhazan H, et al. Biomark Res. 2025)
