Sialic acid-binding immunoglobulin-like lectin 8 ( SIGLEC8) is a transmembrane protein that primarily functions as an inhibitory receptor, acting like a brake on the immune system. By recognizing "self" markers (sialic acid), SIGLEC8 prevents the excessive activation and proliferation of these cells, thereby helping to limit the intensity and duration of inflammatory and allergic reactions and prevent damage to the body.
Expression distribution of SIGLEC8
SIGLEC8 is specifically expressed on blood cells, namely basophils, mast cells, and eosinophils, which are key effector cells in allergic reactions (such as asthma and atopic dermatitis) and certain inflammatory diseases.
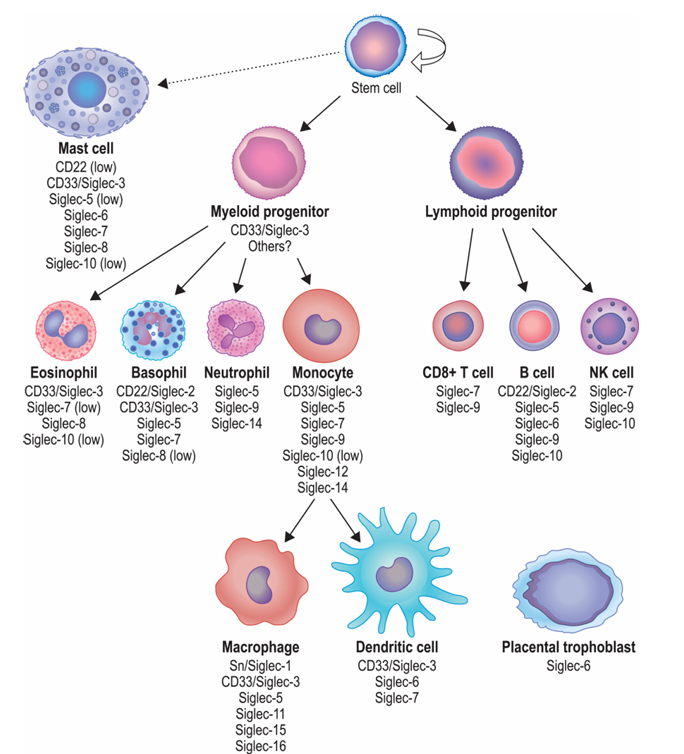
(Data source: O'Sullivan JA , et al. J Leukoc Biol . 2020)
The structure of SIGLEC8
SIGLEC8 is encoded by the SIGLEC8 gene and has a length of 499 aa and a molecular weight of approximately 54 It is a type I transmembrane protein of kDa, composed of three parts: the intracellular region, the transmembrane region and the extracellular region, and generates multiple isoforms (such as long SIGLEC8-b, short SIGLEC8-a/c/d/f, and soluble SIGLEC8-e) through selective splicing.
Extracellular region (17-363 aa): a) N-terminal V-set Ig-like domain: This is the core domain where the sialic acid binding site is located. It specifically recognizes and binds to glycosylated ligands containing sialic acid. This binding is specific, with SIGLEC8 preferentially binding its ligand, 6' - sulfo-sialyl Lewis X. b) Two Ig-like C2 domains: Located behind the V-set domain, they primarily provide support and extension, enabling the V-set domain to more flexibly access glycosylated ligands on the cell surface.
Transmembrane region (364-384 aa): composed of an α-helix, its main function is to anchor the receptor on the cell membrane and transmit conformational changes in the extracellular region to the intracellular region.
The cytoplasmic region (385-499 aa) is key to its signal transduction function. The immunoreceptor tyrosine-based inhibitory motif (ITIM) is central to SIGLEC8's ability to transmit inhibitory signals. When tyrosine residues within the ITIM are phosphorylated, they recruit and activate protein tyrosine phosphatases containing SH2 domains, thereby antagonizing and inhibiting immune cell activation signals, ultimately leading to the suppression of cellular function.
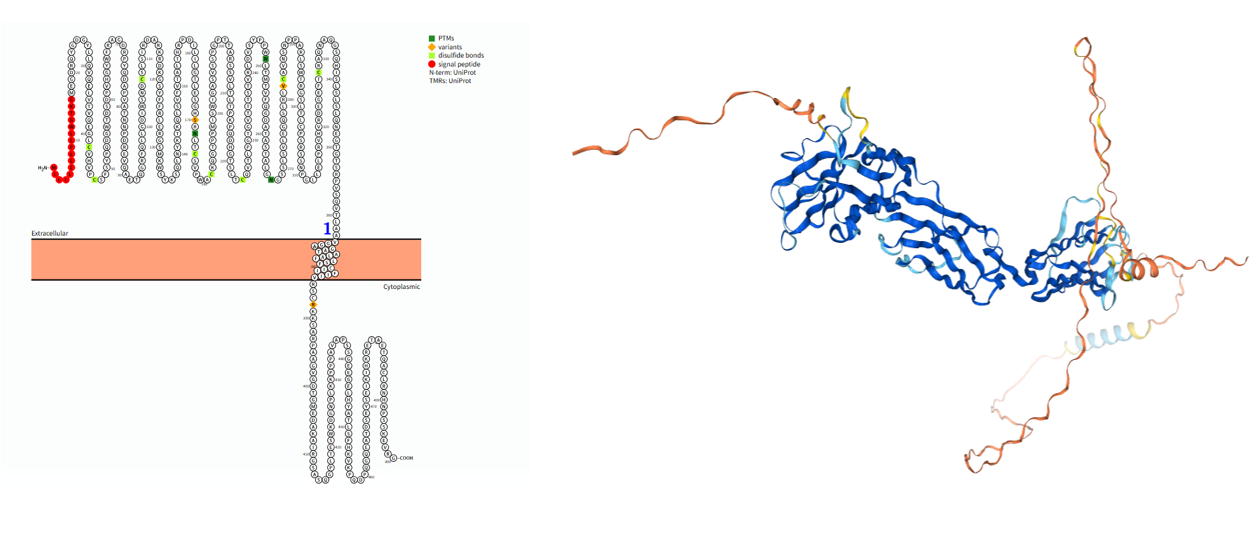
(Data source: protter)
SIGLEC8 signaling pathway
The structural basis of SIGLEC8's inhibitory function lies in the signaling pathway that harbors an immunoreceptor tyrosine-based inhibitory motif (ITIM) and a putative ITIM-like motif within its intracellular domain. While this pathway differs slightly in eosinophils and mast cells, the core mechanisms remain similar.
a) In eosinophils: Anti-SIGLEC8 antibodies first bind to cis-linked SIGLEC8 on the surface of eosinophils to form drug-loaded antibodies, triggering actin rearrangement and activating tyrosine kinases and PKC. Subsequently, the drug enters the cell through clathrin-mediated and lipid raft-mediated endocytosis, and finally releases the cargo in lysosomes.
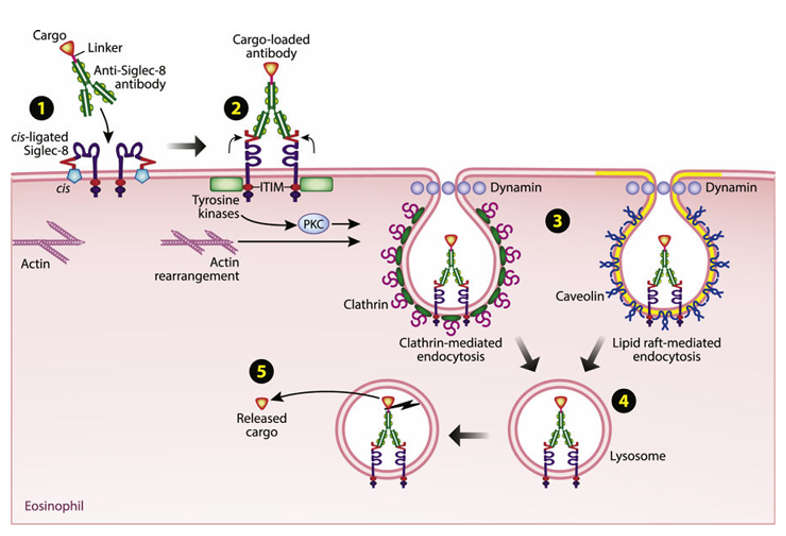
(Data source: O'Sullivan JA, et al. J Allergy Clin Immunol . 2018)
b) In mast cells: This pathway is activated more indirectly. On the one hand, JAK2 phosphorylates insulin receptor substrate (IRS) proteins, which recruit and activate PI3K. On the other hand, the gene transcript of STAT3 may also indirectly influence this pathway.
The primary function of SHP-1/2 is to inhibit activation signals mediated by FcεRI (high-affinity IgE receptor). They dephosphorylate key components of the FcεRI signaling pathway (such as Syk and LAT), thereby inhibiting calcium influx and the activation of inflammatory pathways such as NF-κB and MAPK. Ultimately, they strongly suppress mast cell degranulation (release of histamine, tryptase, etc.) and the production of inflammatory factors.
SIGLEC8 and disease
Severe asthma: Especially eosinophilic asthma, in which eosinophils infiltrate and degranulate the airways, leading to chronic inflammation and bronchoconstriction.
Eosinophilic esophagitis: Eosinophils accumulate in the esophageal wall, causing symptoms such as difficulty swallowing, food impaction, and regurgitation.
Alzheimer's disease: SIGLEC-8's effects are manifested in:
a) Under physiological conditions: Mainly located in cells, it maintains neuronal synaptic function and neural tissue homeostasis by regulating the activities of microglia and other cells.
b) In the state of neuroinflammation: it appears outside the cells, participates in the signal transduction after the activation of microglia and astrocytes, and is associated with pathological substances such as β-amyloid fibers, affecting the process of neuroinflammation.
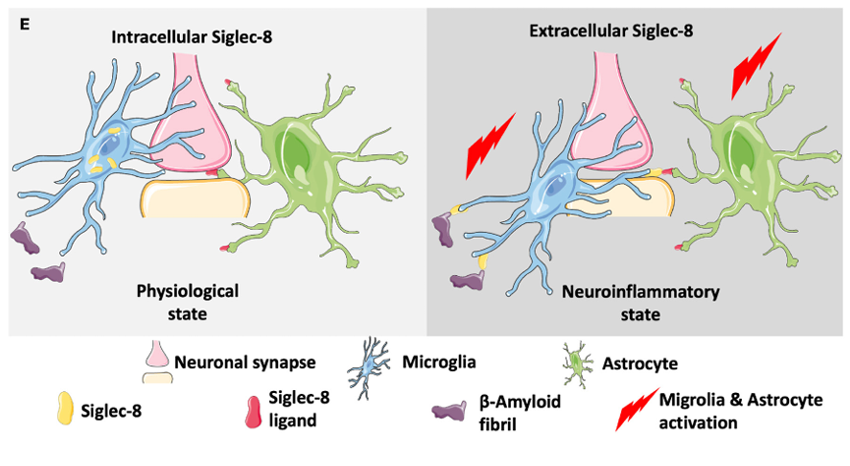
(Data source: Dhar C et al. Front Cell Neurosci. 2023)
Gastrointestinal: SIGLEC-8 functions through immune regulation and maintaining intestinal homeostasis. Expressed on the surface of immune cells such as eosinophils, it regulates cell activity by recognizing ligands. For example, it inhibits the release of mediators such as ECP and MBP from eosinophils, thereby modulating the intensity of the immune response and preventing excessive activation from causing tissue damage. In the intestine, it participates in maintaining barrier function and immune cell balance. By regulating the immune response, it helps maintain the normal physiological state of the intestine and reduces intestinal dysfunction and gut-brain interaction-related comorbidities caused by immune disorders.
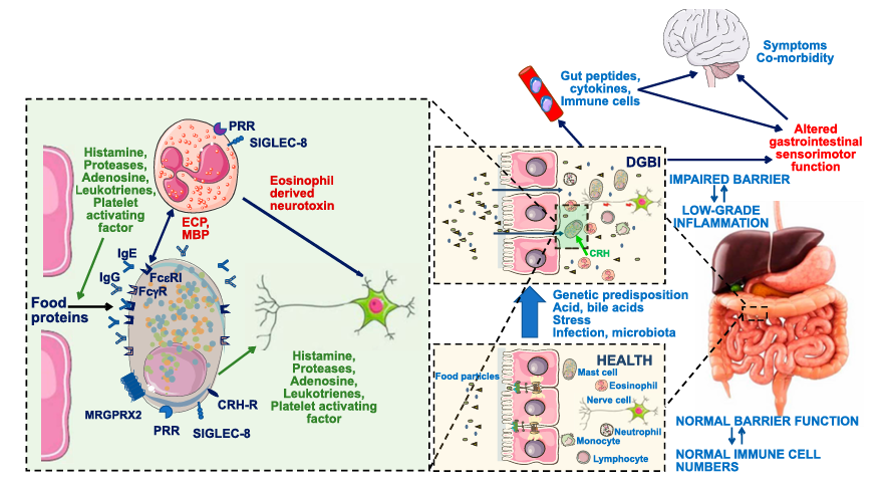
(Data source: Van den Houte K et al. Am J Gastroenterol. 2022)
Urticaria/Inflammatory and Allergic Diseases: SIGLEC8 acts as an inhibitory receptor. a) Inhibiting signal transduction: It prevents excessive mast cell activation and the release of inflammatory mediators such as histamine, tryptase, and leukotrienes, thereby reducing inflammatory responses such as bronchoconstriction and increased vascular permeability caused by these mediators. b) Balancing the immune response: It helps maintain immune homeostasis, preventing allergic or inflammatory diseases caused by abnormal mast cell activation, and plays a role in maintaining a balance in immune regulation.
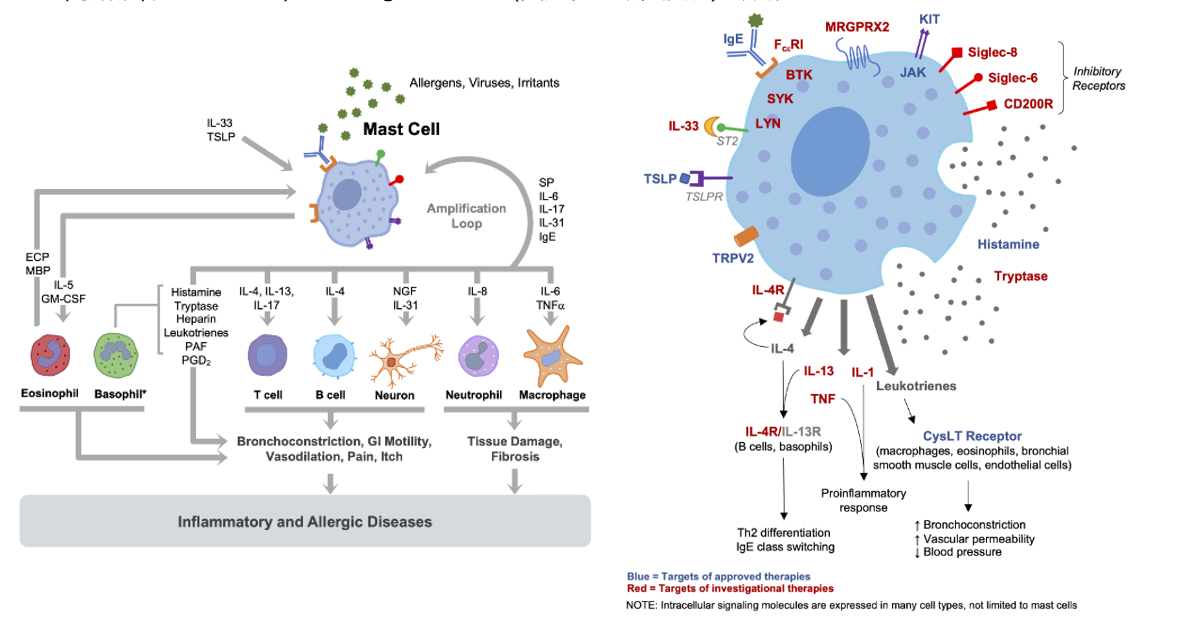
(Data source: Metz M, et al. Allergy. 2024)
Targeted therapy of SIGLEC8
The cell death mechanisms mediated by antibodies targeting Siglec-8 and its functional homolog Siglec-F differ significantly between humans and mice. In humans, antibody binding to Siglec-8 directly induces cell death through multiple intrinsic pathways, including those dependent on ROS, mitochondrial dysfunction, and caspase activation, accompanied by ADCC. Internalization is clathrin- and lipid raft/caveolin-dependent. In contrast, in mice, antibody binding to Siglec-F does not directly induce robust intrinsic cell death. Cell elimination relies primarily on effector mechanisms such as ADCC/ADCP, with internalization occurring in a clathrin-dependent but dynamin-independent manner. Repeated injections are required to enhance cell death.
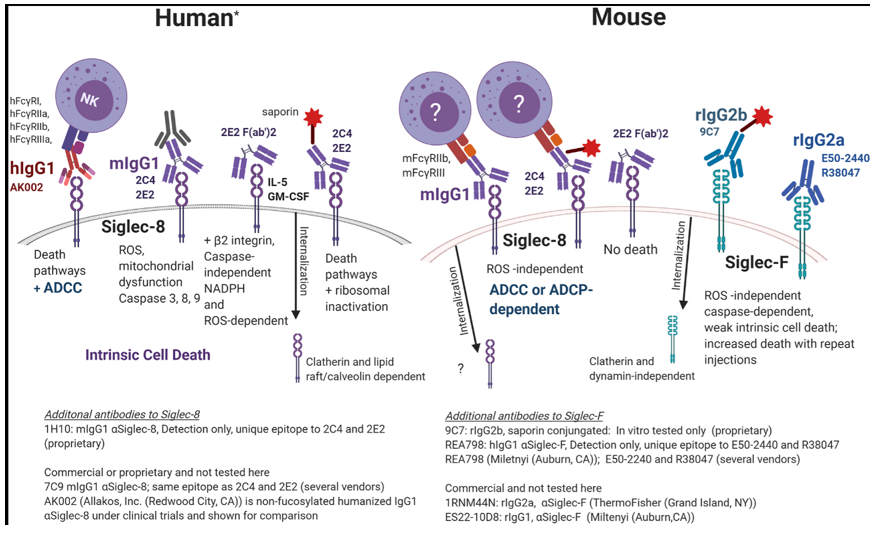
(Data source: Dhar C et al. Front Cell Neurosci. 2023)
Antolimab is a monoclonal antibody drug co -developed by Allakos that targets SIGLEC8. Its primary mechanism of action is as a SIGLEC8 inhibitor , inhibiting Siglec-8 receptor function and blocking its mediated immunosuppressive signaling pathway, thereby reducing mast cell activation and inflammatory responses. 1 It also has mast cell inhibitory effects, reducing the release of inflammatory mediators such as histamine and prostaglandins. It is used to treat chronic urticaria, eosinophilic esophagitis, and myeloid systemic mastocytosis. It was approved for Phase 3 clinical trials on June 18, 2020.
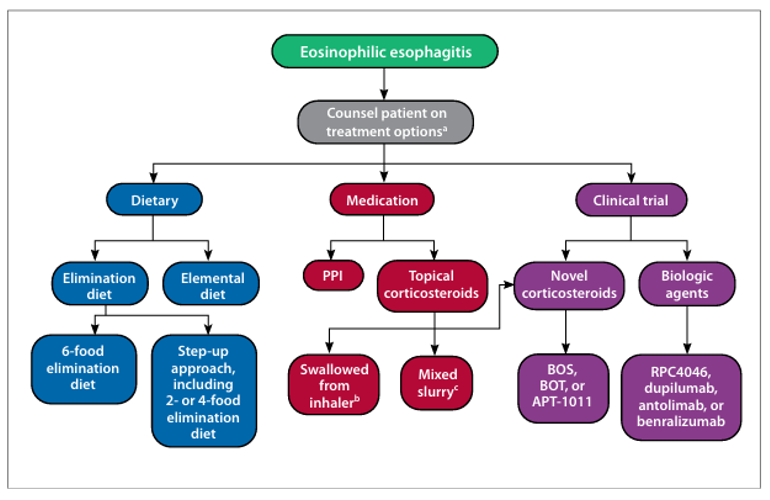
(Data source: Beveridge C, et al. Gastroenterol Hepatol (NY). 2020)
AK-001, a monoclonal antibody developed by Allakos, targets SIGLEC8. Its primary mechanism of action is to act as a SIGLEC8 inhibitor, modulating the activity of immune cells (such as eosinophils and mast cells), suppressing excessive immune responses and improving allergic diseases and chronic inflammatory pathologies through immunomodulatory effects. It is intended for the treatment of otolaryngology, respiratory, skin, and musculoskeletal diseases. Phase 2 clinical trials were approved on April 1, 2016, but progress has been delayed.
