Tumor necrosis factor receptor family member 18 (TNFRSF18), also known as GITR, is a glucocorticoid-induced TNFR-related protein and the receptor for TNFSF18. It is expressed on a variety of cells, including T cells, natural killer cells, and certain myeloid cells. It plays a role in immune cell signaling, activation, and survival. GITR is a therapeutic target for directly activating effector CD4 and CD8 T cells or depleting GITR-expressing regulatory T cells (Tregs), thereby promoting anti-tumor immune responses.
GITR expression distribution
GITR is mainly expressed in basal keratinocytes, plasma cells, Langerhans cells, NK cells, basal epithelial keratinocytes, T cells, and granulocytes.
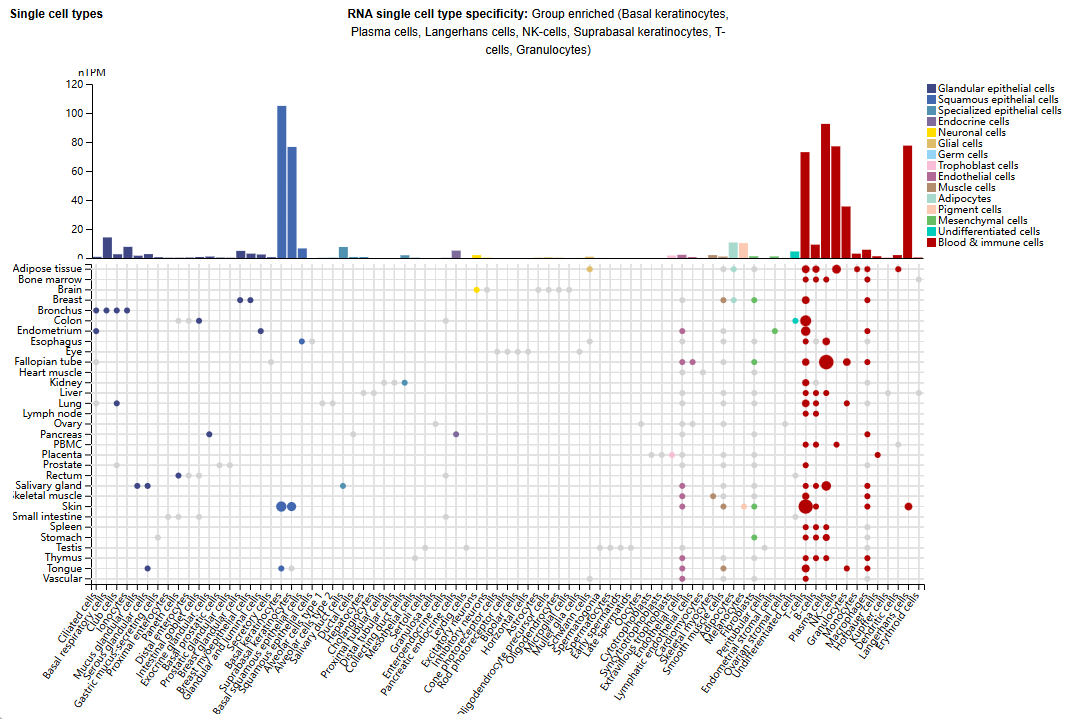
(Data source: Uniprot)
Structure of GITR and its receptor
GITR is a type I membrane protein composed of 241 amino acids, including a signal peptide, an N-terminal extracellular region, a transmembrane domain, and a C-terminal intracellular region. The extracellular region of GITR is composed of three cysteine-rich domains (CDRs) and adopts the typical modular fold of TNF receptors (A1/A2/B1 modules). Human and mouse GITR share a high degree of structural similarity , both exhibiting a curved " C " conformation.
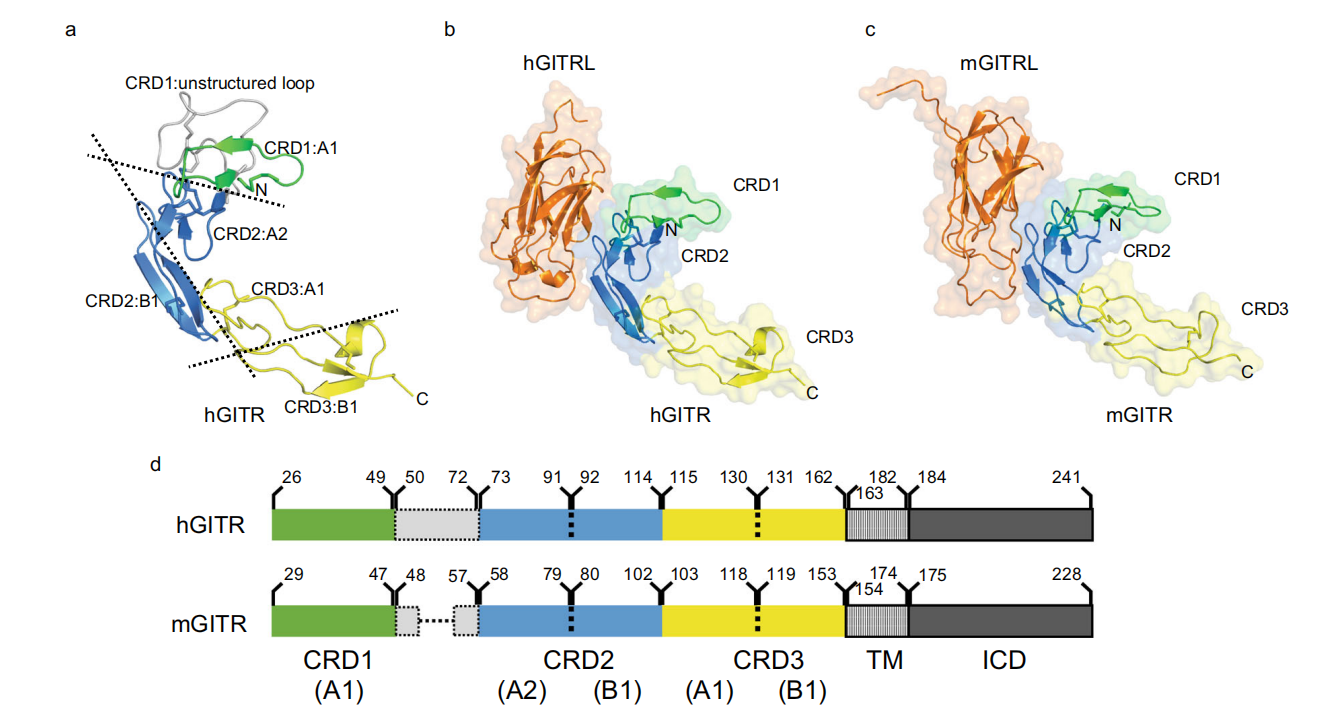
(Data source: Wang F, et al. Nat Commun. 2021)
Function of GITR in immune cells
Triggering GITR with GITR ligand (GITRL) or agonistic anti-GITR antibodies triggers distinct responses in effector T cells and Treg cells. Triggering GITR promotes the differentiation and proliferation of Th17, Th9, and Tfh cells. GITR binding to effector T cells generates positive co-stimulatory signals and promotes T cell activation and proliferation, whereas GITR activation on Tregs abolishes their suppressive function.
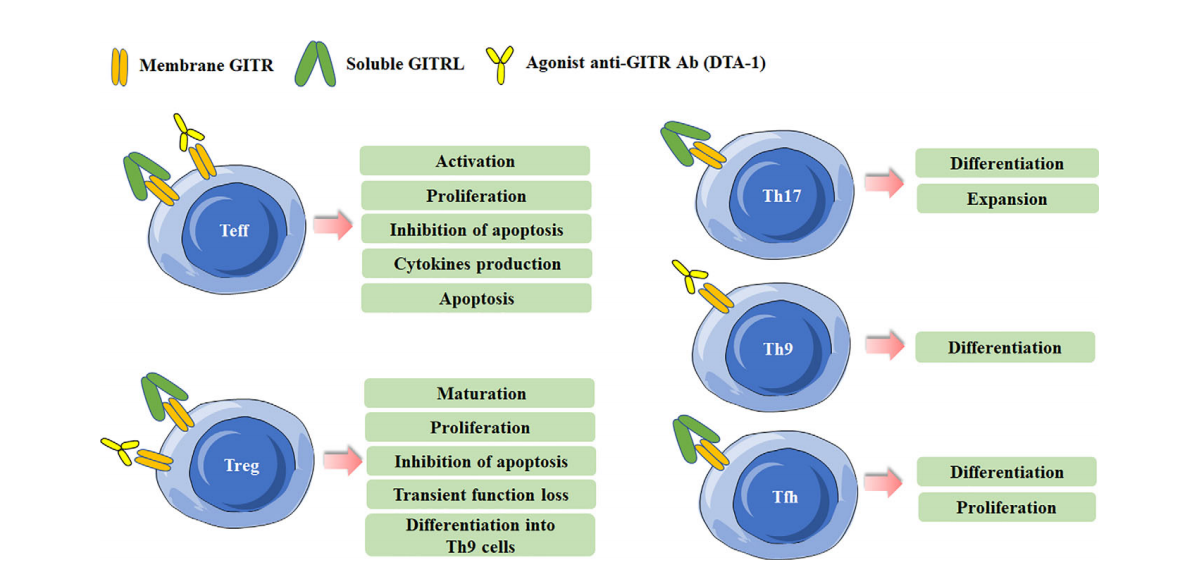
GITR agonists or soluble/membrane GITR trigger multiple effects in DCs and macrophages. GITR is crucial for DC activation and enhances DC-induced T lymphocyte activation. In macrophages, the GITR/GITRL interaction can trigger proinflammatory effects through the activation of GITRL and GITR in macrophages. Binding of GITRL to GITR weakens the suppressive function of MDSCs, thereby promoting the proliferation of CD4+ T cells.

(Data source: Tian J, et al. Front Immunol. 2020)
GITR -targeted therapy
GITR is a co-stimulatory pathway in the immune checkpoint pathway that has attracted much attention in recent years. Monoclonal antibodies targeting GITR have shown great promise as they can enhance the activity of CD8+ and CD4+ effector T cells while suppressing/depleting Tregs.
Leap Therapeutics is developing a GITR agonist currently in Phase II clinical trials. This is an IgG1 aglycosylated monoclonal antibody that enhances NK cell activity. TRX518 has a knockout Fc terminus to prevent the antibody from accidentally targeting cells expressing the GITR receptor, improving the drug's safety. A clinical trial ( NCT-01239134 ) investigating TRX518 demonstrated that targeting GITR was effective without depleting CD4+ and CD8+ naive and memory T cells, B cells, NK cells, monocytes, and macrophages. In the NCT-01239134 trial, GITR monotherapy eliminated Tregs in both peripheral and tumor tissues.
In November 2019, the company announced that it would lower the priority of TRX518 research and development. However, it is currently difficult to find the current status of TRX518 research on the company's official website.
IBI-102 is a monoclonal antibody targeting GITR developed by Innovent Biologics, and was first applied for clinical trials in China in August 2020.
in tumors promotes Treg - mediated immunosuppression and inhibits anti-tumor responses . Studies have found that targeting GITR and combining it with CTLA-4 or PD-1/PD-L1 inhibitors has the potential to improve its efficacy.
GWN-323 is a monoclonal antibody targeting GITR developed by Novartis. NCT02740270 is a Phase I/Ib open-label, multicenter, dose-escalation study of GWN323 (anti-GITR) as a single agent and in combination with PDR001 (anti-PD-1) in patients with advanced solid tumors and lymphomas .
MK-4166, an anti-GITR monoclonal antibody, was found to be well tolerated, and combining GITR agonists with other agents also shows great potential. NCT02132754, a phase 1 trial of MK-4166 alone and in combination with pembrolizumab in patients with advanced malignancies, demonstrated a tolerable safety profile with MK-4166 as monotherapy and in combination with pembrolizumab at doses up to 900 mg; however, objective responses were demonstrated only with the combination therapy. Antidrug antibodies developed in 33% of patients treated with MK-4166 alone or in combination with pembrolizumab, a higher incidence than the 2.6%-6.7% reported with other anti-GITR monoclonal antibodies. Further data are needed to confirm whether the combination of MK-4166 and pembrolizumab may improve efficacy compared with pembrolizumab alone in patients with advanced melanoma who have not received prior ICI therapy.
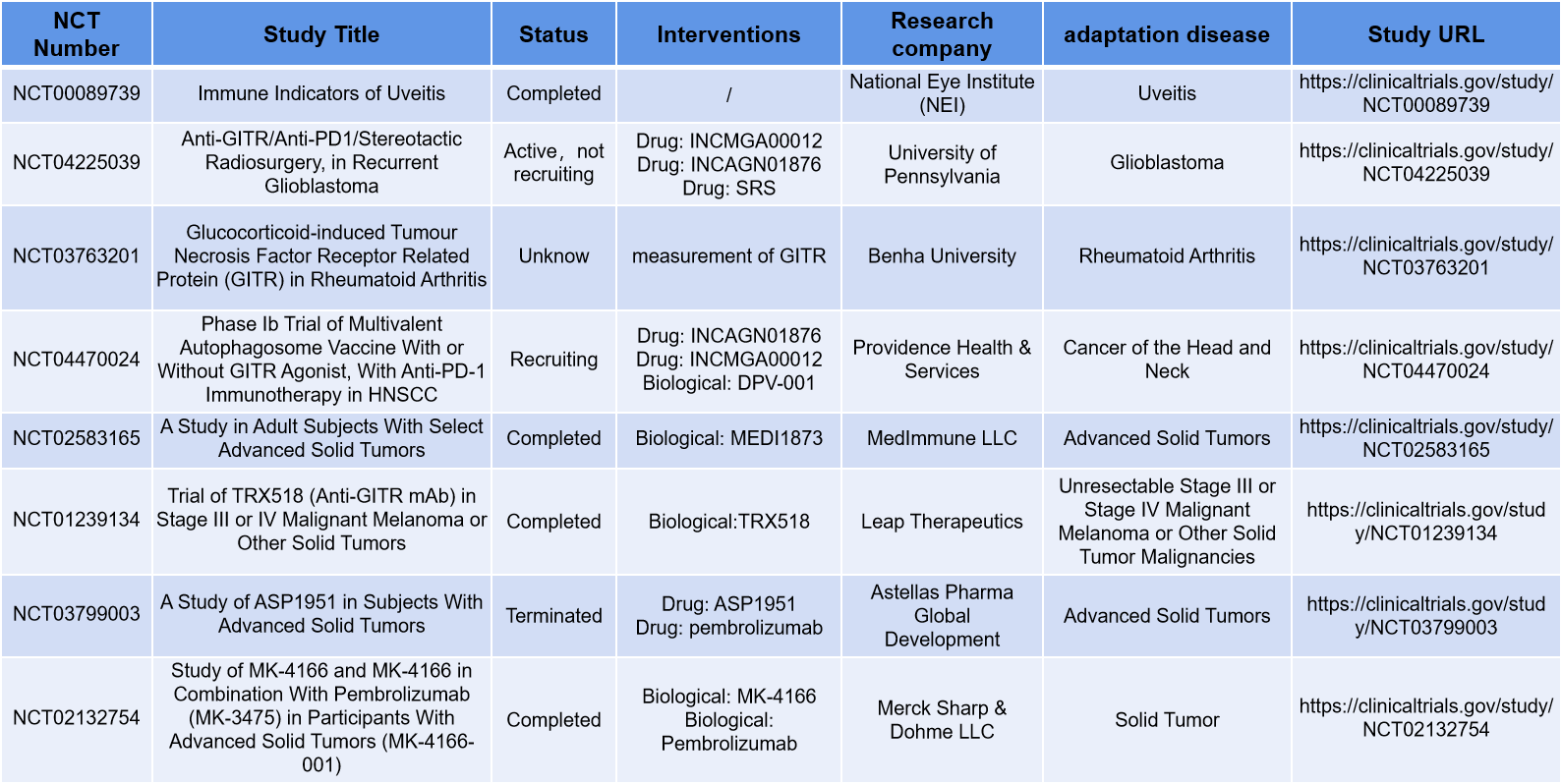
(Data source: https://clinicaltrials.gov/)
