NKG2-D type II integral membrane protein (KLRK1), also known as CD314 and NKG2D, is a member of the killer cell lectin-like receptor subfamily K, member 1. As an activating and co-stimulatory receptor, it participates in immune surveillance upon binding to various cellular stress-induced ligands expressed on the surface of autologous tumor cells and virus-infected cells. It provides stimulatory and co-stimulatory innate immune responses on activated killer (NK) cells, leading to cytotoxic activity. It serves as a co-stimulatory receptor for the T cell receptor (TCR) in CD8+ T cell-mediated adaptive immune responses, amplifying T cell activation. It also stimulates perforin-mediated elimination of tumor cells expressing the ligand.
(Data source: Liu Z, et al. Cytokine Growth Factor Rev. 2024)
Expression distribution of KLRK1
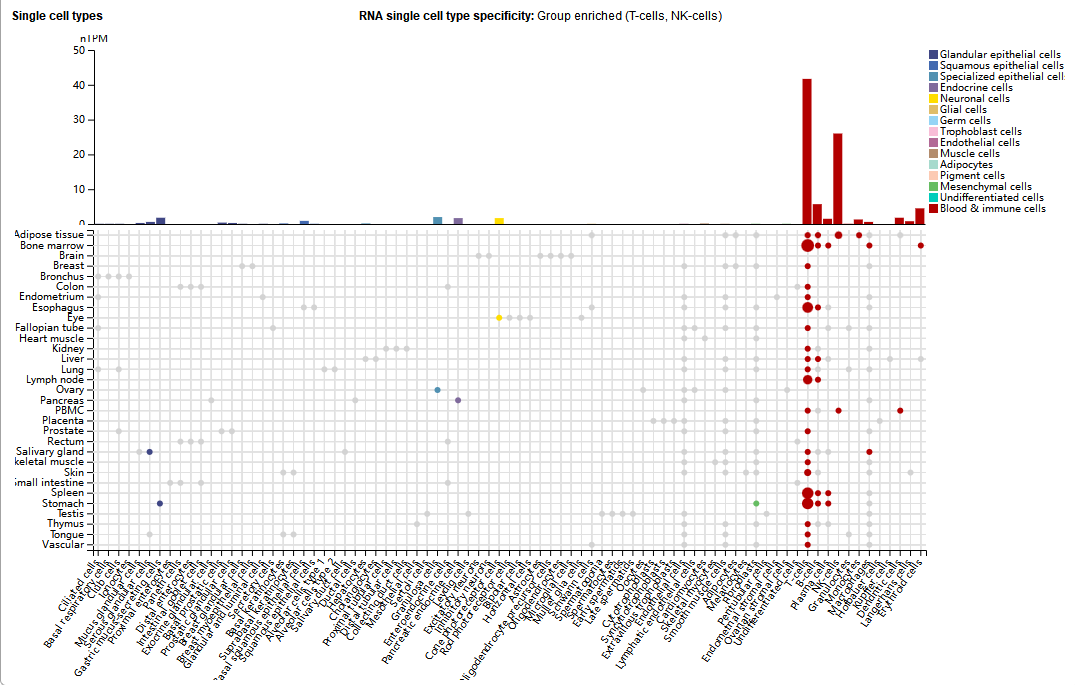
KLRK1 is mainly expressed in NK cells, but is also expressed in large quantities in T cells (such as NKT cells, CD8+T cells, and γδT cells), and in small quantities in immune cells such as B cells, red blood cells, and plasma cells.
(Data source: uniprot)
Structure of KLRK1 and its receptor
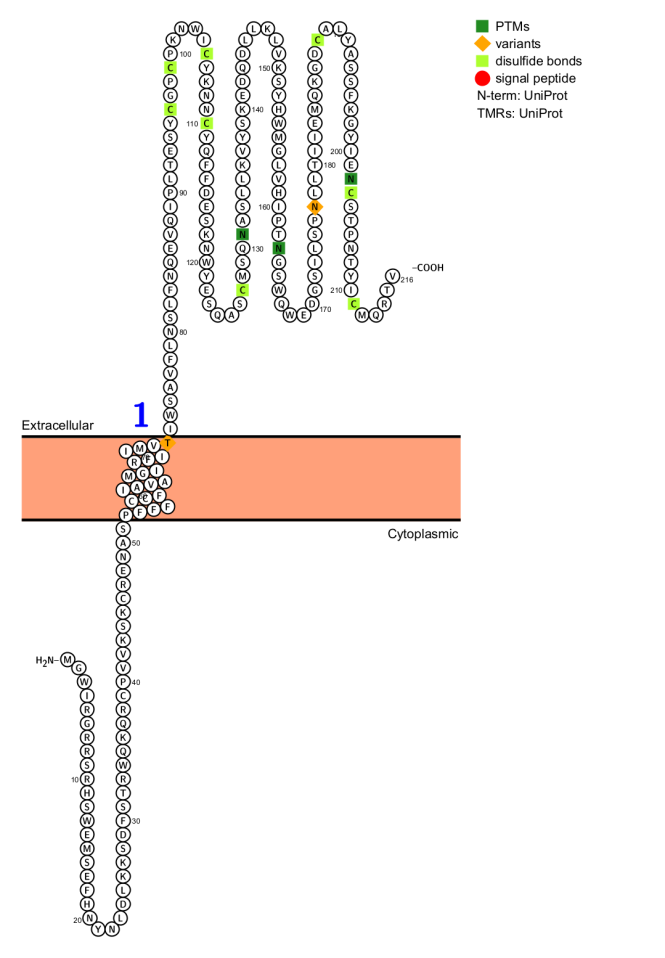
NKG2D is encoded by the KLRK1 gene and is located in the NK gene complex on chromosome 12, at 12p13.2. The NKG2D receptor is a homodimer composed of two type II transmembrane glycoproteins, with a C-type lectin-like structure on the cell membrane. The transmembrane region of NKG2D contains a positively charged lysine residue, which electrostatically interacts with the negatively charged aspartate residue in the transmembrane region of DAP10, forming the structural basis for their stable binding. The intracellular segment of NKG2D itself is very short and lacks signaling capacity.
(Data source: Uniprot)
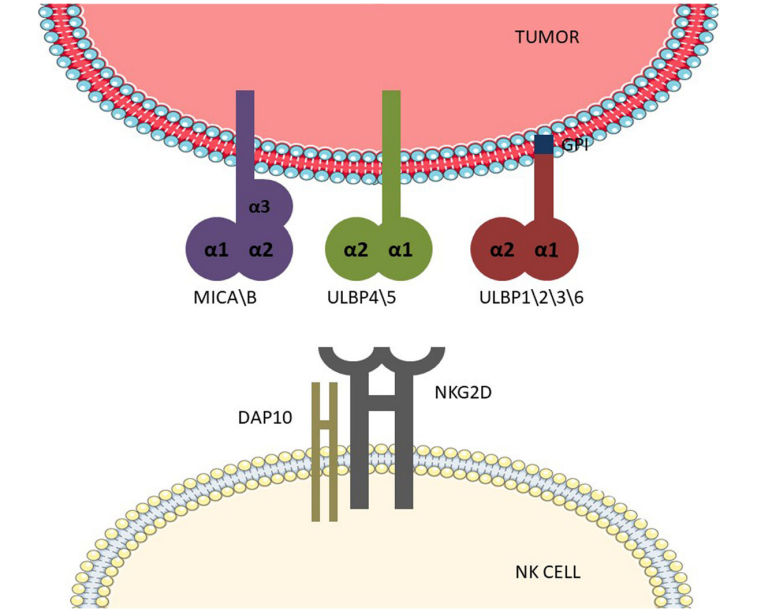
NKG2D recognizes numerous ligands, including MICA/B and UL16-binding proteins 1-6 (ULBP1-6). NKG2DLs are structurally similar to MHC class I molecules. MICA/B share the same α1, α2, and α3 domains as MHC class I molecules, with the α3 domain being an immunoglobulin-like domain, whereas ULBPs contain only α1 and α2 domains. ULBP1, -2, -3, and -6 are GPI-anchored receptors, while ULBP4 and -5 possess a transmembrane domain and a cytoplasmic tail.
(Data source: Duan S, et al. Mol Cancer. 2019)
Function of KLRK1 in NK cells
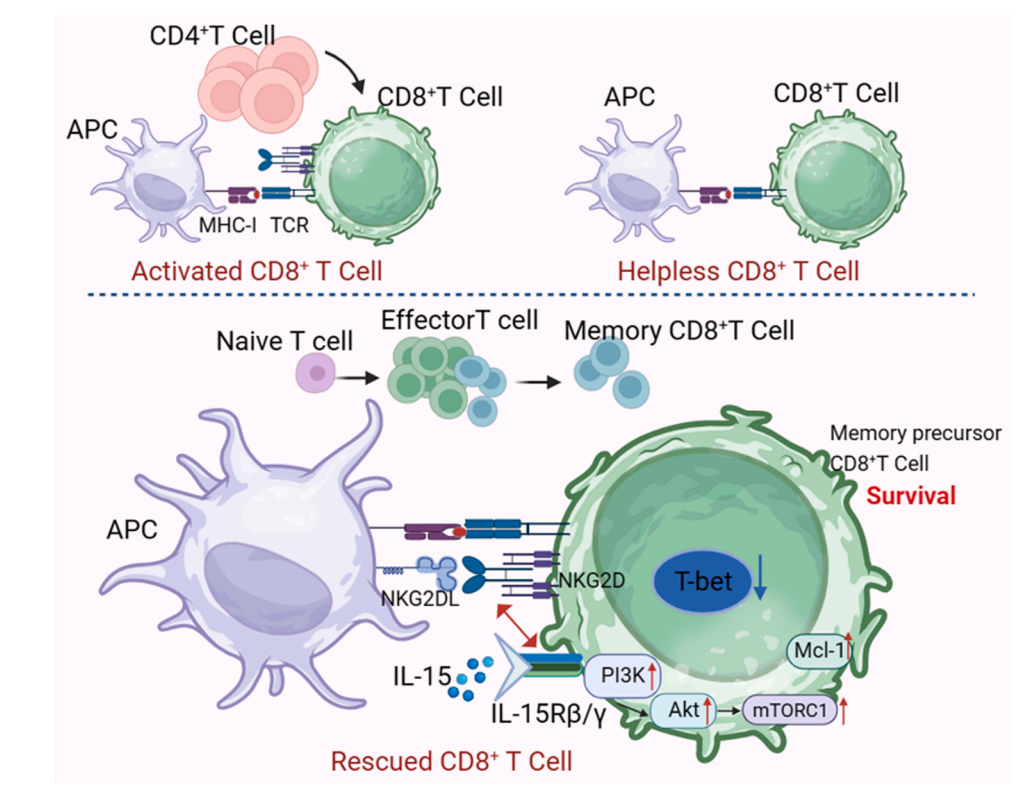
KLRK1 binds to DAP10. DAP10 contains a YXXM motif, which recruits PI3K and GRB2, subsequently activating the Rac1/PAK/c-RAF/MEK/ERK and GrB2/VAV-1 pathways. This ultimately induces NK cell cytotoxicity, releasing cytokines and killing tumor cells through perforin/granzymes, TNF-α/TNF-R1, and Fas/FasL. KG2DL is expressed on tumor cells. NKG2DL activates NKG2D, leading to NK cell activation and tumor cell elimination. Various mechanisms in the body regulate the expression of NKG2D receptors and NKG2DL, thereby affecting the immune system's clearance of tumor cells and contributing to tumor escape.
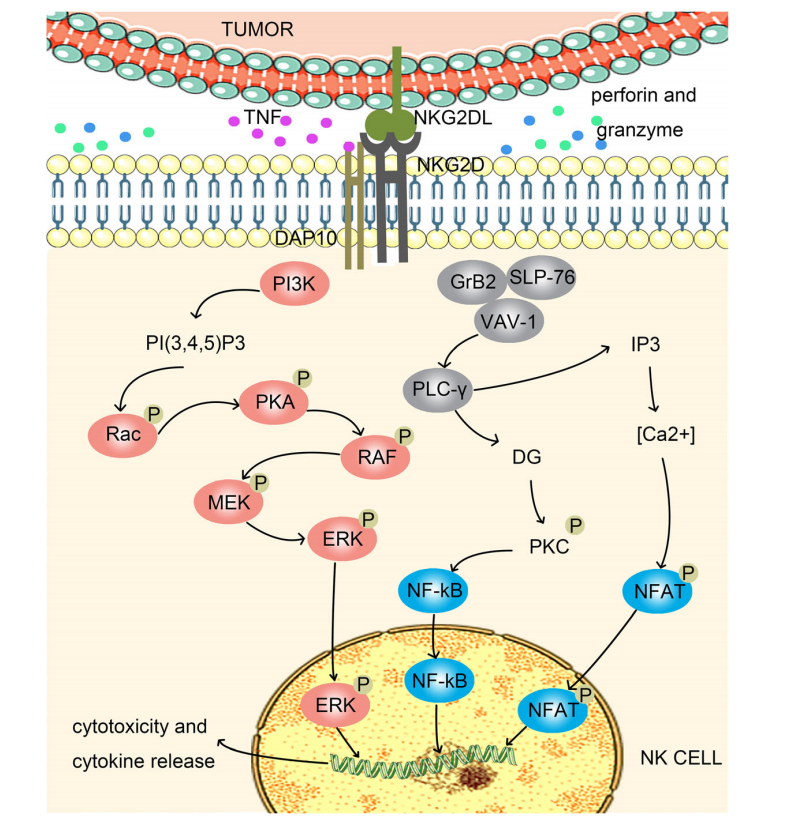
(Data source Duan S, et al. Mol Cancer. 2019)
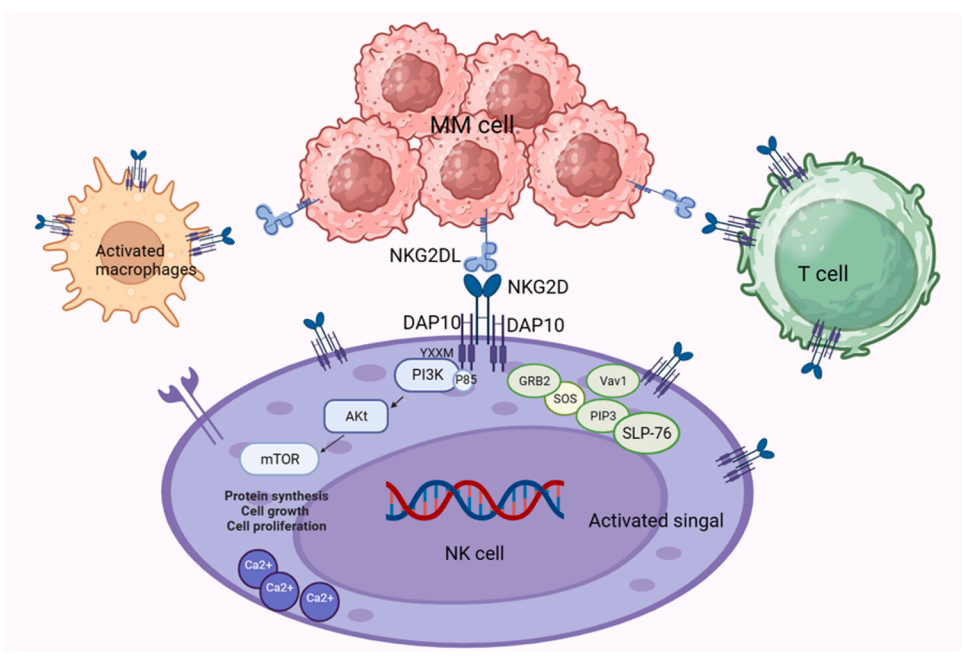
(Data source: Liu Z, et al. Cytokine Growth Factor Rev. 2024)
NKG2D-mediated tumor immune escape
In various cancers, including cervical cancer, pancreatic cancer, and melanoma, the expression of the NKG2D receptor on immune cells (such as NK cells and CD8+ T cells) is significantly reduced, thereby promoting tumor immune escape.
Prostaglandin E2 (PGE2) binds to EP2/4 via the AC/cAMP/PKA pathway and inhibits NKG2D transcription. Indoleamine inhibits NKG2D transcription via the JNK pathway. Tregs and MDSCs secrete cytokines, such as TGF-β, which inhibit NKG2D expression. Tumors themselves also express NKG2D receptors, which bind to NKG2DL on adjacent tumor cells, activating the PI3K/AKT/mTOR signaling pathway, promoting tumor angiogenesis, metastasis, and proliferation.

(Data source: Duan S, et al. Mol Cancer. 2019)
Targeted therapy of KLRK1
Azerutamig (DF1001) is a novel TriNKET® immune engager that stimulates innate and adaptive immune effector cells, including NK, CD8+ T, NKT, and γδ T cells, by binding to the activating receptors CD16a and/or NKG2D. DF1001 redirects immune effector cells to target tumor cells by anchoring HER2.
DF1001 is uniquely different from HER2-targeted drugs due to its unique immune-binding mechanism of action, which allows it to be active even against cancers with low HER2 expression, whereas HER2 monoclonal antibodies (such as trastuzumab) are ineffective against these cancers. In addition to DF1001's ability to target a variety of HER2-expressing tumors, its favorable preclinical safety profile allows it to be combined with standard-of-care therapies, including sacituzumab govitecan-hziy, a Trop2-targeting ADC approved for the treatment of metastatic breast cancer.

(Data source: Dragonfly Therapeutics official website)
Tesnatilimab ( JNJ-64304500 ) is a human immunoglobulin G4 isotype monoclonal antibody developed by Johnson & Johnson that targets NKG2D. It can bind to the NKG2D receptor and block the downstream pro-inflammatory immune response.
