The receptor tyrosine kinase FLT3, also known as stem cell tyrosine kinase 1 (STK-1), fetal liver kinase-2 (FLK-2), and CD135, serves as the cell surface receptor for the cytokine FLT3LG and regulates the differentiation, proliferation, and survival of hematopoietic progenitor cells and dendritic cells . Acute myeloid leukemia (AML) is a heterogeneous hematologic malignancy. Approximately 30% of AML patients carry FLT3 mutations, which are associated with higher relapse rates and lower survival. The development of FLT3 inhibitors has significantly improved the treatment of patients with FLT3-mutated AML. FLT3 is a therapeutic target and diagnostic marker for AML.
Expression distribution of FLT3
FLT3 is mainly expressed on the surface of dendritic cells, inhibitory neurons, Kupffer cells, Langerhans cells, macrophages , hematopoietic stem cells, and early myeloid and lymphoid progenitor cells . Regarding tissue-specific expression, under physiological conditions, hFLT3 is significantly expressed in the spleen, lymph nodes, and bone marrow.
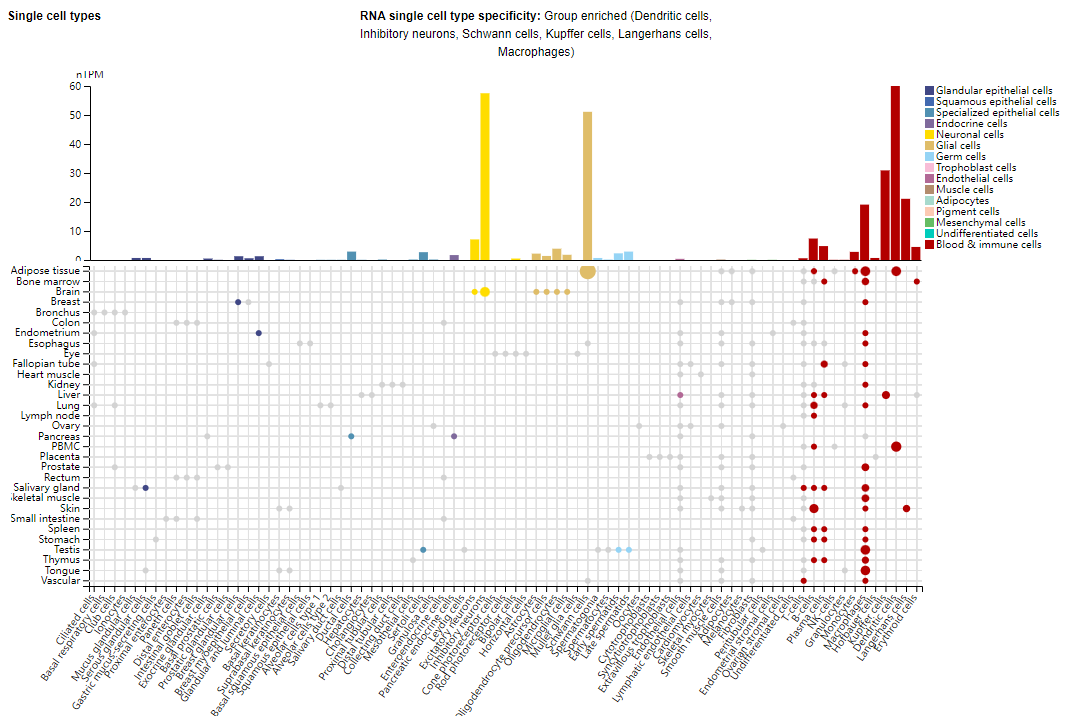
(Data source: Uniprot)
The structure of FLT3 and its receptor
FLT3 is one of the most unique class III receptor tyrosine kinases (RTKs). Located at locus 13q12.2 on chromosome 13 , it consists of 993 amino acids. FLT3 is a transmembrane protein composed of four domains: an extracellular domain (ED) consisting of five immunoglobulin-like (Ig-like) units (D1 to D5); a transmembrane domain (TMD); a juxtamembrane domain (JMD); and a conserved cytoplasmic domain consisting of two tyrosine kinase domains (TKDs) separated by a kinase insert. The JMD, consisting of amino acid residues 572-603 in FLT3, connects the TMD to TKDI and TKDII (amino acid residues 604-958 in FLT3). TKDII is of particular interest, regulating the activation loop, a structural element that allows downstream substrate binding.
FLT3 ligand (FLT3L) is a soluble form of FLT3L that is thought to be released from the cell membrane into the circulation by protease cleavage. Soluble FLT3L is a non-covalently linked dimer composed of two short α-helical bundles that binds to the FLT3 receptor through its N-terminus.
The monomeric receptor is inactive, and this inactive state is maintained by the interaction between the JMD and the TKD (blocking the ATP binding site). FLT3 activation is induced by FLT3L binding to the receptor ED, which causes conformational changes leading to dimerization, autophosphorylation, activation of the kinase domain, and subsequent activation of downstream signal transduction pathways, ultimately promoting the transcription of genes that regulate cell survival, proliferation, and differentiation.

(Data source: Rataj J, et al. Biomed Pharmacother. 2025)
FLT3 signaling pathway and regulation
The interaction between FLT3L and FLT3 leads to a series of rapid conformational changes in the receptor, which adopts a catalytically active form that initially promotes autophosphorylation and homodimerization events. FLT3 exposes binding sites for various adaptor molecules, and docking of these adaptor molecules primarily leads to activation of the PI3K/AKT/mTOR, RAS/MAPK/ERK, and JAK/STAT pathways , which regulate cell proliferation, apoptosis, and cell cycle progression.
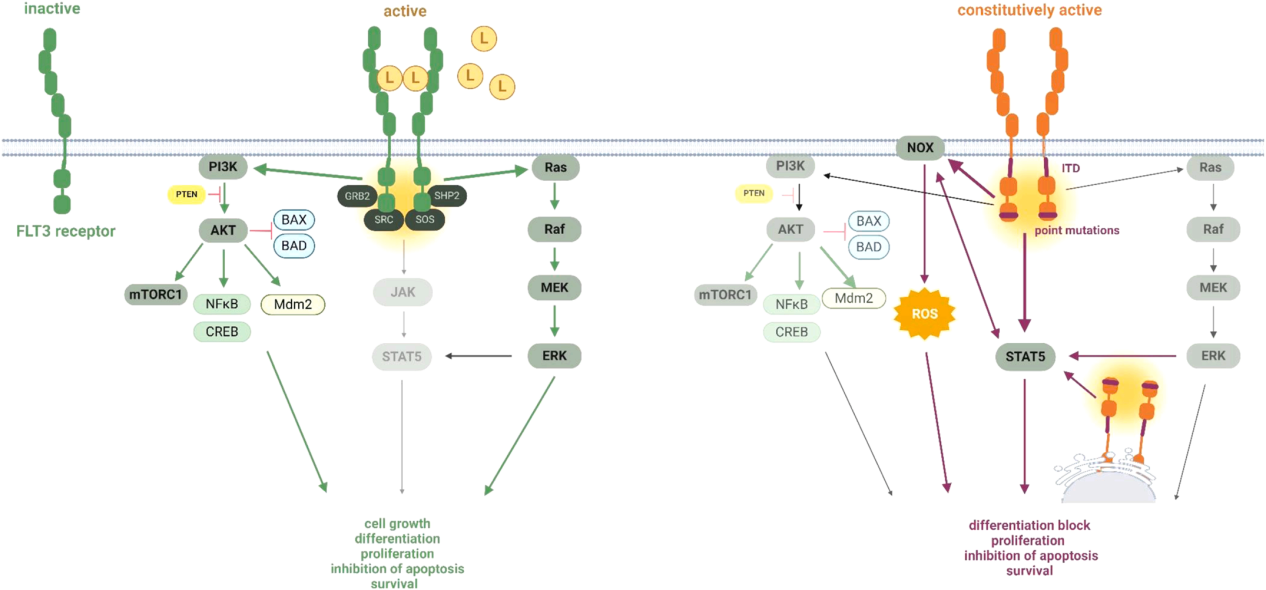
(Data source Rataj J, et al. Biomed Pharmacother. 2025)
FLT3 -targeted therapy
Inhibitors targeting FLT3 play an important role in the treatment of AML. The first-generation FLT3 inhibitors include tandutinib (CT53518), lestatin (CEP-701), sunitinib (SU11248), midostatin (PKC412) and sorafenib (BAY 43-9006), all of which have shown inhibitory effects on FLT3 with varying degrees of specificity and off-target effects. There are many small molecule inhibitors targeting FLT3 in clinical development.
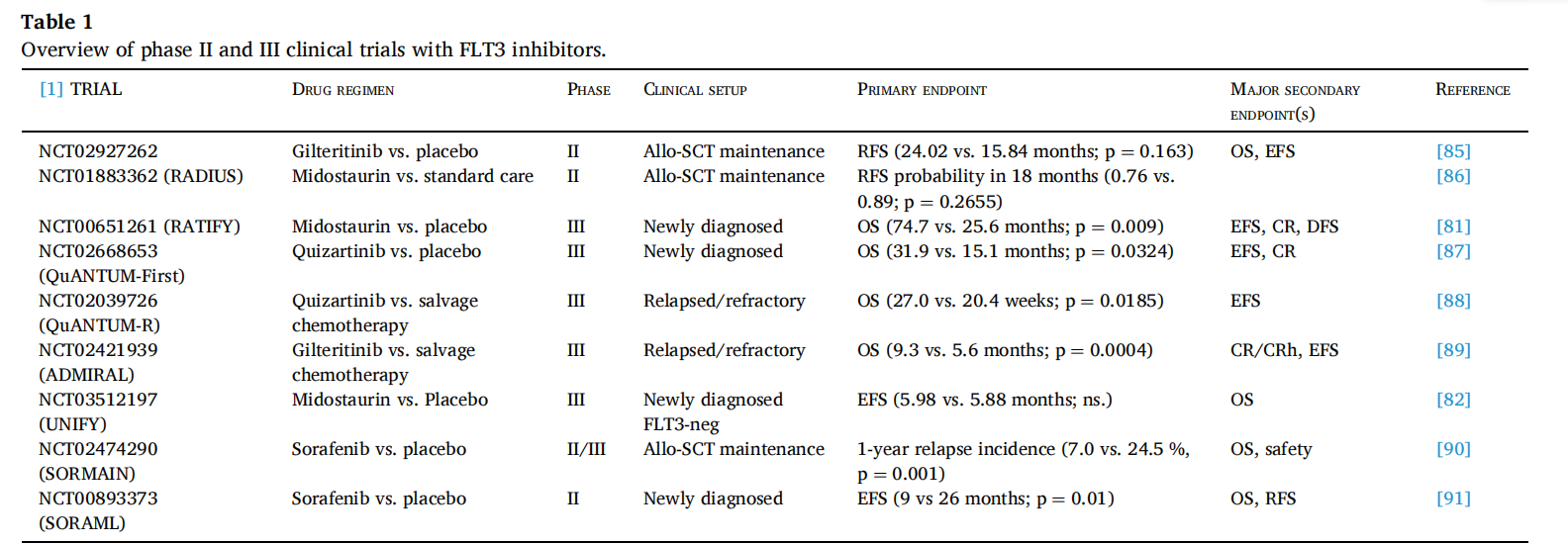
(Data source: Rataj J, et al. Biomed Pharmacother. 2025)
Resistance to targeted FLT3 inhibitors arises during tumor treatment. FLT3 inhibitors are metabolized by CYP34A; FGF2 activates the downstream RAS/MAPK pathway; AXL1 activates the downstream RAS/MAPK and PI3K/AKT/mTOR pathways; FLT3 ligand (FL) binds to wild-type FLT3, activating the downstream RAS/MAPK, JAK/STAT, and PI3K/AKT/mTOR pathways; FLT3 mutations (such as F961L, D835, and Y842) contribute to off-target resistance; and the expression of anti-apoptotic genes and mutations in genes and transcription factors contribute to cancer cell survival. Two strategies are currently being used to combat the rapid development of resistance to selective FLT3 inhibitors: combination strategies and multi-target drug discovery. Dual inhibitors aim to simultaneously target another kinase to mitigate the development of resistance.

(Data source: Rong QY, et al. Drug Discov Today. 2025)
In addition to small molecule-targeted FLT3 therapies, there are other treatments targeting FLT3, such as monoclonal antibodies, antibody-drug conjugates, bispecific antibodies, and CAR-T cell therapy.

(Data source: Rataj J, et al. Biomed Pharmacother. 2025)
CLN-049 is a bispecific T cell engager targeting FLT3 and is currently in Phase 1 clinical development for the treatment of myelodysplastic syndrome, refractory acute myeloid leukemia, and relapsed acute myeloid leukemia.
NCT05143996 is a Phase 1, open-label, preliminary pharmacokinetic (PK) and safety study of CLN-049 in patients with relapsed/refractory acute myeloid leukemia (AML) or myelodysplastic syndrome (MDS). Cullinan completed the single ascending dose (SAD) portion of the CLN-049 study at the end of 2022 and is transitioning to the multiple ascending dose (MAD) portion. Due to dose-limiting injection-site reactions during subcutaneous (SC) administration, the study team decided to continue dose escalation via intravenous (IV) administration.
SENTI-202 , developed by Senti Bio , is a first-in-class logic-gated CAR-NK cell therapy that simultaneously targets CD33 and FLT3. It is designed to address key limitations of NK cell therapies evaluated in clinical trials by integrating three chimeric proteins using a logic-gated gene circuit. This design includes a bivalent ( OR-gated) activating CAR (aCAR) that is independently triggered by one or both CD33 (a validated AML target) and/or FLT3. A NOT gate or an inhibitory CAR (iCAR) is included in the gene circuit to protect healthy cells from potential aCAR-mediated on-target/off-tumor toxicities, thereby enhancing the inherent health cell-protective activity of NK cells. The drug is currently in a Phase I clinical trial for the treatment of relapsed/refractory AML and myelodysplastic syndrome (MDS ). In December 2024, Senti Bio reported positive preliminary clinical results from this trial. In all three patients, SENTI-202 was well tolerated, with an adverse event profile consistent with lymphodepleting chemotherapy in AML patients.

(Data source: Senti Bio official website)
