Background
IgG antibodies, essential for the normal functioning of the immune system, recognize antigens and neutralize pathogens through their Fab regions. Their Fc regions bind to host Fcγ receptors (FcγRs) to signal the immune system. These FcγR interactions initiate and regulate antibody-mediated effector functions, which are crucial for host immunity, the effectiveness of therapeutic monoclonal antibodies, and IgG-mediated pathologies. FcγRs include both activating and inhibitory receptors, and the relative binding affinity of the IgG Fc region interacting with FcγRs that produce opposing signals is a key determinant of immune responses. Extensive research is devoted to understanding and manipulating FcγR interactions to reveal their fundamental biological activities and develop therapeutic monoclonal antibodies with tailored effector functions.

(Data source: Delidakis G, et al. Annu Rev Biomed Eng. 2022)
On April 1, 2025, researchers from Emory University in the United States published an article titled " Mapping affinity and allostery in human IgG antibody Fc regionFcγ receptor interactions " on bioRxiv. The researchers used a saturation mutation library of the Fc region and used the human IgG1 Fc region to determine the effective affinity of more than 98% of all possible single-point amino acid substitutions for all human FcγRs, as well as the most common FcγR polymorphisms. A comprehensive analysis of Fc amino acid variations that determine Fc stability, positive control of FcγR binding, and short-range and long-range allosteric control of FcγR binding was performed. The relative activation and inhibitory effector function capabilities of almost all possible single-point Fc mutations were also predicted.
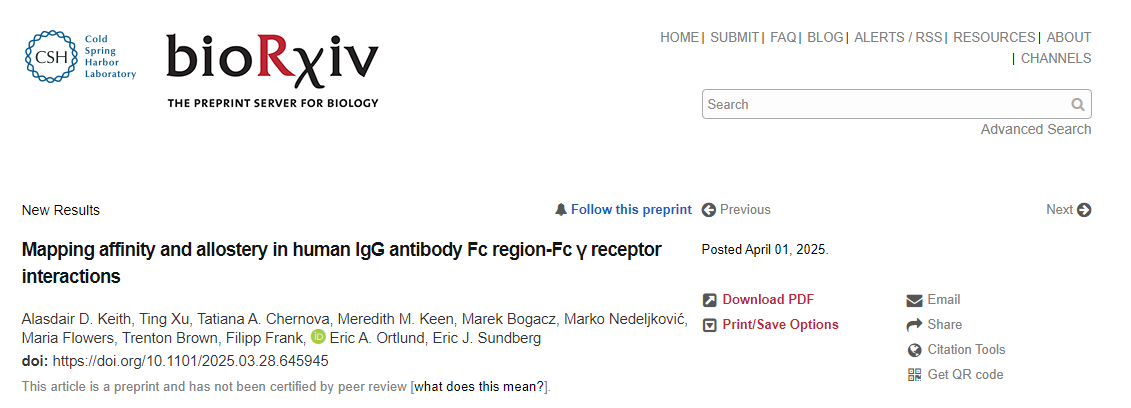
Establishment of a mammalian cell surface display platform for IgG1 Fc region
A lentiviral expression system was modified to contain the hinge and Fc regions of human IgG1 (including residues 216-447). In this construct, IgG1 216-447 is framed by an N-terminal signal peptide (SP) derived from IgG4 and a C-terminal transmembrane (TM) region from the platelet-derived growth factor receptor (PDGFR), with a Myc tag inserted between them to monitor expression. This construct was cloned into a lentiviral expression plasmid containing a 3' internal ribosome entry site (IRES) and green fluorescent protein (GFP), with GFP serving as a selectable marker.
To validate this system, a stable cell line expressing IgG1 216-447 on the surface of mammalian HEK293T cells was developed and human FcγR binding was assessed. Cells were incubated with varying concentrations of individual FcγRs and subsequently stained with fluorescently labeled streptavidin and anti-Myc antibodies to quantify FcγR binding and IgG1 216-447 cell surface expression, respectively. The KD values for the Fc-FcγR interaction of surface-displayed IgG1 determined in this manner are consistent with published data derived from surface plasmon resonance (SPR) analysis of the same Fc-FcγR interaction. When expressed on the HEK293T cell surface, each Fc cluster of IgG1 216-447 pairs as a homodimer, enabling FcγR binding.
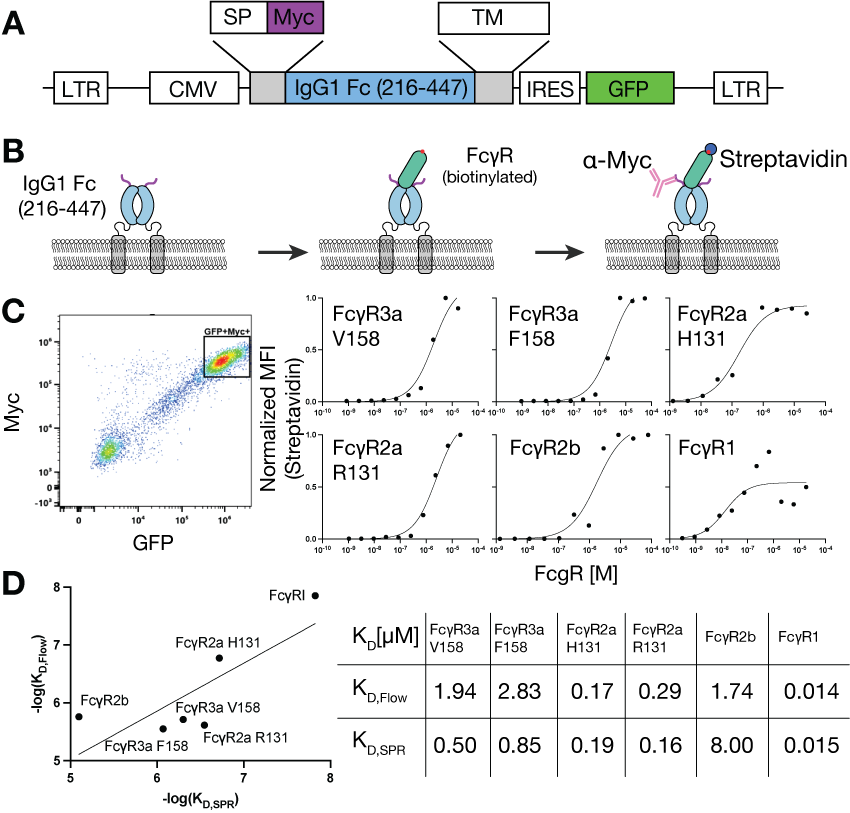
Cell Surface Display of an IgG1 Fc Deep Mutation Scanning Library
The researchers designed a site-saturated library covering all possible amino acid substitutions in IgG1 216-447 while retaining the wild-type surface-displayed flanking regions. A 15-nucleotide random barcode was inserted immediately after the stop codon of IgG1 216-447 by PCR amplification. PacBio long-read sequencing generated a lookup table linking unique barcodes to amino acid variants, achieving 98.9% mutation coverage. Only two positions (242 and 431) were missing due to lack of mutation in the input library. Each variant was associated with an average of nine different encoded barcodes, with the vast majority of variants being single-site substitutions.

IgG1 Fc mutations that inhibit cell surface display are primarily located in the core region of the CH2 domain
Fc mutations can alter protein stability. The CH2 domain has a significantly greater number of sites that negatively impact Fc cell surface display than the CH3 domain (thirteen versus two). Furthermore, each of these sites in the CH2 domain is much less tolerant of substitution with different amino acids than sites in the CH3 domain. The CH2 domain typically exhibits the lowest melting point among the cooperatively folding units (single Ig domains) within a monoclonal antibody, and increasing its stability is highly valuable for designing therapeutic monoclonal antibodies with improved biophysical and developability properties.
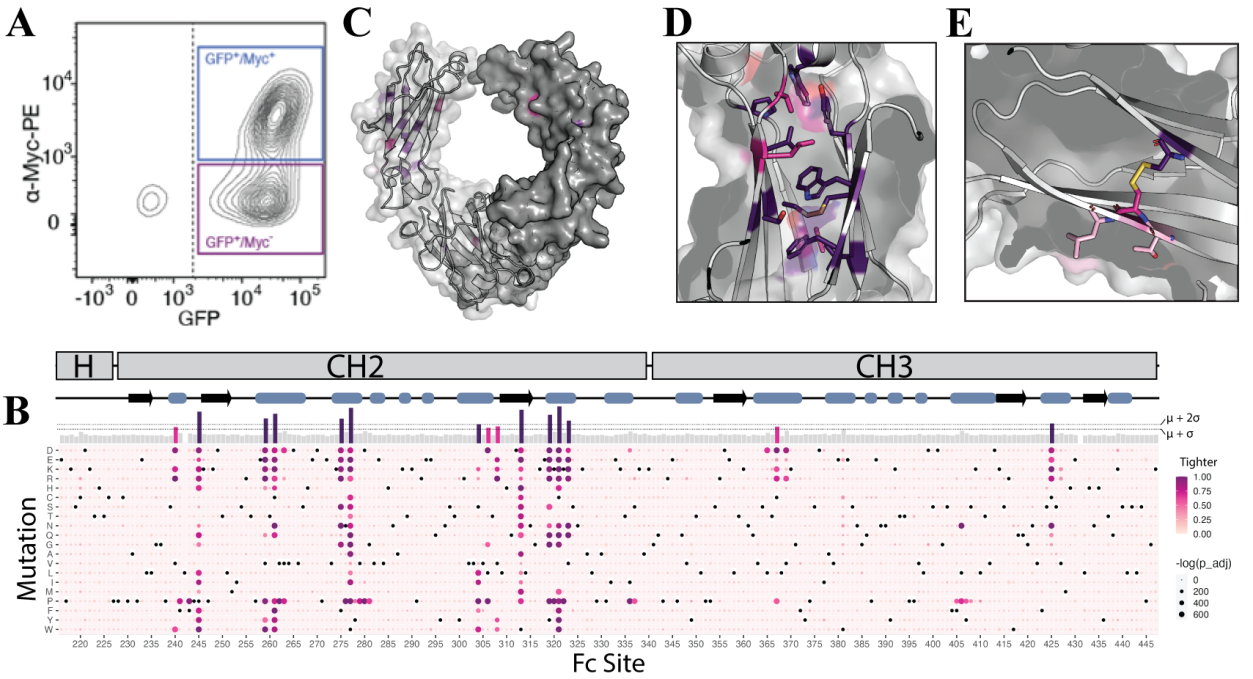
Mapping IgG1 Fc mutations leading to tighter and weaker FcγR binding
An IgG1 Fc deep mutational scanning library was expressed in HEK293T cells and screened against GFP+/Myc+ cells. All mutations at Asn297 abolished Fcγ-R3a-158v binding, with all non-asparagine amino acids at this position exhibiting high binding scores. Asn297 is linked to an N-glycan that is critical for Fc-γ-R binding. Mutations in IgG1 Fc residues that form the FcγR3a-158v interface were associated with weaker binding, with a number of residues distal to the Fc-FcγR3a-158v interface exhibiting significantly tighter and weaker binding, suggesting that a range of allosteric effects can lead to both increased and decreased binding affinity for FcγR3a-158v.

Effective affinity of IgG1 Fc single-site amino acid substitutions for FcγRs
High-resolution deep-scan titration analysis revealed four contiguous sequence-based regions within the human IgG Fc where mutations generally resulted in reduced binding to all human FcγRs. These regions, encompassing approximately positions 230-240, 262-272, 291-300, and 323-331, encompass the lower hinge region and three Fc loops, and contain at least some residues that directly contact FcγRs. Two Fc regions that generally resulted in stronger FcγR binding were located at positions 370-379 and 394-407, respectively. Both sites are located within the Fc methyl domain, extending from the CH2-CH3 junction to the CH3-CH3 interface. Fc regions whose mutations typically reduced binding affinity were located at or near the Fc-FcγR interface, while those whose mutations typically enhanced binding were located further away from the Fc-FcγR interface.

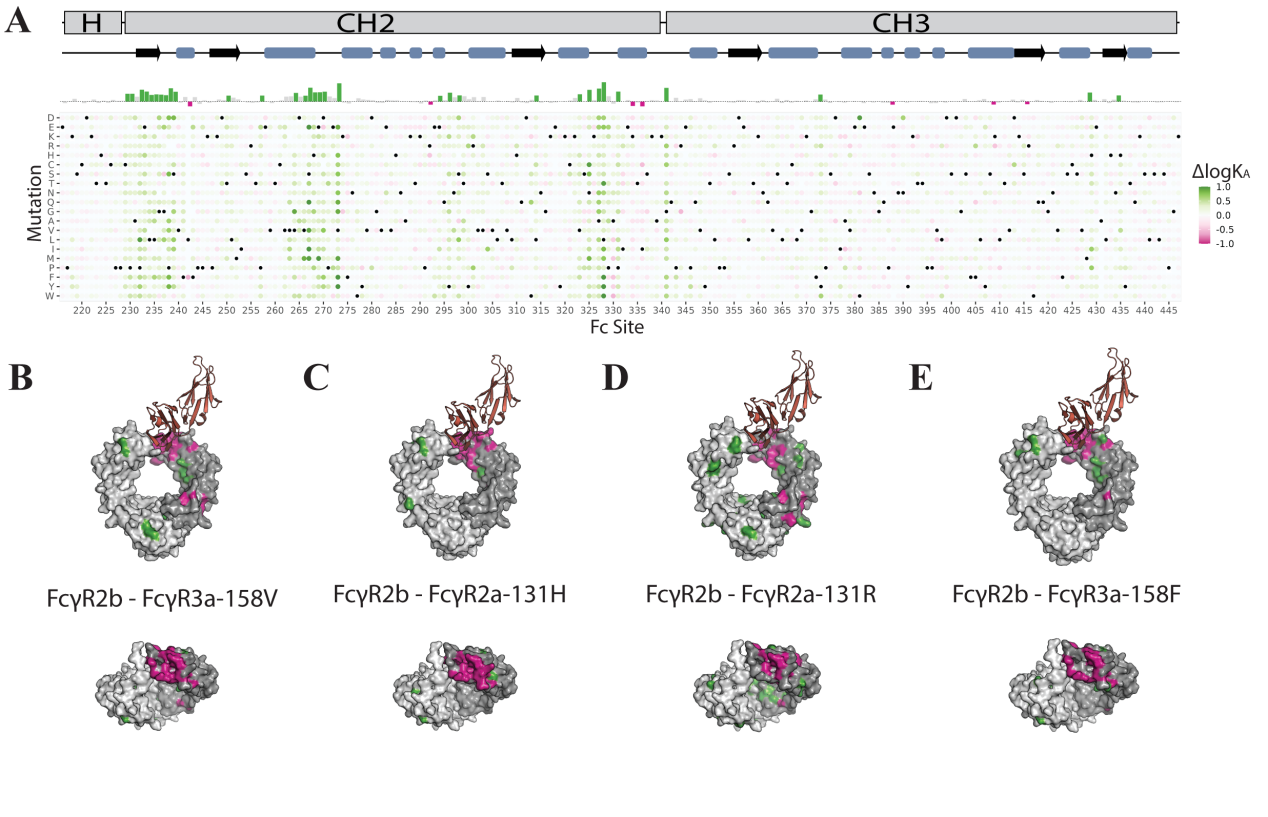
Comparison of the effects of Fc mutations on all low-affinity FcYRs
A key parameter for Fc mutations driving antibody-mediated effector functions is their relative affinity for activating (FcγR2a and FcγR3a) and inhibitory (FcγR2b) low-affinity FcγRs. This is often expressed as an affinity ratio, defined as the affinity for an inhibitory FcγR relative to the affinity for an activating receptor, or the I:A ratio. Because FcγR2b is the only inhibitory FcγR, mapping the differential effective affinity between activating FcγRs and FcγR2b onto their respective activating Fc-FcγR complexes reveals that the most activating (i.e., lowest I:A) Fc mutants are generally clustered at the Fc-FcγR binding interface, while the most inhibitory (i.e., highest I:A) Fc mutants are dispersed throughout the Fc and generally located away from the Fc-FcγR interface.
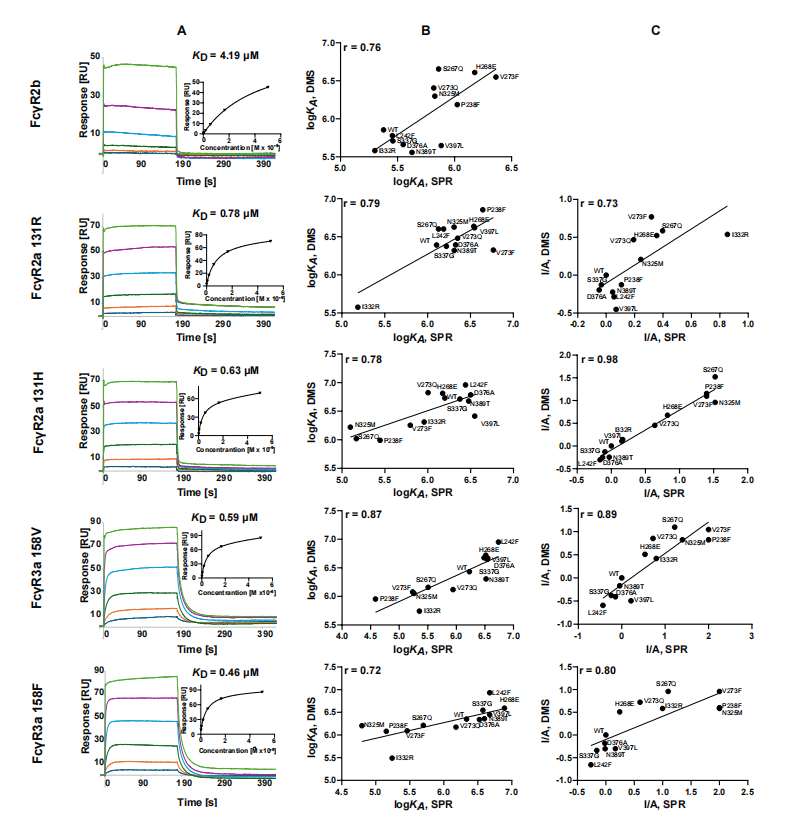
Biophysical validation of deep mutational scanning data
Deep mutation scanning data were verified by surface plasmon resonance (SPR) and differential scanning fluorimetry (DSF), showing that the predicted binding affinity of Fc to FcγRs and the effect of Fc mutations on stability were highly consistent with experimental measurements, confirming the reliability and accuracy of the DMS data.
Allosteric effects on Fc-FcγR binding are restricted to specific regions of the Fc domain
The largest influence on FcγR binding occurs within a layer 50-60 Å from the center of gravity of the receptor, encompassing the CH2-CH3 interface residues on each Fc monomer and the receptor-facing residues at the Fc dimer interface. These residues are crucial to the structure of the Fc region, and the observed changes in binding affinity suggest that these structural changes are exerting allosteric effects on the Fc-FcγR binding interface. Within this 50-60 Å layer, the average ΔlogKA effect is either positive or weakly negative, suggesting that this layer can be used to engineer Fc variants with varying affinities to target activating and inhibitory FcγRs.
D376T was most effective in improving activation and forms part of a larger network of residues that could be mutated to improve activation (S375W, D376T, and P395V). These residues all face the receptor and are close to the interface between the two Fc chains. T307 was the only residue that met both criteria: conversion to cysteine improved activation, while conversion to phenylalanine or leucine improved inhibition. This suggests that this residue is a promising target for engineering. T307 is located at the interface of the CH3 and CH2 domains, suggesting that this part of the Fc exerts allosteric control on receptor binding. All 18 mutations within the 20-30 Å layer improved inhibition, while none improved activation, suggesting that residues in the Fc binding interface may have evolved specifically to activate receptor binding rather than inhibit it.

Synergistic effects of combined Fc mutations
The synergistic effects of IgG1 Fc mutation combinations on FcγR binding were generally weak, with most combinations exhibiting near-additive effects and median synergistic effects close to zero, indicating that most mutations independently affect FcγR binding. However, some mutation combinations exhibited significant positive or negative synergistic effects, with the effects of these mutation combinations significantly deviating from additive effects, providing potential optimization targets for Fc engineering.

Summarize
The interaction between the IgG1 Fc and FcγRs is influenced by multiple factors, including the direct binding interface and allosteric effects. Modulating the relative affinity of the Fc for activating and inhibitory FcγRs can modulate the I:A ratio, which is crucial for designing therapeutic antibodies with specific immune effector functions. By modulating the intensity and type of immune responses, it has important implications for treating a variety of diseases, such as cancer and autoimmune diseases. The comprehensiveness and systematic nature of deep mutational scanning (DMS) enabled the evaluation of the effects of nearly all possible single-site mutations in the IgG1 Fc and approximately 1% of all possible double mutations on all human FcγRs. This data provides the most comprehensive analysis of Fc-FcγR interactions to date and offers unprecedented insights into how the interactions between the Fc region and activating and inhibitory FcγRs are regulated by allosteric effects. However, this approach also has limitations, as it does not include all possible combinations of mutations and relies on the endogenous glycosylation machinery of mammalian cells, limiting the diversity of Fc glycosylation patterns. Future exploration requires the combination of more complex libraries and glycosylation engineering.
