Lymphocyte activation gene 3 (LAG-3), also known as CD223, is an inhibitory receptor highly expressed by exhausted T cells. In the tumor microenvironment, elevated expression of immune checkpoint receptors on exhausted T cells limits their anti-tumor activity. LAG-3 is a major target for cancer immunotherapy.
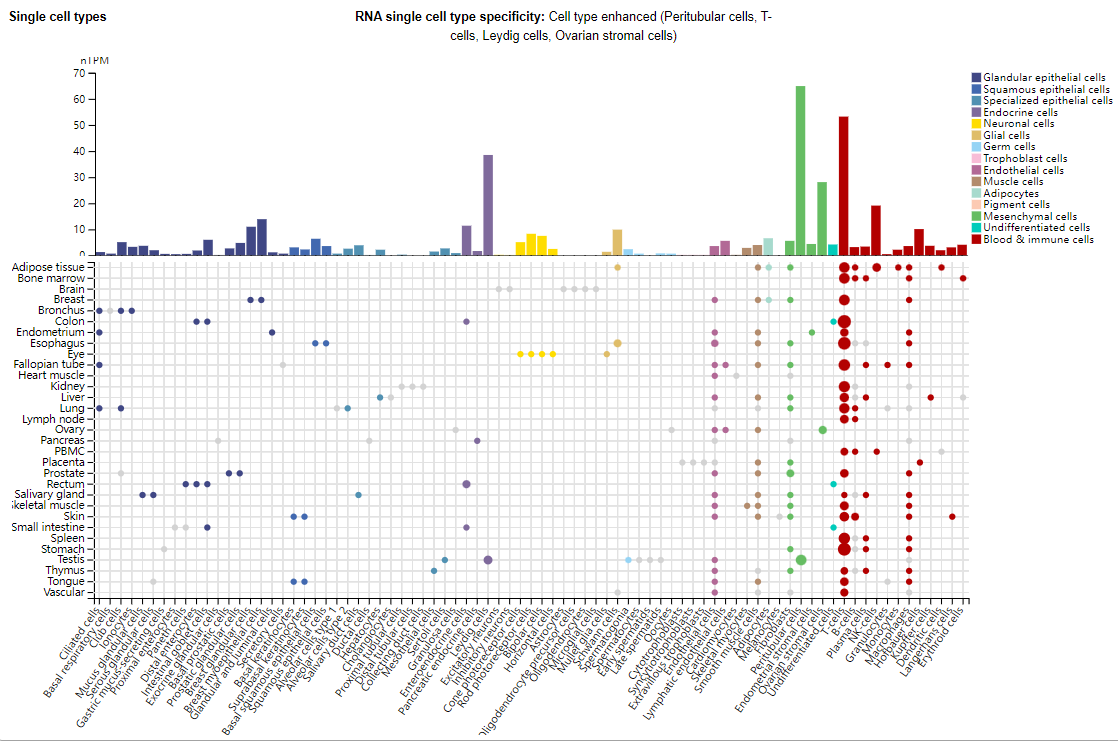
LAG-3 expression distribution
LAG-3 is mainly expressed in T cells, but is also expressed in NK cells, B cells, cytoplasmic dendritic cells, neurons, mesenchymal cells, and endocrine cells.
(Data source: Uniprot)
Structure of LAG-3
LAG-3 is a 55 kDa type I transmembrane protein composed of 525 amino acids. It contains four extracellular immunoglobulin (Ig)-like domains (D1-D4), a linker peptide, and an intracellular region that transmits inhibitory signals to T cells upon binding to MHC class II and other ligands. The four Ig-like domains of hLAG3 and mLAG3 adopt an elongated, strip-like arrangement similar to CD4. D1 is a V-type domain, while D2, D3, and D4 are C2-type domains. D1 and D2 fold as discrete domains, while D3 and D4 form a continuous unit connected by extended β-strands.

(Data source: Mariuzza RA, et al. J Biol Chem. 2024)
LAG-3 ligands
LAG-3 mediates T cell inhibition by interacting with its canonical ligand , major histocompatibility complex (MHC) class II molecules. The interaction between LAG-3 and MHC class II is mediated through loop 2 (103-112 aa) of the LAG-3 D1 domain. Other ligands for LAG-3 include galectin-3 (Gal-3), liver and lymph node sinusoidal endothelial cell C-type lectin (LSECtin), fibrinogen-like protein 1 (FGL1), α-synuclein precursor fibrils (α-syn PFFs), and the T cell antigen receptor (TCR)-CD3 complex.

(Data source: Aggarwal V, Workman CJ, Vignali DAA. Nat Immunol. 2023)
LAG-3 signaling pathway and regulation
LAG-3 binds to MHC class II molecules on cancer cells or antigen-presenting cells (APCs) and inhibits T cell receptor (TCR) signaling, thereby reducing the transcription of key transcription factors (AP-1, NFAT, and NF-κB) . LAG-3 mediates its inhibitory function through a cytoplasmic domain containing four phylogenetically conserved motifs: a membrane-proximal FXXL motif, a putative serine phosphorylation site, a KIEELE motif, and repeated C-terminal EP motifs. LAG-3 interaction with MHC class II molecules transmits two independent inhibitory signals through distinct intracellular motifs: the EP repeats and the FXXL motif. This is the mechanism by which LAG-3 mediates T cell immunoregulation.
LAG-3 activity is also regulated by cleavage of its extracellular domain or shedding mediated by the metalloproteases ADAM10 and ADAM17, forming free LAG-3 (sLAG-3). sLAG-3 has immunostimulatory functions, leading to increased cytokine production (IL-12 and IFN-γ), dendritic cell maturation, and T cell proliferation.

(Data source: Alejandra Martínez-Pérez. Biomark Res.2024)
LAG-3 targeted therapy
LAG-3-targeted therapies primarily involve LAG-3 inhibitors, primarily monoclonal antibodies and bispecific antibodies. LAG-3 is often combined with PD-1 therapy. The combination of PD-1 and LAG-3 blockers has been shown to promote anti-tumor immune responses in vitro and in vivo in a variety of tumors, including melanoma, renal cell carcinoma, and chronic lymphocytic leukemia. Currently, numerous drugs in clinical development are in clinical trials for the treatment of both solid tumors and hematologic malignancies.

Relatlimab (BMS-986016, Bristol-Myers Squibb) is a first-in-class anti-LAG-3 blocking antibody that represents the third approved ICB therapy for cancer patients and is currently in Phase 3 clinical studies.
Fianlimab and favezelimab are currently being tested in combination with anti-PD-1 blocking antibodies in Phase III clinical trials for the treatment of melanoma, non-small cell lung cancer (NSCLC), and colorectal cancer. In patients with advanced melanoma, the combination of fianlimab and cemiplimab (an anti-PD-1 monoclonal antibody) has resulted in higher clinical response rates than either agent alone.
Teboteliab, a bispecific antibody targeting PD-1 and LAG-3, is undergoing a Phase III study in patients with HER2-positive gastric cancer and other solid tumors.
FS118, a tetravalent anti-LAG-3/anti-PD-L1 antibody, was evaluated in a dose-escalation and expansion clinical trial in patients with advanced/metastatic disease who had previously received anti-PD-1/PD-L1 monoclonal antibody therapy. At the study's endpoint, approximately 55% of patients who had developed acquired resistance to prior ICB therapy demonstrated stable disease, strengthening the case for dual LAG-3/PD-L1 blockade as an alternative for rechallenging resistant tumors.


(Data source: Alejandra Martínez-Pérez. Biomark Res.2024)
