Regulatory Protein Alpha ( SIRPA) is a type I transmembrane protein whose core function is to transmit the "don't eat me" signal through the SIRP A -CD47 axis, inhibiting macrophage phagocytic activity and inflammatory responses, thereby maintaining autoimmune tolerance and homeostasis. Targeting this pathway offers new avenues for the treatment of diseases such as cancer.
Expression distribution of SIRPA
SIRPA is generally highly expressed in the brain. It is detectable on myeloid cells but not on T cells. Lower levels are detected in the heart, placenta, lung, testis, ovary, colon, liver, small intestine, prostate, spleen, kidney, skeletal muscle, and pancreas.
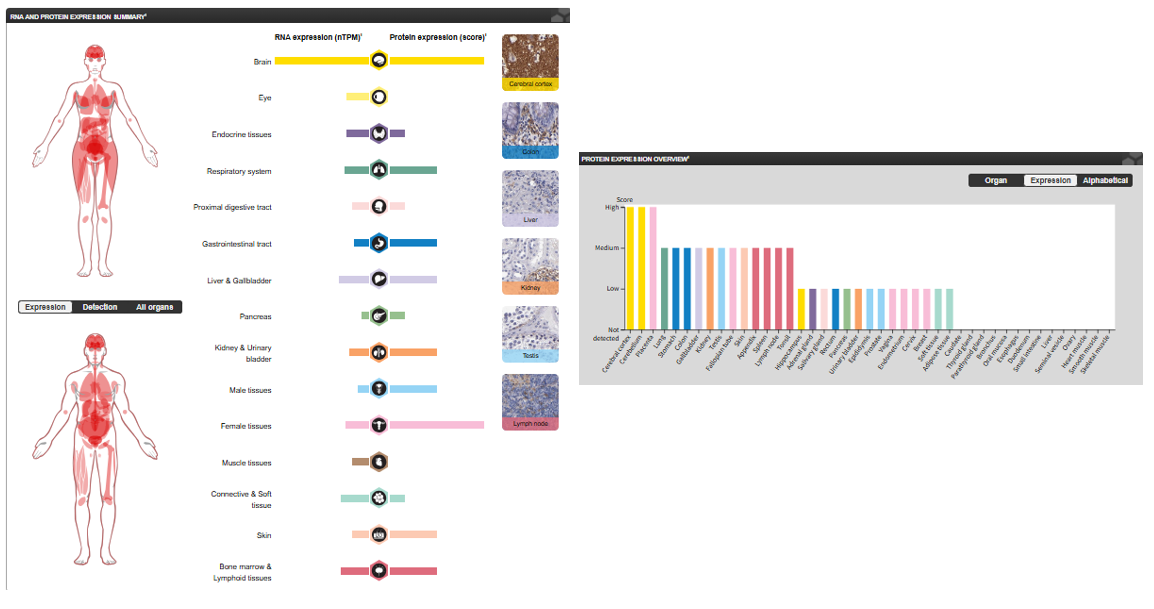
(Data source: Protein Atlas)
SIRPA 's structure
SIRPA is a type I transmembrane protein encoded by the SIRPA gene with a length of 504 aa and a molecular weight of approximately 54.9 kDa. It consists of three parts: the intracellular region, the transmembrane region and the extracellular region. The core function of SIRP A is to act as an inhibitory receptor, which transmits the " don't eat me " signal to the downstream through interaction with its ligand CD47.
Extracellular region (31-373 aa): Contains three immunoglobulin superfamily domains: V (variable region-like), C1 (constant region 1-like), and C2 (constant region 2-like). The V domain is the core functional domain responsible for binding to the ligand CD47. The V domain of SIRPα is polymorphic among individuals, meaning its amino acid sequence varies. This may affect its binding affinity to CD47 and be associated with an individual's susceptibility to certain diseases or allograft rejection.
Transmembrane region (374-394 aa): composed of an α- helix, its main function is to anchor the receptor on the cell membrane and transmit conformational changes in the extracellular region to the intracellular region.
The cytoplasmic region (395-504 aa): Contains immunoreceptor tyrosine-based inhibitory motifs (ITIMs). Human SIRPα typically contains two ITIMs. Upon binding to CD47, the tyrosine residues within the ITIMs become phosphorylated, recruiting and activating SH2 domain-containing protein tyrosine phosphatases (such as SHP-1 and SHP-2). This is a key step in transmitting inhibitory signals.

(Data source: Che Z, et al. J Pharm Anal . 2025)
SIRPA signaling pathway
Signal source (ligand): CD47 is widely expressed in normal healthy cells, such as red blood cells, lymphocytes, etc.; many cancer cells also highly express CD47 to escape immune surveillance.
Receptor: SIRPA is mainly expressed on myeloid cells, such as macrophages, dendritic cells, and neurons.
Key effector molecules: SH2 domain-containing protein tyrosine phosphatases, mainly SHP-1 and SHP-2.

(Data source : Zhao K, et al. Front Immunol . 2024)
SIRPA and disease
Immune escape: Many cancer cells overexpress the ligand CD47, which binds extensively to SIRPα on myeloid immune cells such as macrophages , leading to overactivation of inhibitory signaling pathways. This strongly inhibits the phagocytic function of macrophages, allowing cancer cells to escape surveillance and clearance by the innate immune system, promoting tumor growth and metastasis. The SIRPα -CD47 axis has become a key cancer immune checkpoint.
In the field of gastrointestinal GI cancer: CD47-SIRPα is a key checkpoint in tumor immunity. Tumor cells overexpress CD47, which binds to SIRPα on the surface of immune cells such as macrophages, transmitting a "don't eat me" signal and enabling immune evasion.
Blocking this pathway with anti-CD47 antibodies, SIRPα-Fc fusion proteins, or bispecific antibodies can relieve this inhibitory effect, promote macrophage tumor phagocytosis through ADCP, and enhance NK cell-mediated ADCC. Simultaneously, tumor antigen release activates dendritic cells and T cells, triggering a specific immune response. Furthermore, CAR-T/M cell targeting of CD47 and complement activation also enhance anti-tumor efficacy.
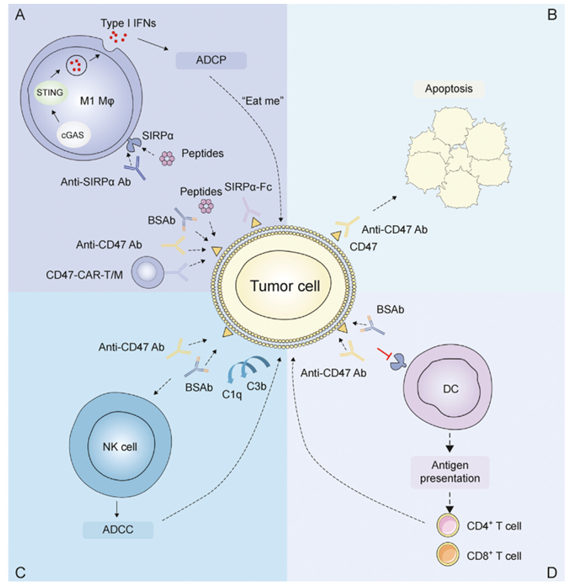
(Data source : Che Z, et al. J Pharm Anal. 2025)
In the field of high-risk neuroblastoma: Macrophages (M1-M1 and tumor cell-macrophage) transmit "don't eat me" signals through CD47-SIRPA binding, inhibiting phagocytosis and aiding tumor escape. Neuroblastoma (NB ) cells transmit "don't eat me" signals through CD47 binding to macrophage SIRPA, directly inhibiting phagocytosis by M1 macrophages (M1 is a pro-inflammatory type). Anti-CD47 antibodies block the binding of tumor cell CD47 to macrophage SIRPA, disabling the "don't eat me" signal. Furthermore, they bind to macrophage FcγRs, transmitting the "don't eat me" signal, activating M2 macrophages (M2 is an anti-inflammatory/pro-tumor type) to transform into a pro-phagocytic phenotype, enhancing phagocytosis of NB. Blocking CD47-SIRPA binding with anti-SIRPA F(ab')₂ restores the phagocytic function of M1 macrophages toward NB.

(Data source: Tang XX, et al. Curr Oncol. 2024)
In the field of colorectal cancer: a) Within the tumor microenvironment, activation of the unfolded protein response/endoplasmic reticulum-XBP1 (UPR/ER-XBP1) signaling pathway in tumor-associated macrophages (TAMs) induces cytokine production. These cytokines, by disrupting their self-recognition, inhibit macrophage phagocytosis of tumor cells. Consequently, TAMs promote colon cancer metastasis. b) Disrupting the UPR/ER-XBP1 signaling pathway or using anti- SIRPα antibody therapy may enhance anti-cancer capabilities in this hostile tumor microenvironment.
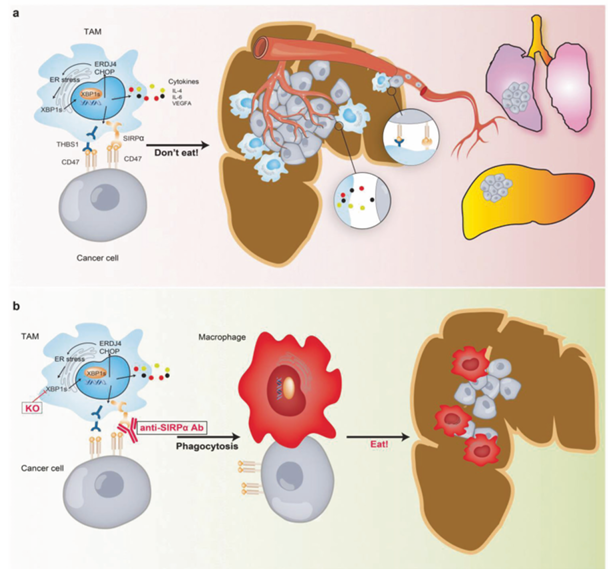
(Data source: Zhao Y, et al. Signal Transduct Target Ther. 2021)
Targeted therapy for SIRPA
The discovery of the CD47–SIRPα interaction (1999–2000) laid the foundation for self-recognition. Between 2007 and 2014, research focused on mechanisms of self/non-self recognition, culminating in the identification of calreticulin, SIRPα, and MHC-I/LIRB1 as phagocytic checkpoints. Since 2015, the field has rapidly expanded to encompass complement-dependent tumor killing, novel checkpoint molecules such as PD-L1, CD24/CD22/STC1/IGSF8/ENPP1/TREX1, as well as synergistic therapeutic strategies and regulation of the cGAS/STING pathway. These advances systematically revealed the regulatory network underlying phagocytosis, laying a solid foundation for cancer immunotherapy targeting phagocytic checkpoints.
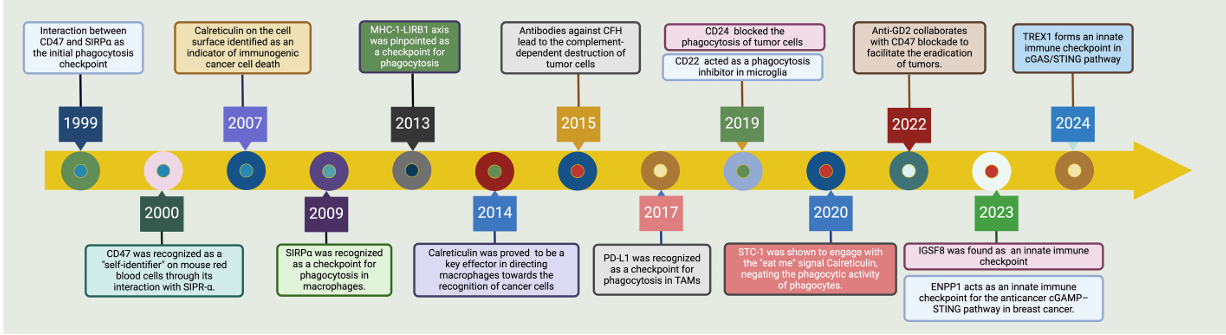
(Data source: Zhao K, et al. Front Immunol . 2024)
BSI-082 is a monoclonal antibody drug targeting SIRPA, developed by Biosion, Inc. Its primary mechanism of action is as a SIRPA inhibitor and macrophage stimulator, inhibiting SIRPα and directly stimulating phagocyte function. It is intended for the treatment of locally advanced malignant solid tumors. It was approved for Phase 1 clinical trials on August 7, 2025.
ELA-026, developed by Electra Therapeutics, is a monoclonal antibody targeting SIRPA, SIRPβ1, SIRPγ, and tubulin. Its primary mechanism of action is as a SIRPα modulator, simultaneously regulating SIRPβ1 and SIRPγ, and inhibiting tubulin function. It is intended for the treatment of hemophagocytic lymphohistiocytosis (sHLH). It was approved for Phase 3 clinical trials on May 19, 2022.
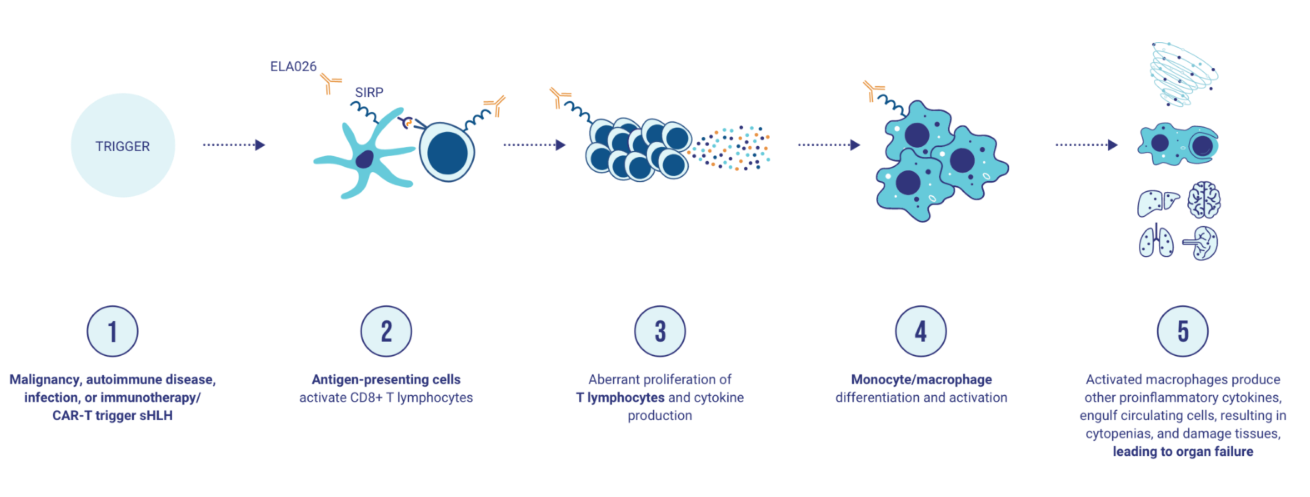
(Data source: electra official website)
BI-770371 is a monoclonal antibody developed by Boehringer Ingelheim GmbH that targets SIRPα and Chk. Its primary mechanism of action is as a SIRPα inhibitor. By inhibiting SIRPα and Chk kinases , it enhances antibody-dependent cellular cytotoxicity (ADCC) and stimulates T lymphocyte activity for the treatment of advanced solid tumors. It was approved for Phase 2 clinical trials on February 3, 2025.
LM-101, a monoclonal antibody targeting SIRP α, was developed by LaNova Medicines Ltd. Its primary mechanism of action is to specifically inhibit SIRP α, blocking its interaction with CD47 and enhancing anti- tumor immunity. It is used to treat hepatocellular carcinoma and obesity. It was approved for Phase 2 clinical trials on January 11, 2023.
JY47, a monoclonal antibody targeting SIRP α, is being developed by Hangzhou Jiuyuan Gene Biopharmaceutical Co., Ltd. Its primary mechanism of action is to target SIRP α, blocking the CD47 - SIRP α signaling axis to activate the immune system. It is intended for the treatment of advanced malignant solid tumors. Phase 1 clinical trials were approved on July 1, 2025.

(Data source: Sang YS , et al. Industrial Crops and Products . 2025)
ADU-1805, developed by Sairopa BV, is a monoclonal antibody targeting SIRP α. Its primary mechanism of action is that the monoclonal antibody targets SIRP α, blocking the immunosuppressive signaling pathway. It is intended for the treatment of advanced malignant and metastatic solid tumors. It was approved for Phase 1 clinical trials on April 3, 2023.
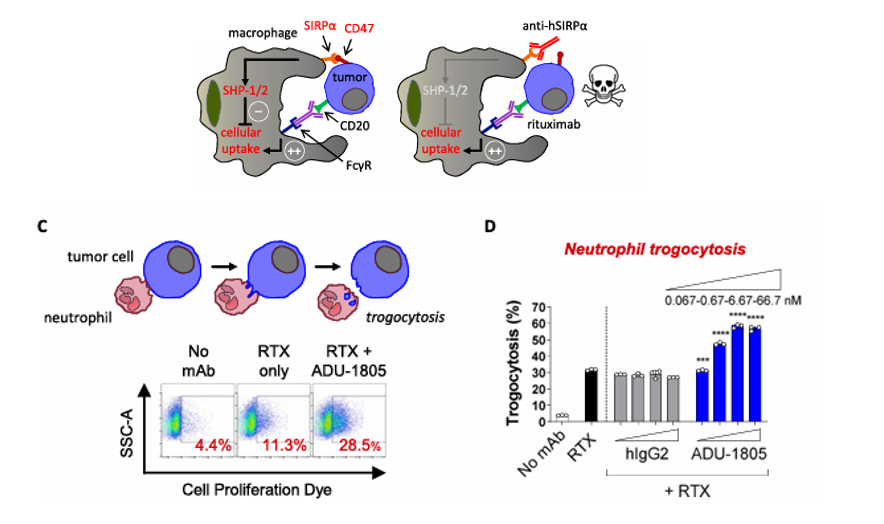
(Data source: Voets E, et al. J Immunother Cancer . 2019)
ES004-B5 is a monoclonal antibody targeting SIRP α, developed by Elpiscience Biopharma, Ltd. Its primary mechanism of action is as a SIRP α inhibitor, intended for the treatment of tumors. It is currently in the drug discovery phase.

(Data source: Niu X, et al. Antib Ther . 2024)
