Background
The emergence and rapid spread of multidrug-resistant Botrytis cinerea poses a significant challenge to the quality and safety of agricultural products and the efficient use of pesticides. There is an urgent need to identify previously unknown fungicides and targets as alternative treatments against infections associated with B. cinerea.

On March 12, 2025, a team led by Liu Yingqian from the School of Pharmacy at Lanzhou University published a study titled "Indoloquinoline alkaloid neocryptolepine derivative inhibits Botrytis cinerea by targeting thiamine thiazole synthase" in Sci Adv. In this study, compound Z24 demonstrated efficacy against all tested plant pathogenic fungi. Using drug affinity response target stability (DARTS), cellular thermal shift assay (CETSA), and surface plasmon resonance (SPR) assays, Z24's target protein was identified as thiamine thiazole synthase (Bcthi4). Molecular docking and enzyme activity experiments demonstrated that Z24 could affect Bcthi4 function. Finally, mechanistic studies revealed that Z24 inhibits thiamine biosynthesis by binding to Bcthi4 and inducing upregulation of alternative splicing of the Bcthi4 gene. By targeting Bcthi4, Z24 has the potential to be developed as a previously unknown anti-B. cinerea drug candidate.
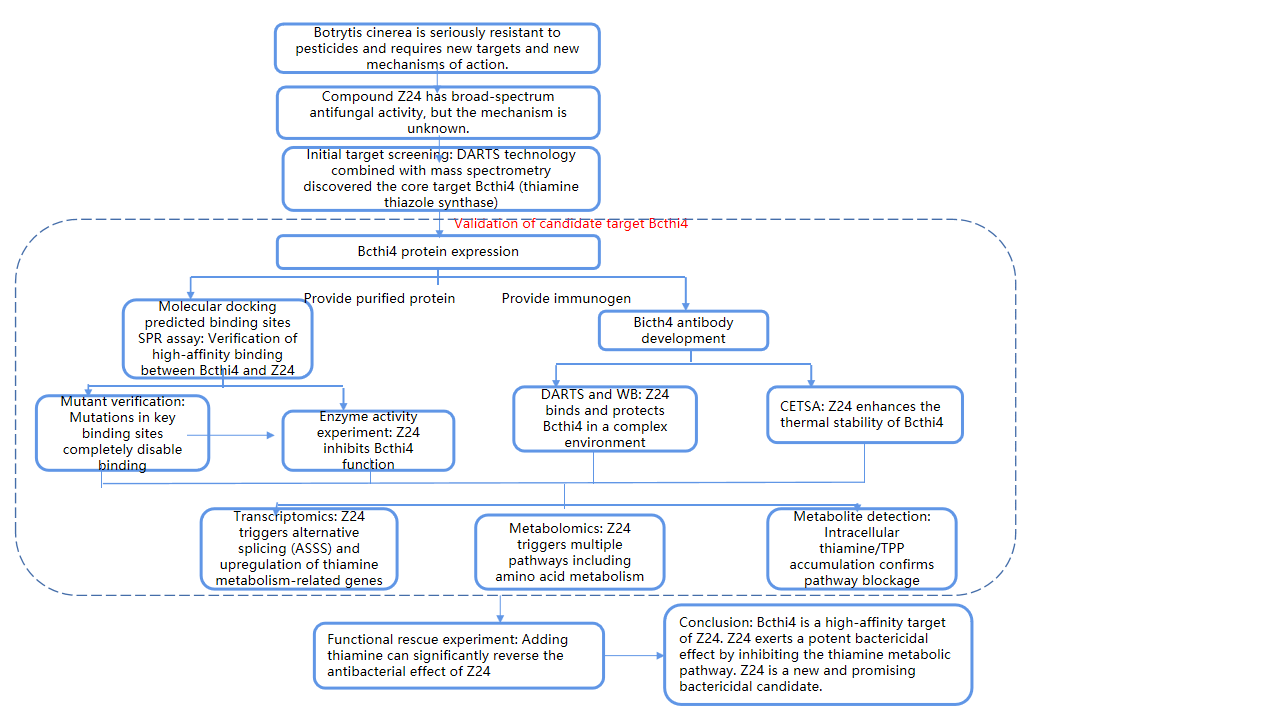
This research not only provides a novel solution for combating pesticide resistance but also serves as a classic example of leveraging modern biotechnology to decipher drug mechanisms of action. This study systematically validated the target of Z24 by combining protein expression, antibody development, and SPR detection.
Recombinant protein expression
Recombinant protein expression utilizes recombinant DNA technology to construct a target gene (exogenous gene) into a specific expression vector. This vector is then introduced into host cells, where the host cells' biological machinery allows the expression of the specific protein. The main steps include codon optimization, gene synthesis, expression vector construction, protein expression, and protein purification. Common expression systems include prokaryotic Escherichia coli, mammalian cells, insect cells, and yeast. Protein expression is crucial for functional research and drug discovery.
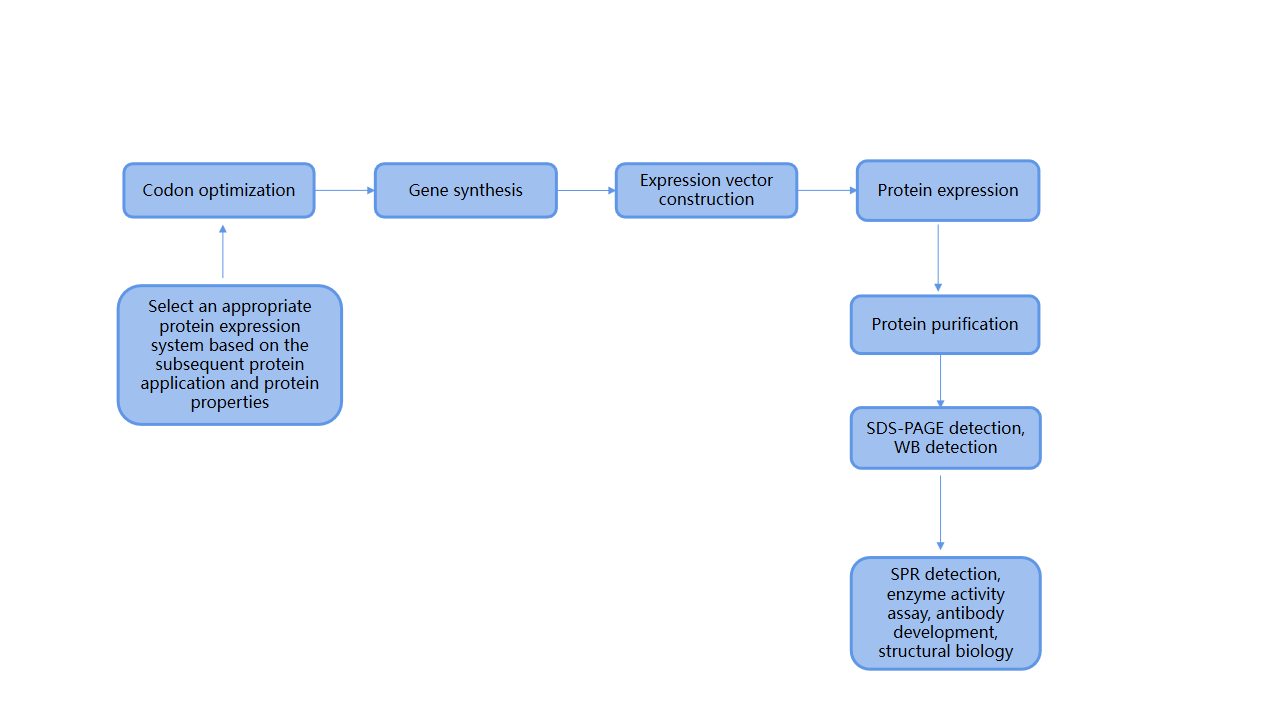
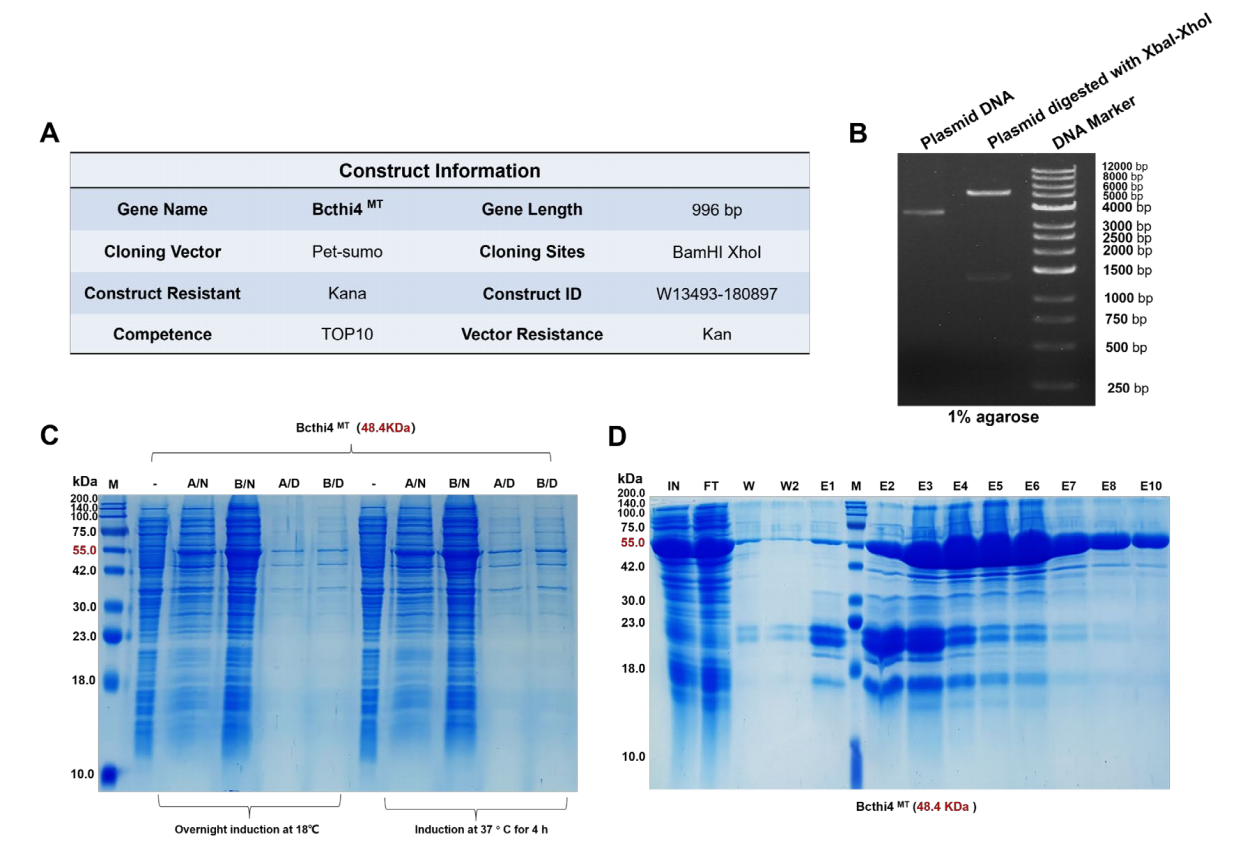
Case Study: Expression of Bcthi4 Wild-Type and Mutant Proteins
Bcthi4 protein was expressed to verify the mechanism of action of Bcthi4 and Z24, provide "bait" (ligand immobilized on the chip) for subsequent SPR binding experiments, and provide immunogen (antigen used to immunize animals to produce antibodies) for antibody development. To verify the predicted binding mode, Bcthi4 mutant proteins were generated: Met304, Thr232, and Thr231.

SPR affinity detection
Surface plasmon resonance (SPR), a new technology developed in the 1990s, uses the SPR principle to monitor interactions between ligands and analytes on biosensor chips. Through real-time analysis, it can quickly and easily monitor interactions between biomolecules, such as DNA and protein, protein and protein, drug and protein, antigen and antibody, receptor and ligand, and more. It has broad applications in life science testing, drug screening, food testing, and environmental monitoring. Compared to traditional methods, SPR eliminates the need for fluorescent or isotopic labeling, thus avoiding potential functional effects. It can visualize the entire binding and dissociation process in real time and provide quantitative kinetic and affinity data. Therefore, it is recognized by pharmaceutical companies and regulatory agencies worldwide as the gold standard for drug-target interaction research.
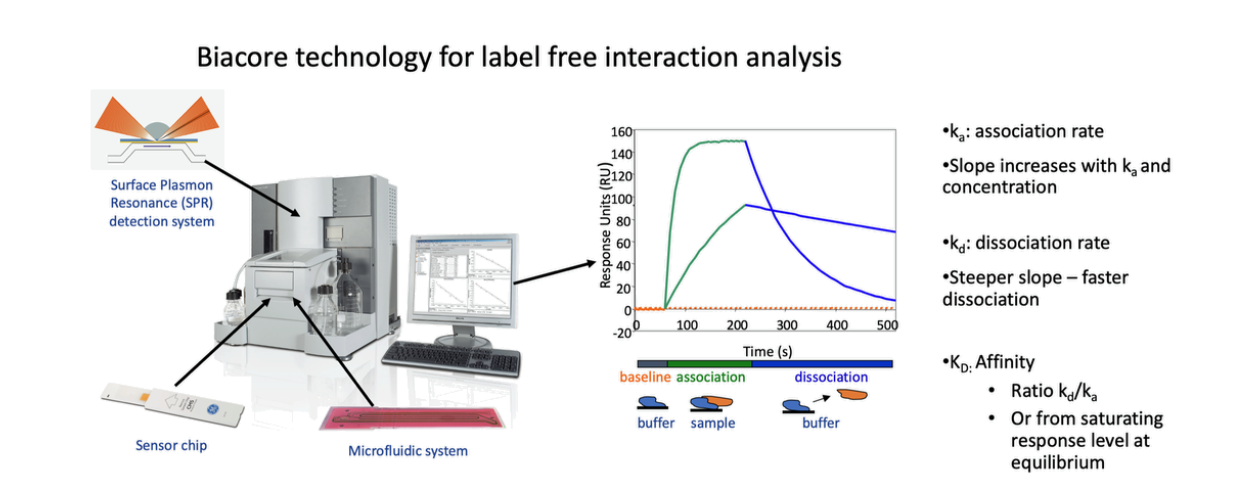
Case Study: SPR Affinity Detection of Bcthi4 and Z24
The study predicted the binding mode of Z24 and Bcthi4 using molecular docking (Autodock Vina). The docking results showed a binding energy of -8.185 kcal/mol. The authors further confirmed the binding of Z24 to Bcthi4 using surface plasmon resonance (SPR) experiments, measuring a KD value of 94.25 μM. To validate the predicted binding mode, the authors used SPR to test the binding ability of Bcthi4 mutants: Met304, Thr232, and Thr231, to Z24. The results showed that these mutants completely abolished the binding of Z24 to Bcthi4. Bcthi4, as a thiazole synthase, plays a crucial role in the thiamine biosynthesis pathway.

Antibody Development
Antibody development is the process of producing highly specific antibodies capable of recognizing and binding to a target molecule (antigen) through techniques such as animal immunization or in vitro cell screening. Antibody development can be used to detect protein expression in samples (WB), locate the distribution of target proteins in tissues and cells (IHC/IF), study protein-protein interactions or DNA-protein interactions (Co-IP/CHIP), and serve as a detection antibody to validate drug-target protein binding in complex environments (DARTS/CETSA).
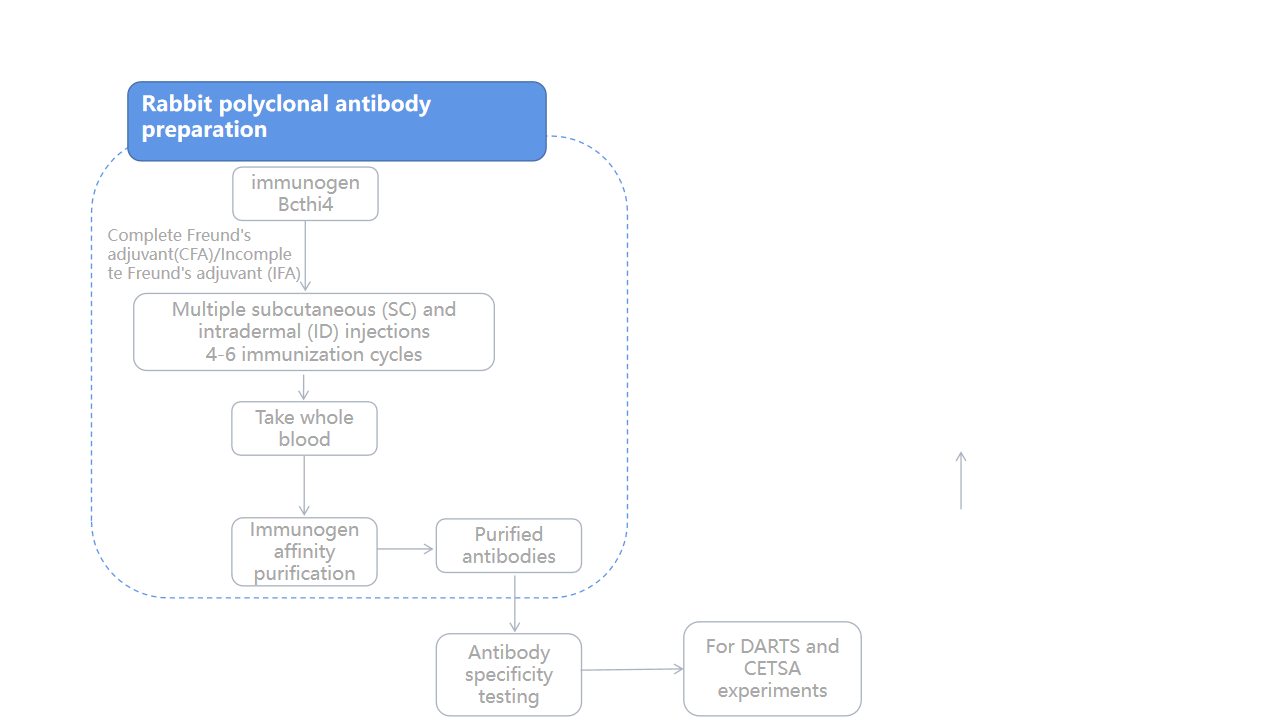
Case Study: Development of Bcthi4 Antibody
To verify the binding of Z24 to Bcthi4 , the authors successfully prepared a rabbit polyclonal antibody against Bcthi4 protein, which showed high sensitivity and specificity.
By analyzing the transmembrane region, signal peptide, and antigenicity of Bcthi4, we selected an appropriate expression region for immunogen expression using a prokaryotic expression system. This immunogen was injected multiple times into rabbits to stimulate their immune systems to produce Bcthi4-specific antibodies . The purified antibodies were then specifically tested for use as detection antibodies in Western blotting (WB) assays for DARTS and CETSA.

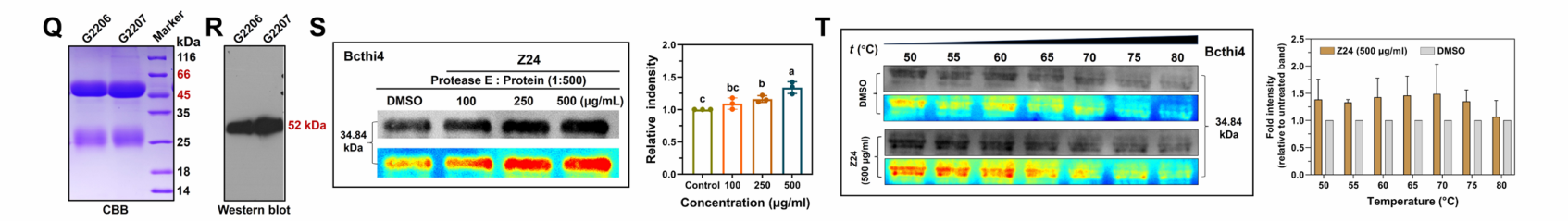
Summarize
This study used DARTS technology to identify Bcthi4, which is involved in thiamine biosynthesis, as a direct target of Z24. By expressing Bcthi4 recombinant protein and antibodies, its binding activity was further confirmed using DARTS, CETSA, and SPR. This study not only revealed a novel antifungal mechanism, but also provided new ideas for drug development based on the target Bcthi4. As demonstrated in this study, protein expression, antibody development, and SPR detection have become the core driving forces for innovative drug research and development. By integrating the above three technology platforms, Mabnus Bio is committed to providing universities, research institutes, and pharmaceutical companies with one-stop, high-quality target validation and bioanalysis solutions to facilitate original innovation.
