Transmembrane mucins are major components of the gastric mucosal barrier, comprising a family of high-molecular-weight glycoproteins that are expressed by specialized epithelial cells as secreted or membrane-bound mucins. MUC17, also known as MUC3, is a member of the mucin family and is expressed on the apical membrane of normal gastrointestinal epithelial cells. Loss of apical localization of MUC17 is characteristic of malignant tissue. Gene expression analysis of The Cancer Genome Atlas (TCGA) and other research data revealed that MUC17 is overexpressed in 23.3%-52.2% of gastric cancer patients, with expression levels significantly higher in gastric cancer tissue than in adjacent normal tissue, suggesting that MUC17 is an attractive target for gastric cancer treatment.
Expression distribution and tissue specificity of MUC17
MUC17 is expressed almost exclusively in the intestine. Its expression levels are particularly high in the duodenum and transverse colon . The gene is expressed in mature absorptive cells of the small intestinal villi. No expression of the gene has been detected in goblet cells. The gene is highly expressed (at the protein level) in pancreatic adenocarcinoma tissue. Expression of the gene has not been detected in normal pancreas, pancreatitis, or cell lines derived from other cancers.

(Data source: uniprot )
Features of MUC17:
Interaction with PDZK1: MUC17 interacts with the PDZK1 protein through its C-terminus, and this interaction is important for the correct localization of MUC17 in cells. PDZK1 may act as a scaffold protein, helping MUC17 find its proper location on the cell membrane, allowing it to better perform its function.
Maintaining mucosal surface homeostasis: MUC17 may play a role in maintaining mucosal surface homeostasis. The mucosal surface is a critical barrier between the human body and the external environment. MUC17, through its domains interacting with other molecules, helps maintain mucosal integrity and regulates secretory and absorptive functions, thereby maintaining normal physiological conditions on the mucosal surface.
The structure of MUC17:
MUC17 was first discovered and characterized in 2002. It is a 4493-amino acid type I transmembrane protein encoded by the MUC17 gene. It consists of a transmembrane segment, an intracellular segment, and an extracellular segment. From the amino terminus, it contains a mucin-like structure (tandem repeats), an EGF (epidermal growth factor)-like region, an N-glycosylation region, a second EGF-like region, a hydrophobic transmembrane segment, and a cytoplasmic shuttle terminus. The MUC17 protein contains five key domains: a glycosylation domain, an EGF-like domain, a SEA module domain, a transmembrane domain, and a cytoplasmic domain. The extracellular glycosylation domain forms the glycoprotein and mediates interactions; the carboxy-terminal EGF-like domain is involved in signaling and adhesion; the SEA module domain is hypothesized to be involved in glucose metabolism or recognition; the transmembrane domain provides membrane anchoring; and the cytoplasmic domain contains phosphorylation sites that regulate its activity and downstream signaling.
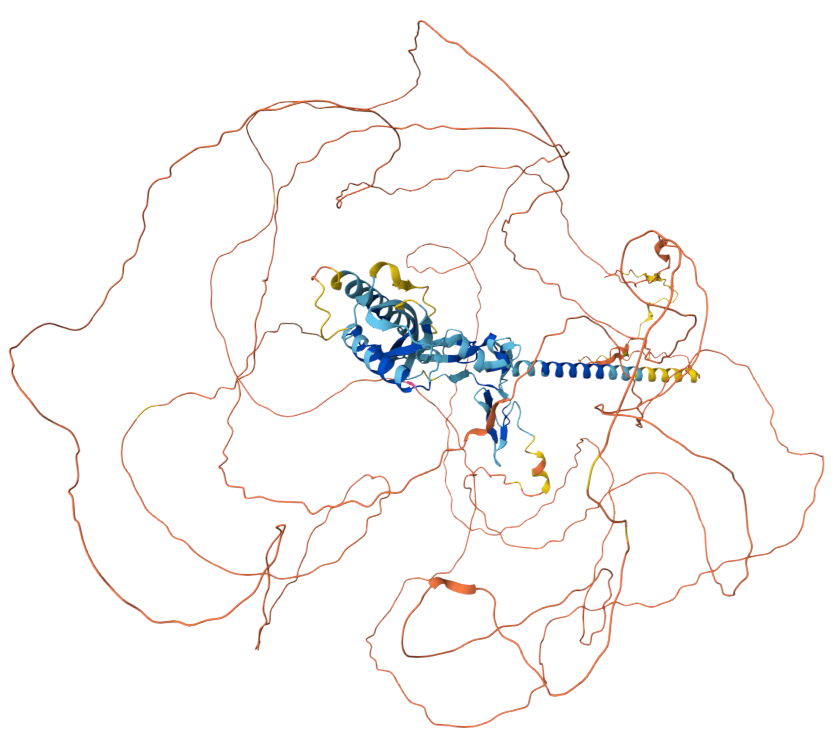
(Data source: AlphaFold)
MUC17 signaling pathway and immune regulation:
Tumor-associated MUCs promote an immunosuppressive microenvironment by masking Toll-like receptors (TLRs) on antigen-presenting cells (APCs) or inhibiting synapse formation between cytolytic natural killer (NK) cells and cancer cells. Mucins increase immune tolerance by enhancing reprogramming toward a regulatory T cell (T-CTL) phenotype and reducing the expression of activation markers on dendritic cells (DCs). Furthermore, mucins activate tumor-specific humoral and cellular immune responses in the presence of adjuvants or TLR agonists, including antibody-dependent cellular cytotoxicity (ADCC), sialyl Tn antigen (sTn), cytotoxic T lymphocytes (CTLs), interleukins (ILs), transforming growth factor β1 (TGF-β1), and clusters of differentiation (CDs).

(Data source: Bhatia R, et al. Cancer Metastasis Rev. 2019)
MUC17 and disease:
Loss of MUC17 apical localization is a characteristic finding of malignant tissue. Gene expression analysis of data from The Cancer Genome Atlas (TCGA) and other studies revealed that MUC17 is overexpressed in 23.3%-52.2% of gastric cancer patients, with expression levels significantly higher in gastric cancer tissue than in adjacent normal tissue, suggesting that MUC17 is an attractive therapeutic target for gastric cancer. Related studies have shown that MUC17 can inhibit human gastric cancer by limiting inflammatory responses . Inflammatory factors induce MUC17 expression by upregulating CDX1. MUC17 relies on its conserved epidermal growth factor domain and downstream sequences to interact with myosin-9, forming a continuous regulatory feedback loop between myosin-9, p53, and RhoA, which then activates p38 to negatively regulate the NFκB pathway in gastric cancer cells. Inflammatory factors promote cancer cell growth by activating the NFκB pathway, so MUC17 can limit inflammatory responses.
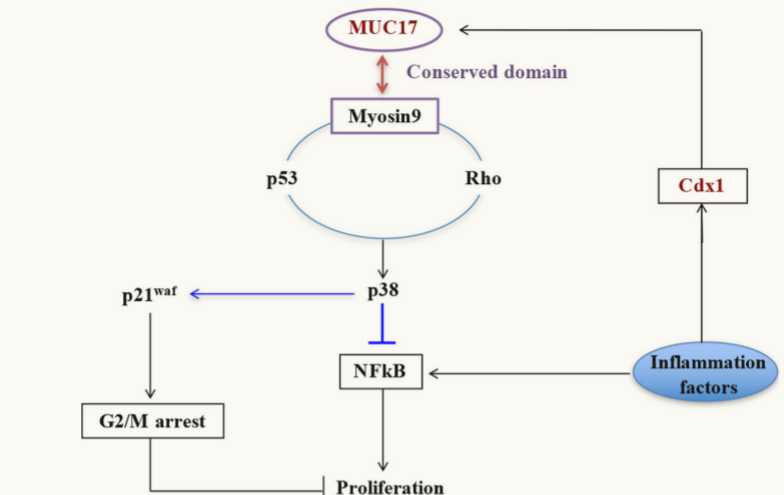
(Data source: J Exp Clin Cancer Res. 2019.)
Targeted therapy of MUC17
AMG199 is a bispecific antibody targeting MUC17/CD3, developed by Amgen. Composed of two single-chain variable fragments, one targeting the tumor-associated antigen (TAA) human mucin 17 (MUC17) and the other targeting human CD3, a T-cell surface antigen found on T lymphocytes, it has potential anti-tumor activity. Upon administration, AMG199 binds to CD3 on T cells and MUC17 expressed on tumor cells, promoting cross-linking between the two cells and inducing a potent cytotoxic T lymphocyte (CTL) response against MUC17-expressing tumor cells.

(Data from Wen D, et al. Mol Pharm. 2025)
SCR9171 is a bispecific T cell engager (BiTE) developed by Simcere Pharmaceutical Group Limited that simultaneously targets CD3 and MUC17. Through this dual-target design, the drug bridges tumor cells and T cells, enabling precise immune killing.
Its primary mechanisms of action are: 1. Precise tumor targeting: The anti-MUC17 arm binds to the MUC17 protein, which is highly expressed on the surface of gastric cancer cells; 2. T cell activation: The anti-CD3 arm binds to the CD3 receptor on the surface of T cells, activating T cells and inducing their proliferation and differentiation. Currently, its efficacy and safety data are based on preclinical models and require further clinical validation.
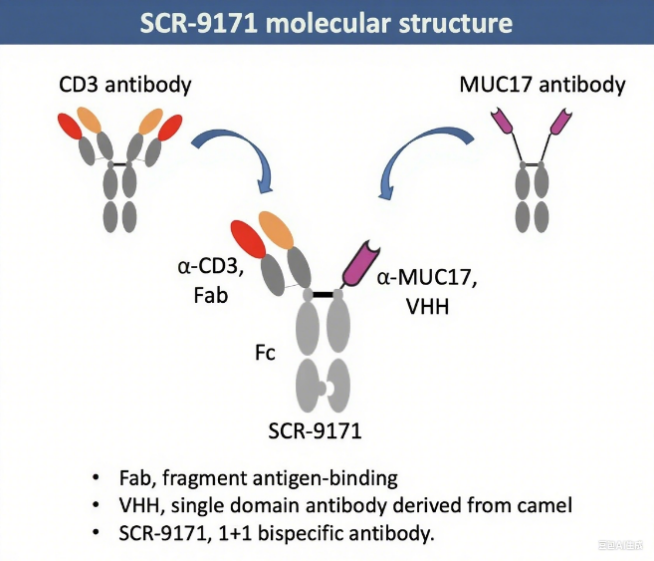
(Data source: (Simcere Pharmaceutical Group Limited official website)
SSS59 is a trispecific antibody drug targeting MUC17/CD3/CD28 developed by 3SBio Inc. It is currently in Phase I clinical trial for advanced gastric cancer and gastroesophageal junction (GEJ) cancer (NCT06944444). Its primary mechanism of action is similar to that of AMG199 and SCR9171 , but with the addition of a CD28 target, enabling it to provide a co-stimulatory signal ("second signal"), promoting full T cell activation, proliferation, and persistence, overcoming the T cell exhaustion associated with traditional bispecific antibodies (such as those targeting only CD3 ) . The first Phase I clinical trial in China was approved on April 27, 2025.
HDM2012 is an ADC drug targeting MUC17 that is coupled with a topoisomerase I inhibitor and developed by Huadong Medicine.
Its primary mechanism of action is as follows: 1. Targeted Recognition and Binding: HDM2012, through its anti- MUC17 monoclonal antibody, specifically recognizes the MUC17 protein overexpressed on the surface of tumor cells. 2. Internalization and Toxin Release: Binding of the antibody to MUC17 triggers antigen-mediated endocytosis, leading to internalization of the ADC by tumor cells. Within the cell, the linker is cleaved, releasing the topoisomerase I inhibitor (toxin payload). This toxin inhibits topoisomerase I , which is essential for DNA replication , causing DNA double-strand breaks and triggering apoptosis in tumor cells. 3. Bystander Effect Enhances Cytotoxicity: The released toxin small molecule has transmembrane penetration and can diffuse into neighboring tumor cells (even those with low MUC17 expression levels), amplifying its cytotoxic effect within the tumor microenvironment. This property is particularly important for the heterogeneous cell populations found in solid tumors.
