The FZD receptor family consists of seven transmembrane receptors, structurally belonging to the G protein-coupled receptor (GPCR) superfamily, but their signal transduction mechanisms are atypical. The FZD receptor family comprises ten members (FZD1-FZD10). FZDs are activated by binding to Wnt ligands, which regulate key processes such as cell proliferation, differentiation, and polarity. Abnormal FZD/Wnt signaling pathways drive tumorigenesis, progression, and metastasis, and mediate treatment resistance, making it a key therapeutic target of great interest in oncology.
FZD structure
FZDs belong to the F class of GPCRs, and the FZD receptor family shares a set of common functional domains that are crucial for Wnt binding and intracellular signal transduction. These receptors share a common structure, including an extracellular cysteine-rich domain (CRD), a hydrophilic linker region, a seven-transmembrane (7TM) domain, and an intracellular C-terminal region.
Extracellular cysteine-rich domain (CRD): A hallmark feature of the FZD receptor, crucial for binding to the Wnt ligand. This domain is stabilized by 10 conserved cysteine residues that form disulfide bonds, creating a structure that interacts with lipid-modified regions of the Wnt ligand, such as palmitoleic acid.
The seven-transmembrane (7TM) domain: It anchors the FZD receptor to the plasma membrane and facilitates the transduction of extracellular signals into intracellular responses. Wnt binding is transmitted via conformational changes.
The intracellular C-terminal region: It contains the PDZ binding motif and is key to recruiting signal transduction effectors such as DVL.
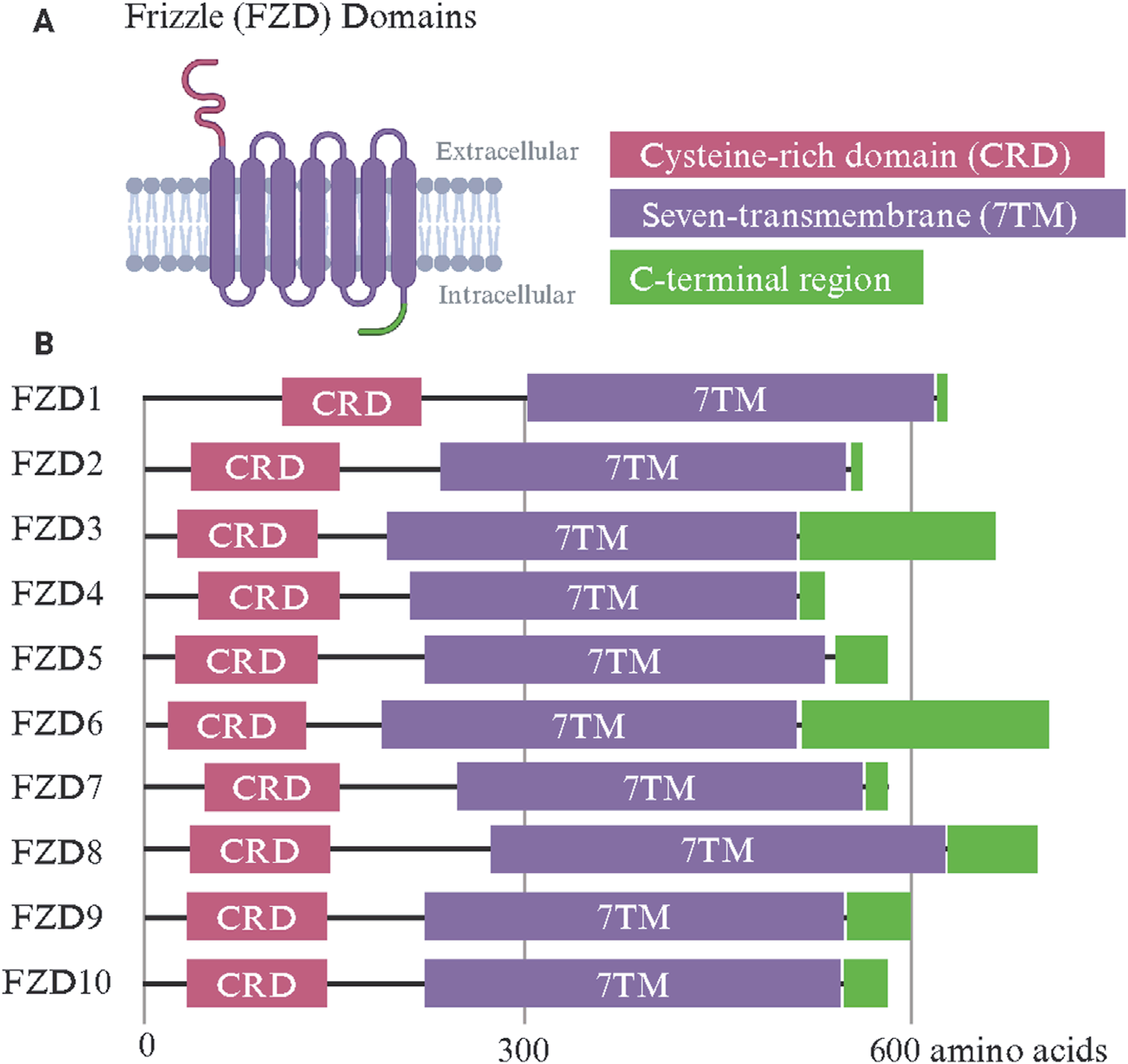
(Data source: Martinez-Marin D, et al. Front Cell Dev Biol. 2025)
Signaling pathways and regulation of FZD
FZD receptors are essential mediators of the Wnt signaling pathway, which controls key processes such as embryonic development, tissue homeostasis, and disease progression. These pathways are broadly classified into the classical Wnt/β-catenin pathway and non-classical pathways, including the Wnt/Ca2+ and PCP pathways.
Classic Wnt approach
In the classical Wnt signaling pathway, Wnt ligands that bind to the FZD and LRP5/6 co-receptors trigger the binding of DVL to the C- terminal domain of FZD. DVL recruits the β-catenin disruption complex via the TCF/LEF transcription factor, leading to β-catenin stabilization, nuclear aggregation, and expression of Wnt target genes.
Non-classical Wnt approach
In the non-canonical Wnt/PCP signaling pathway, Wnt ligands binding to the FZD and ROR/Ryk co-receptors trigger the formation of the FZD-PCP complex (i.e., Vangl, Prickle , Cels, DVL). DVL induces small Rho GTPase activation, cytoskeleton remodeling, and c-jun-mediated gene transcription. In the non-canonical Wnt/Ca2+ signaling pathway, Wnt ligands binding to the FZD and ROR/Ryk co-receptors trigger the formation of FZD.
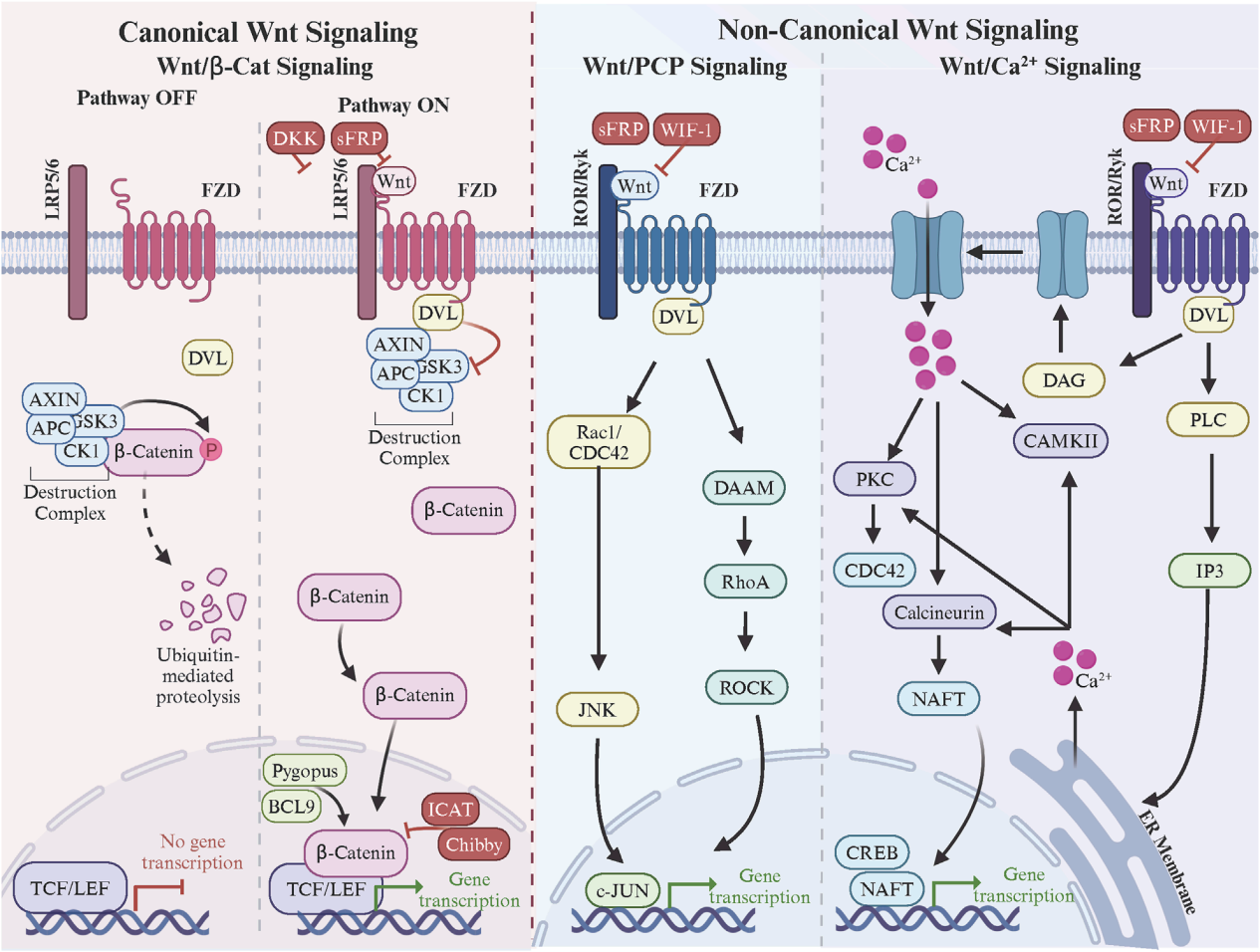
(Data source: Martinez-Marin D, et al. Front Cell Dev Biol. 2025)
FZD Expression and Role in Tumors
FZD1: One of the proteins expressed in the central nervous system, closely related to the development and evolution of the human nervous system. It plays a neuroprotective role.
FZD2: Expressed in various metastatic cancer cell lines.
FZD3: Primarily expressed in the nervous system, and also highly expressed in various tumors.
FZD4: Expressed in vascular endothelial cells and various tumors.
FZD5: Widely expressed in various tumors.
FZD6: Expressed in various tumors, especially associated with the non-canonical Wnt/PCP pathway.
FZD7: Highly expressed in various cancers, especially in stem cells.
FZD8: Highly similar to FZD5, expressed in various tumors.
FZD9: Primarily expressed in brain, muscle, testis, eye, skeletal muscle, and kidney tissues, playing a crucial role in maintaining the stable growth and differentiation of stem cell populations in blood, skin, and intestines.
FZD10: Absent in almost all normally developing healthy tissues; therefore, FZD10's high tumor specificity makes it a potentially highly sensitive tumor marker. Increased FZD10 expression may become an important indicator for human tumor diagnosis and malignancy determination.

(Data source: Liu HY, et al. Acta Pharmacol Sin. 2024)
The tumor microenvironment (TME) facilitates the bioavailability of Wnt ligands, the localization of FZD receptors, and the signal transduction of downstream receptors. Extracellular matrix (ECM) components bind to Wnt, promoting ligand presentation and stabilizing Wnt-FZD interactions. The ECM regulates FZD receptor aggregation and intracellular transport, thereby enhancing the Wnt signaling pathway. A hypoxic tumor microenvironment can stimulate the overexpression of both FZD receptors and Wnt ligands. Growth factors present in the TME synergistically enhance Wnt/FZD signaling to promote tumor growth and metastasis. Fibroblasts and macrophages in the TME secrete Wnt ligands and ECM components, further promoting FZD signaling.

(Data source: Martinez-Marin D, et al. Front Cell Dev Biol. 2025)
Targeted therapy for FZD
FZD receptors are attractive therapeutic targets due to their crucial role in regulating the Wnt signaling pathway, which is frequently dysregulated in cancer. Aberrant FZD signaling contributes to tumor development, progression, metastasis, and treatment resistance, making these receptors key intervention points in oncology.
Treatment options for the FZD receptor include monoclonal antibodies, small molecule inhibitors, decoy receptors, RNA-based therapies, and emerging gene editing technologies.

(Data source: Martinez-Marin D, et al. Front Cell Dev Biol. 2025)
Tabituximab (OTSA101, ONC-005) is a monoclonal antibody targeting FZD10, developed by OncoTherapy Science. A Phase I clinical trial of OTSA101 for synovial sarcoma in Japan has been successfully completed. Furthermore, the safety and tolerability of radiolabeled OTSA101 in patients with refractory, recurrent, or advanced synovial sarcoma have been confirmed.
Vantictumab is a fully humanized monoclonal antibody that targets FZD and can bind to certain FZD receptors and inhibit the Wnt signaling pathway.
